🔄
The Ortho Effect in Benzoic Acids
A fascinating phenomenon where ortho-substituted benzoic acids defy conventional electronic effects, revealing the intricate interplay between steric hindrance, resonance, and through-space interactions.
Introduction: The Paradox
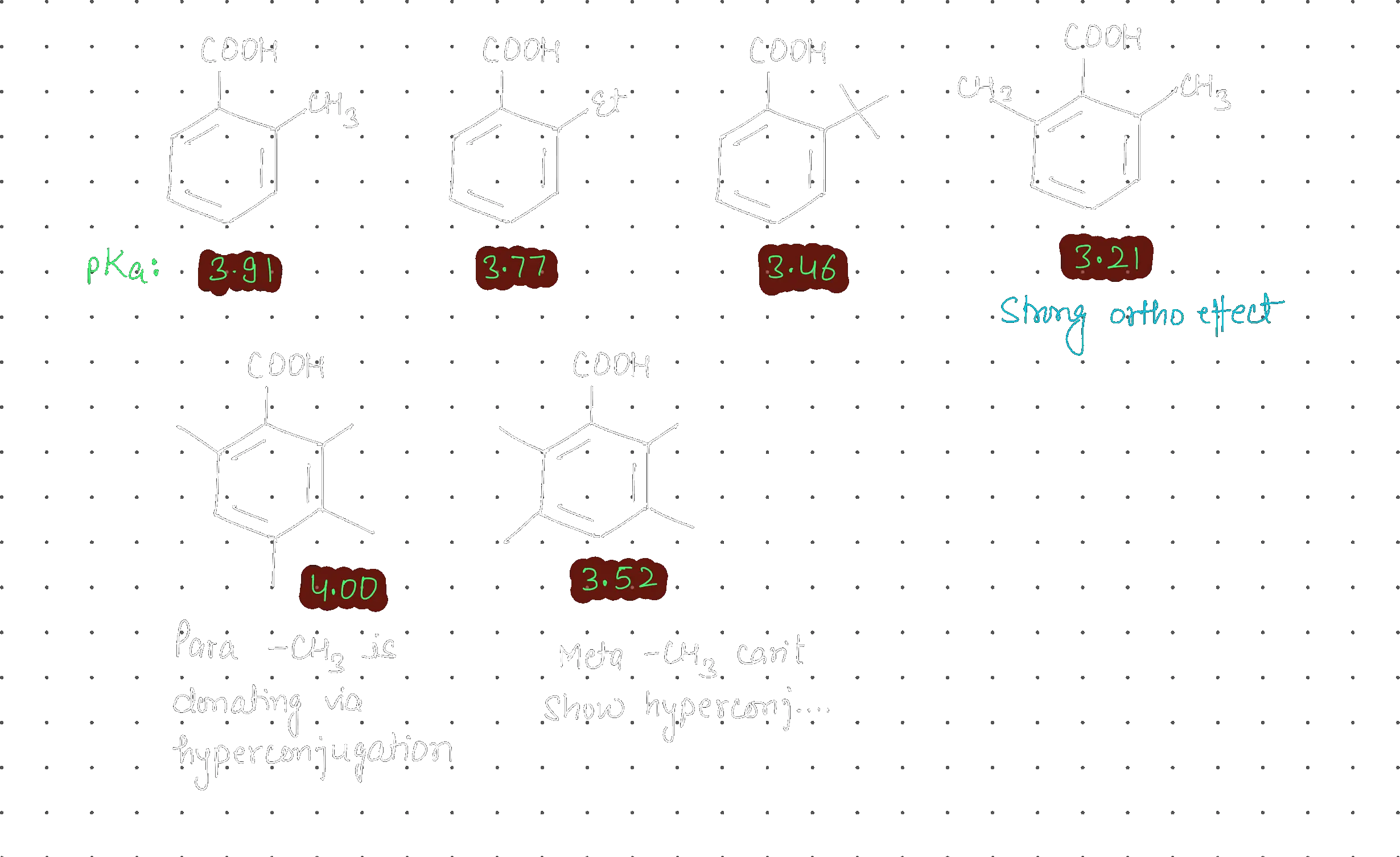
In organic chemistry, electron-donating groups (EDGs) like alkyl groups (–CH₃, –C₂H₅) typically decrease acidity by destabilizing the conjugate base. This holds true for para and meta positions in benzoic acid derivatives. However, when these same alkyl groups occupy the ortho position, they paradoxically increase acidity. This counterintuitive behavior is known as the ortho effect, arising from two primary mechanisms: steric inhibition of resonance (where bulky ortho groups twist the –COOH out of plane, disrupting resonance stabilization) and field effects through space (electrostatic interactions between the ortho substituent and the carboxyl group).
Experimental Evidence
| Compound | pKa | Observation |
|---|---|---|
| Benzoic acid (reference) | 4.20 | Baseline |
| ortho-Methylbenzoic acid | 3.91 | More acidic despite –CH₃ being EDG |
| ortho-Ethylbenzoic acid | 3.79 | Bulkier group → stronger ortho effect |
| para-Methylbenzoic acid | 4.38 | Less acidic (normal EDG behavior) |
| meta-Methylbenzoic acid | 4.27 | Slightly less acidic (weak +I effect) |
💡 Key observation: ortho-CH₃ (pKa = 3.91) is more acidic than benzoic acid (pKa = 4.20), while para-CH₃ (pKa = 4.38) is less acidic. This reversal cannot be explained by electronic effects alone.
Visualizing the Ortho Effect
Mechanism 1: Steric Inhibition of Resonance
When a bulky substituent (like –CH₃ or –C₂H₅) is placed at the ortho position, it experiences steric repulsion with the carboxyl group (–COOH), forcing the –COOH to twist out of the benzene ring plane. In a planar conformation, the carboxyl group can engage in resonance with the benzene ring, delocalizing and stabilizing the negative charge of the carboxylate ion (–COO⁻). However, when twisted out of plane, this resonance is disrupted. The key insight is that the neutral acid is destabilized more than the conjugate base: the twisted –COOH in the neutral molecule loses resonance stabilization with the ring, while the carboxylate anion's negative charge is already delocalized within the –COO⁻ group itself (between the two oxygen atoms), making it less dependent on ring resonance. This differential destabilization shifts the equilibrium toward deprotonation, increasing acidity.
📌 Key Principle: Acidity is determined by the relative stability of the acid and its conjugate base. If the neutral acid is destabilized more than the anion, acidity increases.
Mechanism 2: Field Effect Through Space
Even when the ortho substituent is not directly conjugated with the –COOH group (due to twisted geometry), it can still interact through electrostatic field effects. The ortho substituent's close spatial proximity to the –COOH group allows it to stabilize or destabilize the developing negative charge on the carboxylate oxygen through through-space interactions. For alkyl groups, despite being typically electron-donating via the inductive effect (+I), their polarizable electron cloud can interact with the carboxylate oxygen to provide partial stabilization of the negative charge through polarization and dispersion forces. This contributes to the overall acidifying effect, though steric inhibition of resonance is generally the dominant factor.
Position Dependence: ortho vs. meta vs. para
The ortho effect is position-specific, with the same substituent producing vastly different effects depending on its location:
- ortho Position: Maximum steric hindrance forces –COOH out of plane. Field effects are strongest due to close proximity. For alkyl groups, acidity increases (pKa ~3.9).
- meta Position: No direct steric clash. Electronic effects are weak because resonance cannot directly reach the carboxyl group. Alkyl groups show weak +I effect, slightly decreasing acidity (pKa = 4.27).
- para Position: No steric hindrance. Alkyl groups donate electrons through resonance and inductive effects, destabilizing the carboxylate anion and decreasing acidity (pKa = 4.38) — the expected behavior for EDGs.
Acidity trend for alkyl substituents: ortho (pKa ~3.9) > benzoic acid (4.20) > meta (4.27) > para (4.38)(Lower pKa = more acidic)
Ortho Effect with Electron-Withdrawing Groups (EWGs)
For electron-withdrawing groups (like –NO₂, –Cl, –CF₃), the ortho position is always the most acidic, but for different reasons than alkyl groups. EWGs withdraw electrons through σ-bonds (inductive effect, strongest at ortho due to proximity), through π-conjugation (resonance effect, especially –NO₂, directly stabilizing the carboxylate anion), and through electrostatic attraction (field effect, where electronegative atoms stabilize the nearby negative charge on –COO⁻). For example, ortho-nitrobenzoic acid (pKa ≈ 2.2) is significantly more acidic than benzoic acid (pKa = 4.20) and even more acidic than para-nitrobenzoic acid (pKa ≈ 3.4), due to these combined effects.
Summary
- The ortho effect causes ortho-substituted benzoic acids to exhibit anomalous acidity trends.
- Steric inhibition of resonance is the primary mechanism: bulky ortho groups twist –COOH out of plane, destabilizing the neutral acid more than the anion.
- Field effects through space provide additional stabilization/destabilization of the carboxylate ion.
- For alkyl groups (EDGs): ortho > H > meta > para (in terms of acidity).
- For EWGs: ortho is always most acidic due to combined inductive, resonance, and field effects.