General Organic Chemistry MCQ for JEE Main Revision
A 30-question revision quiz on General Organic Chemistry (GOC) — every question is a star-marked JEE Main previous-year question (PYQ) hand-picked by Paaras Sir's team. Covers electronic effects (inductive, resonance, hyperconjugation), acidity and basicity, carbocation/carbanion/free-radical stability, structural and stereo-isomerism, aromaticity, and IUPAC nomenclature. Built for JEE Main, JEE Advanced, NEET and BITSAT aspirants.
What you'll revise
- ▸Apply electronic effects (Inductive, Mesomeric, Hyperconjugation) to predict stability and reactivity
- ▸Compare acidity and basicity across organic compound families using reasoning chains
- ▸Rank reaction intermediates (carbocation, carbanion, free radical) by stability
- ▸Assign IUPAC names and identify isomerism (structural, geometrical, optical)
- ▸Recognise aromaticity using Hückel's rule across charged and neutral systems
Questions (30)
Click an option — correct picks go green, wrong picks go red.The IUPAC name for the following compound is:
2,5-Dimethyl-6-oxo-hex-3-enoic acid
Step 1: Determine Principal Functional Group Between the carboxylic acid () and the aldehyde (), the carboxylic acid has higher priority. The parent chain must start from the acid group.
Step 2: Numbering and Chain
- Numbering begins at .
- The longest chain containing the double bond and the other carbonyl has 6 carbons Hex-3-enoic acid.
Step 3: Substituents
- C2: Methyl group.
- C5: Methyl group.
- C6: Carbonyl group (aldehyde carbon) Named as "oxo" when part of the chain.
Step 4: Final IUPAC Name 2,5-Dimethyl-6-oxohex-3-enoic acid.
Correct stability order of resonance structures of CH₃-CH=CH-CHO:
III > II > I
Resonance Stability Principles

How many compounds show inductive, mesomeric AND hyperconjugation effects?
4
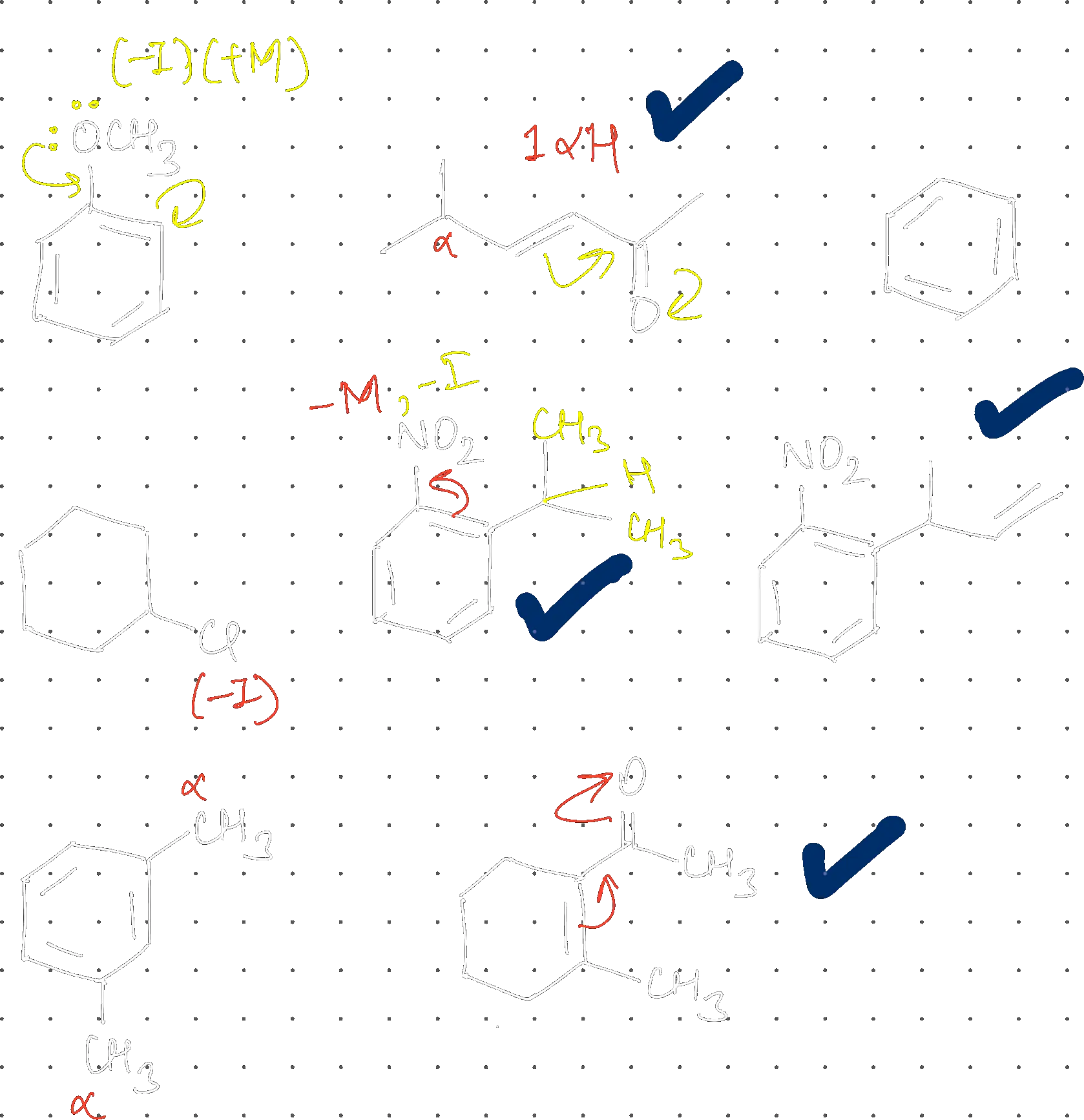
Increasing order of stability of resonance structures:
(C) < (A) < (B) < (D)
Match List-I with List-II (Electronic effects):
(A)-(IV), (B)-(III), (C)-(I), (D)-(II)
Step 1: Matching Effects to Mechanisms
- (A) Aniline resonance: Electrons move into the ring. This is effect. (A-IV)
- (B) Cyclohexene + : Double bond electrons migrate to the attacking reagent (). This is effect. (B-III)
- (C) Cyclohexene + : Double bond electrons move away from the attacking reagent (). This is effect. (C-I)
- (D) Nitrobenzene resonance: Electrons move toward the substituent. This is effect. (D-II)
Step 2: Correct Match (A)-(IV), (B)-(III), (C)-(I), (D)-(II).
The decreasing order of electron density on the ring is:
(IV) > (I) > (III) > (II)
Strategy: Electron density on the benzene ring is increased by electron-donating groups () and decreased by electron-withdrawing groups ().
Step 1: Analyze the substituents
- (IV): The nitrogen of the amide/amine is directly attached to the ring and bears a negative charge (anion). This is a very strong effect, making the ring extremely electron-rich.
- (I): Standard saturated ring fused to benzene. No strong M-effects.
- (III): Carbonyl group () attached to the ring withdraws electron density.
- (II): Two carbonyl groups () attached to the ring withdraw density even more effectively.
Step 2: Determine Order Decreasing density: IV > I > III > II
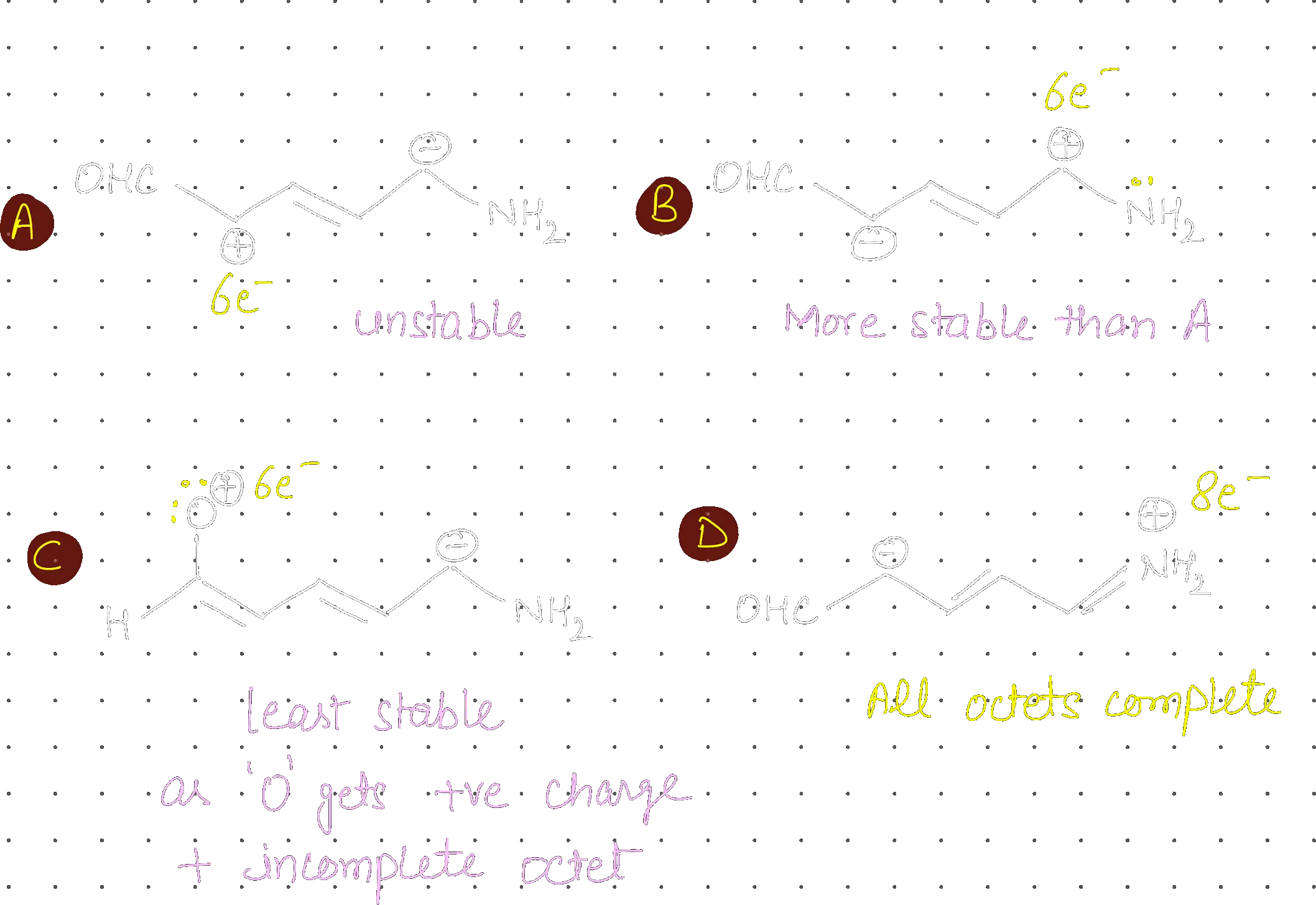
In the following sets of resonating structure, label the major contributors towards resonance hybrid.
II, II, II, II
Strategy: Major resonating contributors are determined by: (1) Completeness of octets, (2) Greater number of covalent bonds, (3) Lack of charge separation, and (4) Negative charge on more electronegative atoms.
Step 1: Analyze each set
- (P): (II) is major because both C and N have complete octets and there are more bonds compared to (I).
- (Q): (II) is major because oxygen is more electronegative than carbon and carries the negative charge more stably than the carbanion in (I).
- (R): (II) is major because all atoms (including Carbon and Nitrogen) have complete octets, whereas in (I) the carbocation has an incomplete octet.
- (S): (II) is major because it has no charge separation on the ring and all atoms have complete octets.
Step 2: Conclusion The major contributors are II, II, II, II.
Select the correct option related to stability of following structures.
c
Strategy: Stability of ions is determined by resonance, inductive effects, and charge distribution.
Step 1: Analyze Option (C) Anion (1) is the phenoxide ion (). Anion (2) is the benzoate ion (). Benzoate is much more stable than phenoxide because the negative charge is delocalized over two equivalent oxygen atoms in the carboxylate group, whereas in phenoxide it is delocalized into the less electronegative carbon atoms of the ring.
Step 2: Verify other options
- (A): Cation on carbonyl carbon vs vinylic. (B): Pyridinium derivative vs O-anion. (D): Formate vs Trifluoroacetate. Option (C) correctly states Phenoxide < Benzoate.
Hyperconjugation observed in
d
Strategy: Hyperconjugation requires (1) a vacant p-orbital (carbocation), a partially filled p-orbital (radical), or a orbital (alkene), and (2) adjacent -bonds that are not constrained by geometry (Bredt's Rule).
Step 1: Analyze the species
- (A): Tertiary radical at a bridgehead-like or constrained position with availabe -H.
- (B): Triphenylmethyl radical. Stabilized purely by resonance, no -H available for hyperconjugation.
- (C): Radical at a bridgehead. According to Bredt's Rule, hyperconjugation is inhibited because the required planar transition state for the double bond cannot be achieved.
- (D): Allylic radical system where -hydrogens are available on the adjacent carbons of the rings.
Step 2: Conclusion Hyperconjugation is most clearly and effectively observed in the systems with unconstrained -H like in Option (D).
Correct order of acidic strength for following compounds is:
(iii) > (i) > (iv) > (ii)
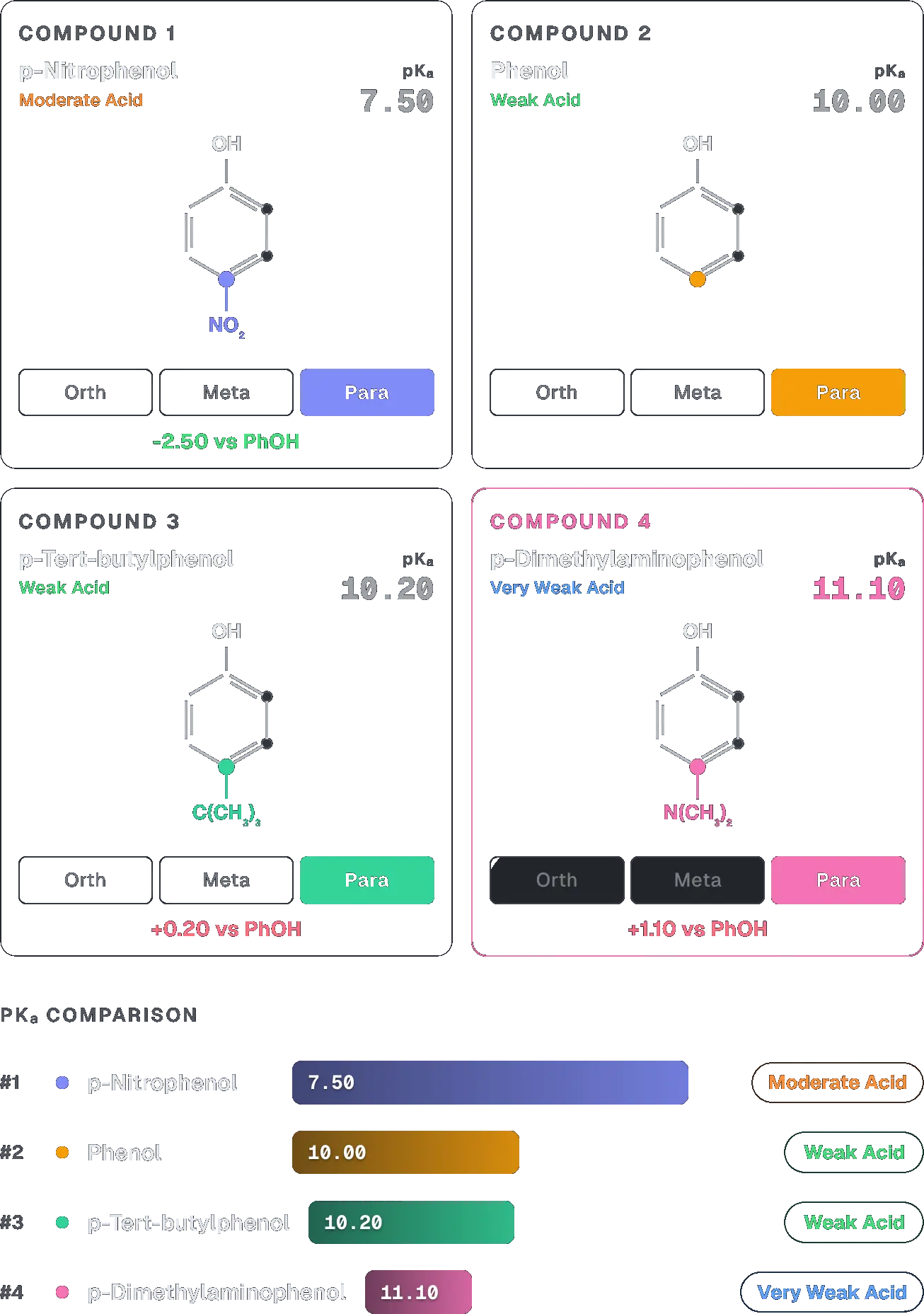 Check the Acidity Lab in the Organic Chemistry Hub page under revision tools on www.canvasclasses.in to visualise such comparisons.
Check the Acidity Lab in the Organic Chemistry Hub page under revision tools on www.canvasclasses.in to visualise such comparisons.
The least acidic compound, among the following is:
D
Step 1: Comparing Acidic Strength Acidity depends on the stability of the conjugate base.
- Phenol: Resonace stabilized phenoxide ion.
- Sulfonic Acid (): Highly stabilized by resonance with three oxygens.
- Carboxylic Acid (): Stabilized by resonance with two oxygens.
- Terminal Alkyne (): The negative charge on -hybridized carbon is less stable than resonance-stabilized oxygens.
Step 2: Identifying the Least Acidic Terminal alkynes () are significantly less acidic than carboxylic acids (), phenols (), or sulfonic acids ().
The order of relative stability of the contributing structure is:
I > II > III

Which will undergo deprotonation most readily in basic medium?
A only
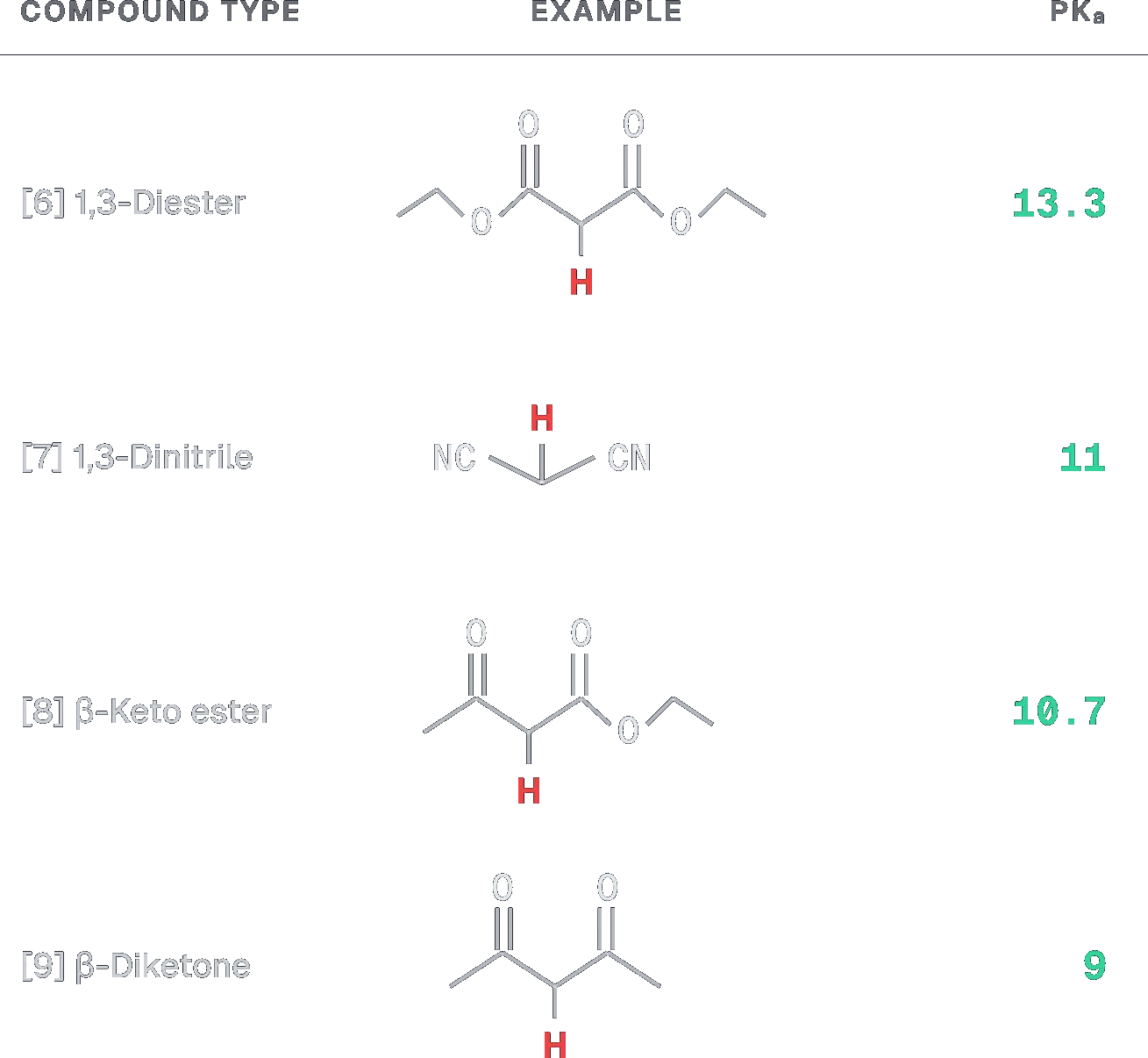 Step 1: Acidity of Active Methylene Protons
Deprotonation occurs most readily where the conjugate base is most stable.
Step 1: Acidity of Active Methylene Protons
Deprotonation occurs most readily where the conjugate base is most stable.
- Structure A: 2,4-pentanedione. The active methylene hydrogens are flanked by two methyl ketones. The and effects of two keto groups stabilize the anion.
- Structure B: Dimethyl malonate. Esters are less electron-withdrawing than ketones due to the effect of the alkoxy group.
- Structure C: Ethyl acetoacetate. One ketone and one ester.
Step 2: Conclusion Structure A has two ketone groups, which provide the strongest stabilization to the conjugate base compared to esters.
Increasing order of basicity:
(B) < (A) < (D) < (C)
Step 1: Comparing Nitrogen Basicity Basicity depends on the availability of the nitrogen lone pair.
- (C) Pyrrolidine: nitrogen, localized lone pair. Most basic.
- (D) Imidazole: One nitrogen lone pair is localized (), the other is aromatic. Still fairly basic as the nitrogen can accept a proton.
- (A) Pyridine: nitrogen lone pair is localized but in a more electronegative orbital than .
- (B) Pyrrole: lone pair is involved in aromaticity (6π system). Least basic.
Step 2: Conclusion Order: (C) > (D) > (A) > (B).
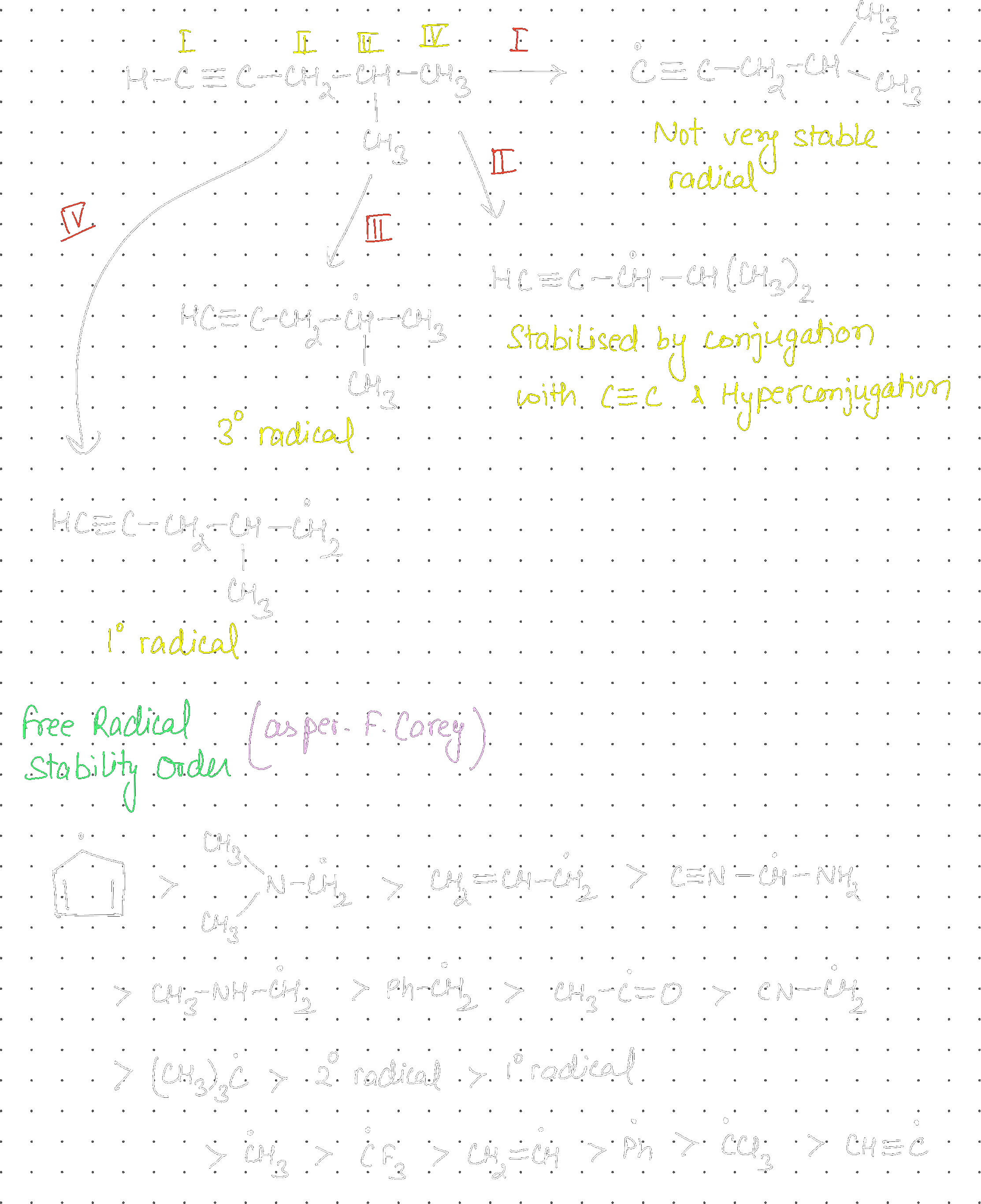
Consider the following compound (X):
The most stable and least stable carbon radicals, respectively, produced by homolytic cleavage of corresponding C-H bond are:
II, I
The correct order of stability of following carbocations is:
C > A > B > D
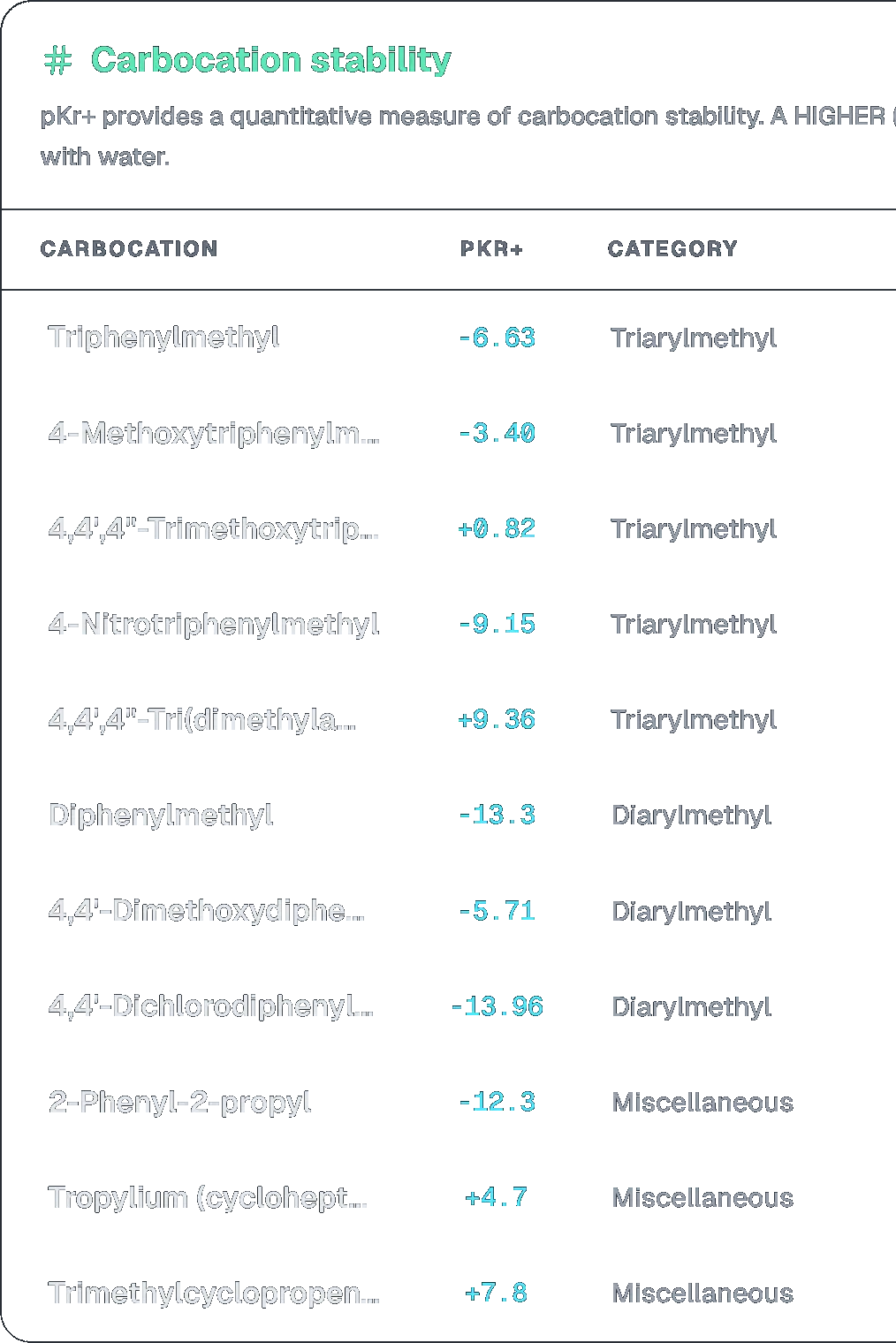 pKr+ provides a quantitative measure of carbocation stability. A HIGHER (more positive) value indicates GREATER stability.
pKr+ provides a quantitative measure of carbocation stability. A HIGHER (more positive) value indicates GREATER stability.
- Tropylium Cation (C): It is aromatic ( electrons, where ), giving it exceptional stability.
- Triphenylmethyl Cation (A): The positive charge is delocalized over 3 phenyl rings.
- Diphenylmethyl Cation (B): The charge is delocalized over 2 phenyl rings.
- Sec-butyl Cation (D): Only stabilized by inductive effect and hyperconjugation.
Step 2: Stability Order Stability decreases in the order: Tropylium > Triphenylmethyl > Diphenylmethyl > Secondary alkyl.
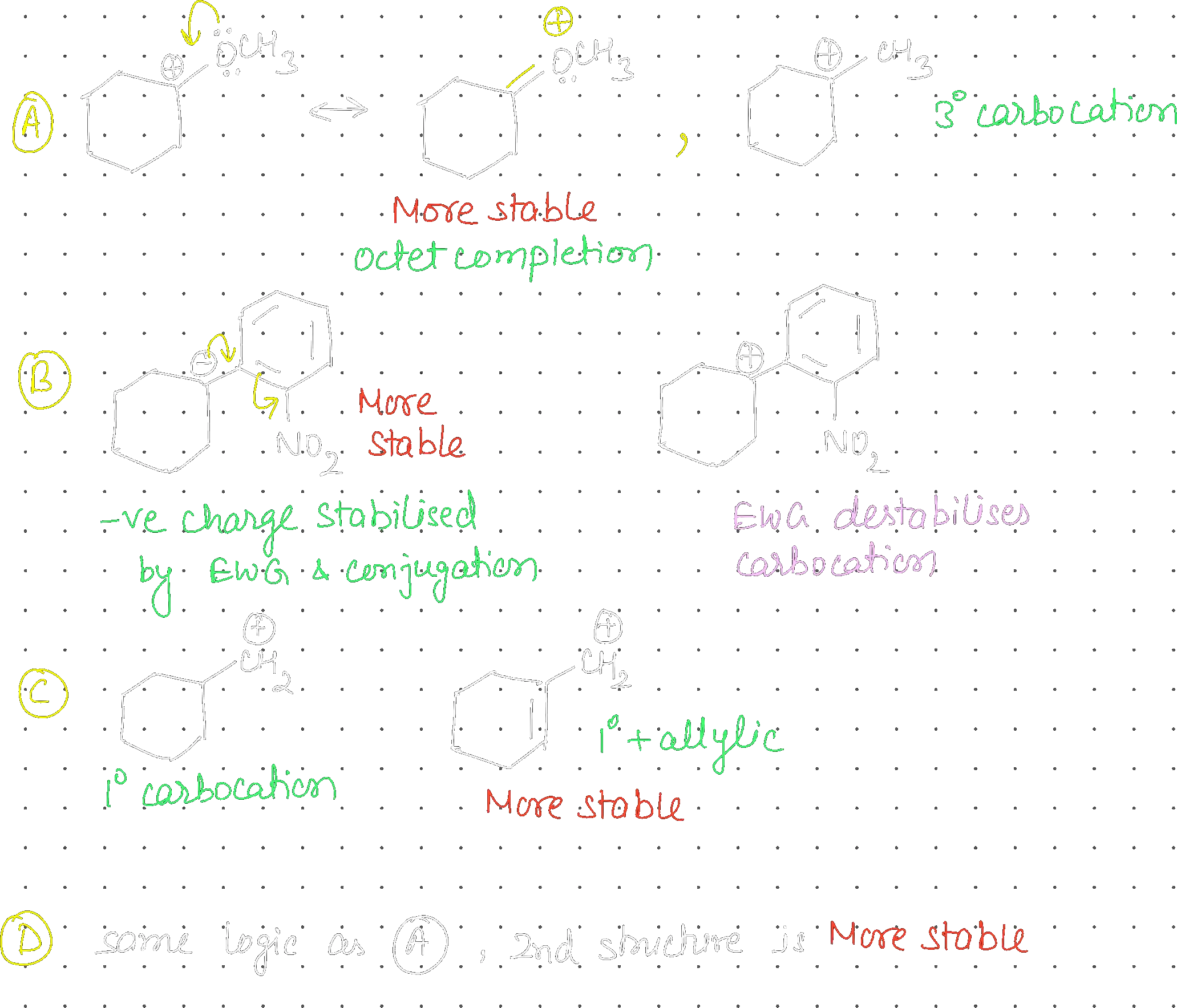
In which pairs, the first ion is more stable than the second?
(A) & (B) only
Which one of the carbocations from the following is most stable?
Option (c)
Identifying Most Stable Carbocation
Carbocation stability is determined by resonance, inductive effect, and hyperconjugation.
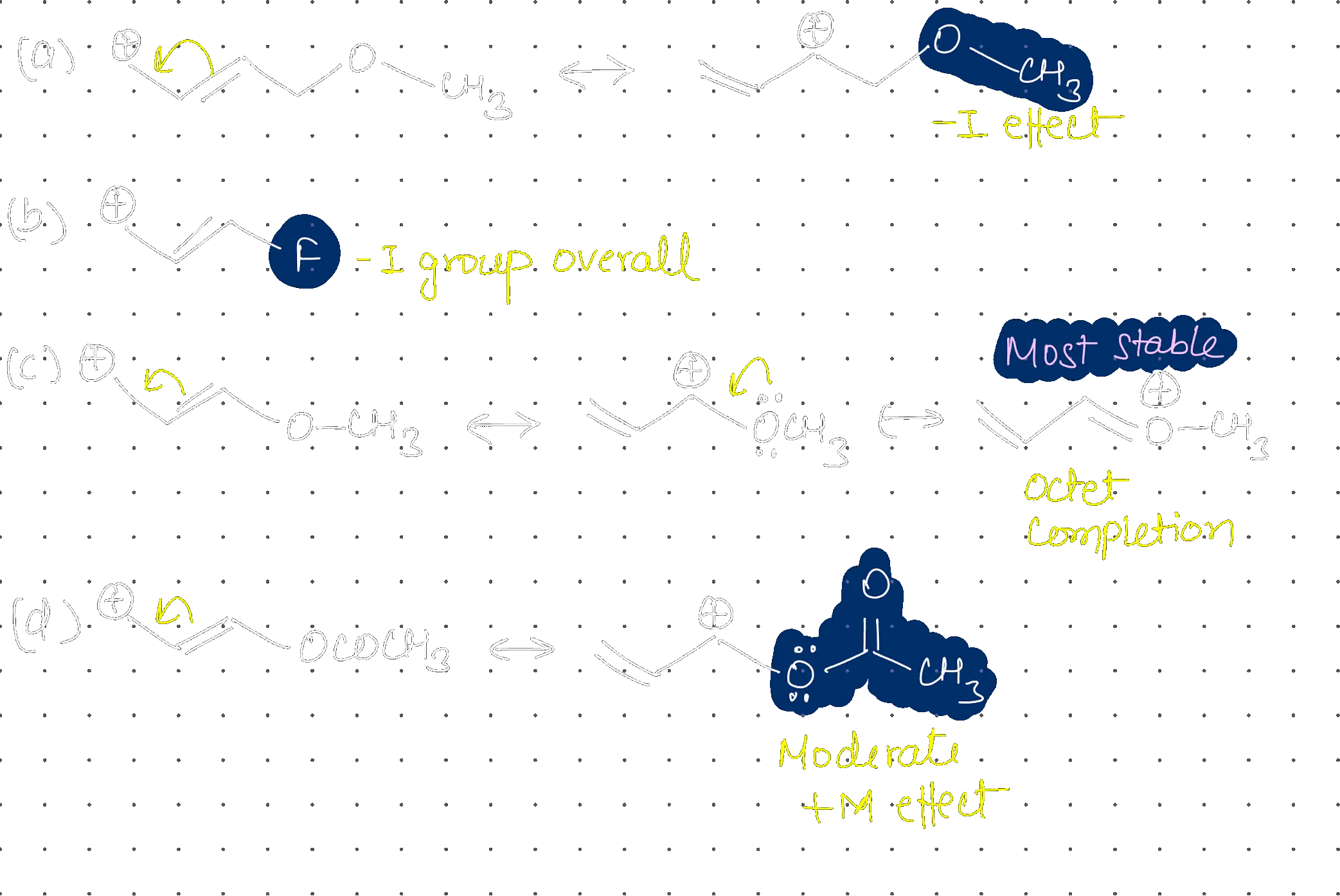
Decreasing order of hydride affinity for carbocations:
(C) > (A) > (B) > (D)
Hydride Affinity and Carbocation Stability Hydride affinity is the tendency of a carbocation to accept a hydride ion. It is inversely proportional to the stability of the carbocation. The more stable the carbocation, the lower its affinity for hydride.
- (D): Tricyclopropylmethyl cation (Exceptionally stable due to Bent orbital overlap). Lowest hydride affinity.
- (B): Triphenylmethyl cation (Stable due to resonance with three rings).
- (A): Allylic + tertiary stabilized cation.
- (C): tert-butyl cation (Least stable among the four).
Step 2: Order of Affinity Affinity order: (C) > (A) > (B) > (D).
Which of the following has highly acidic hydrogen?
Structure - d
Step 1: Analyzing Acidic Hydrogens in Ketones
Acidity depends on the resonance stabilization of the conjugate base (enolate ion).
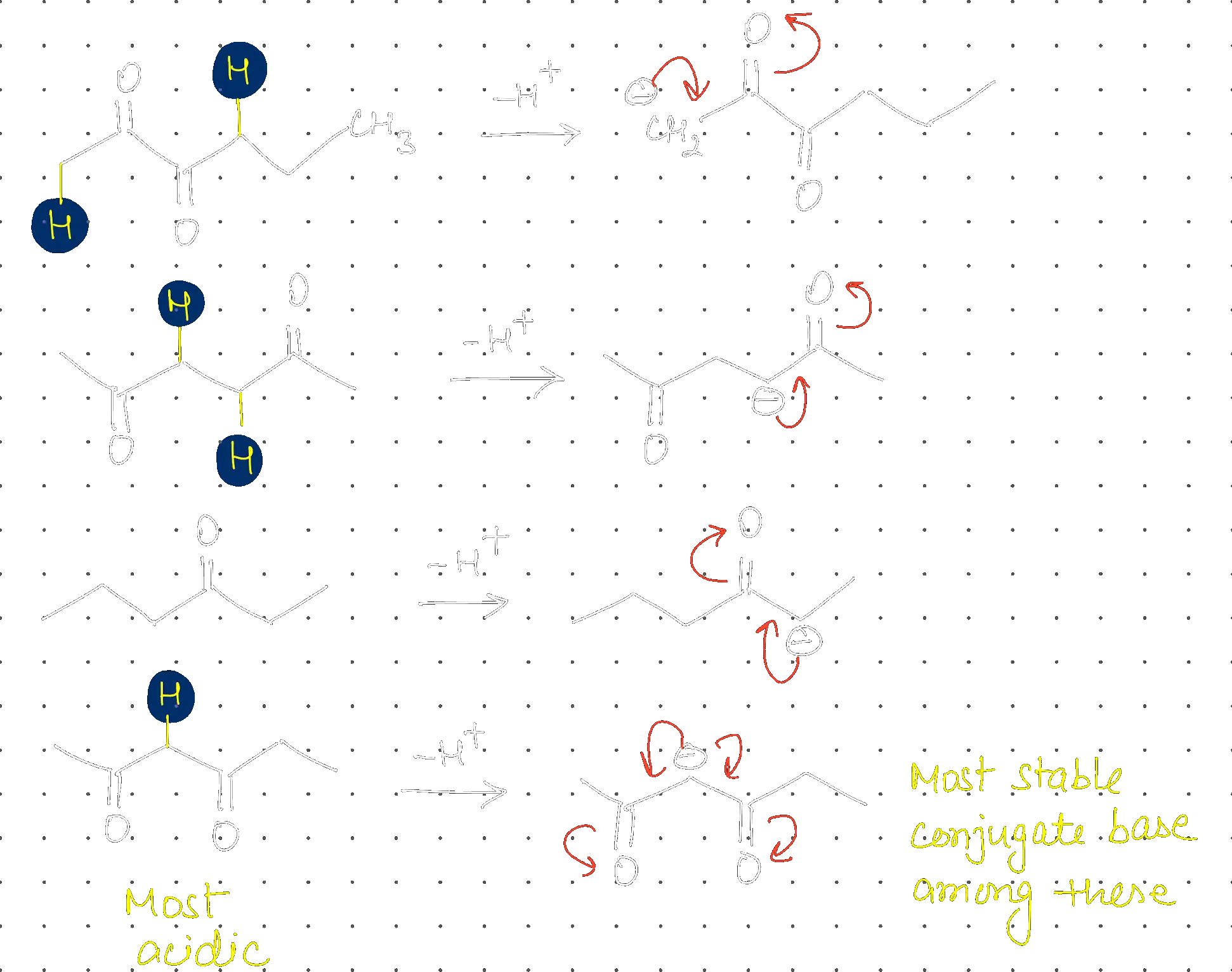 Step 2: Determining Maximum Acidity
The enolate formed in structure (d) is stabilized by resonance with two adjacent groups, making these hydrogens highly acidic
().
Step 2: Determining Maximum Acidity
The enolate formed in structure (d) is stabilized by resonance with two adjacent groups, making these hydrogens highly acidic
().
Total number of nucleophiles from the following is:
, PhSH, , , , , ,
5
Identifying Nucleophiles
Nucleophiles are species that have a lone pair of electrons or a bond that can be donated to an electrophile.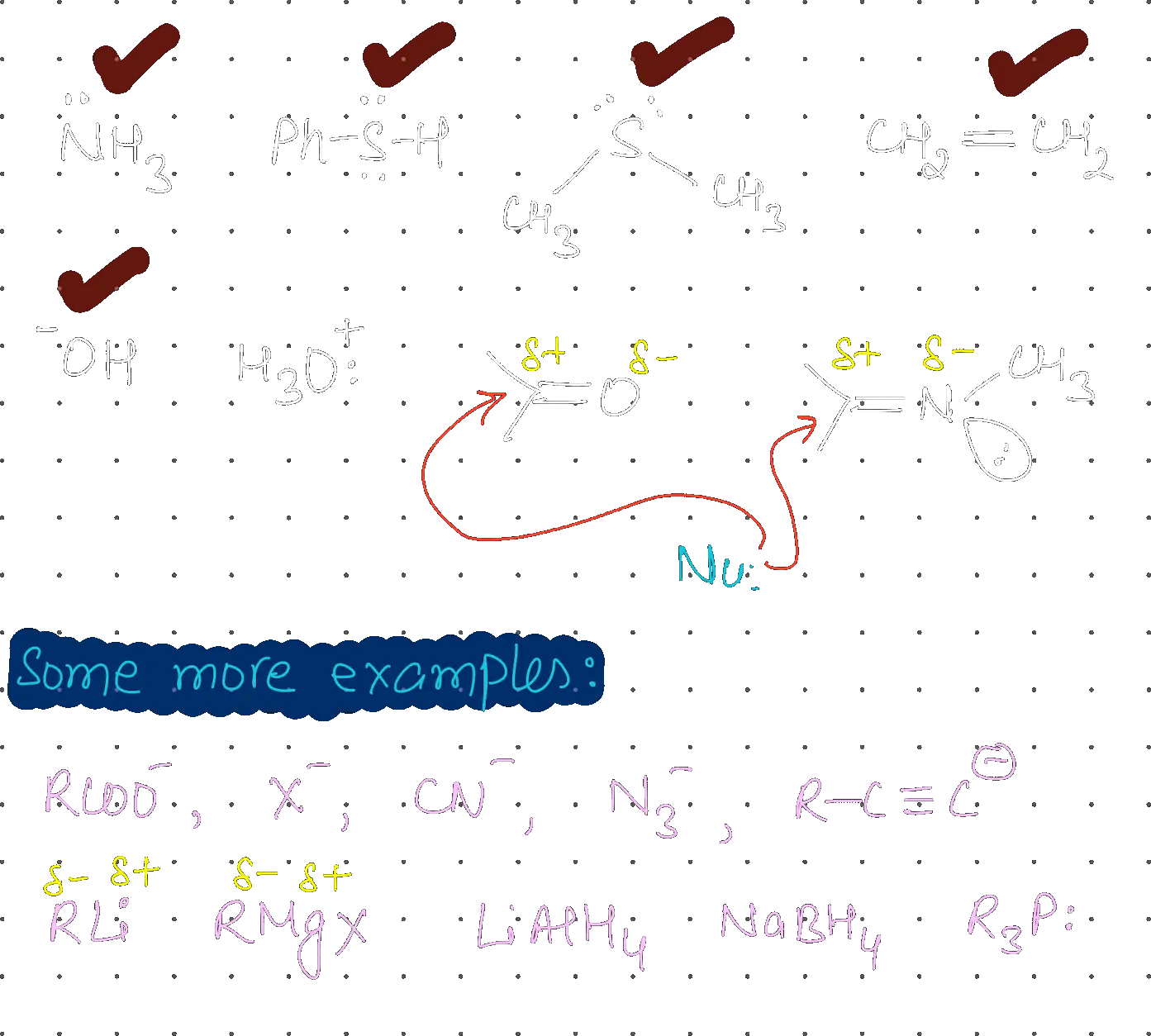 Evaluating Other Species
Evaluating Other Species
- : Electrophile (positive charge, no lone pair to donate).
- and : Electrophilic at the C-atom. Total count = 5.
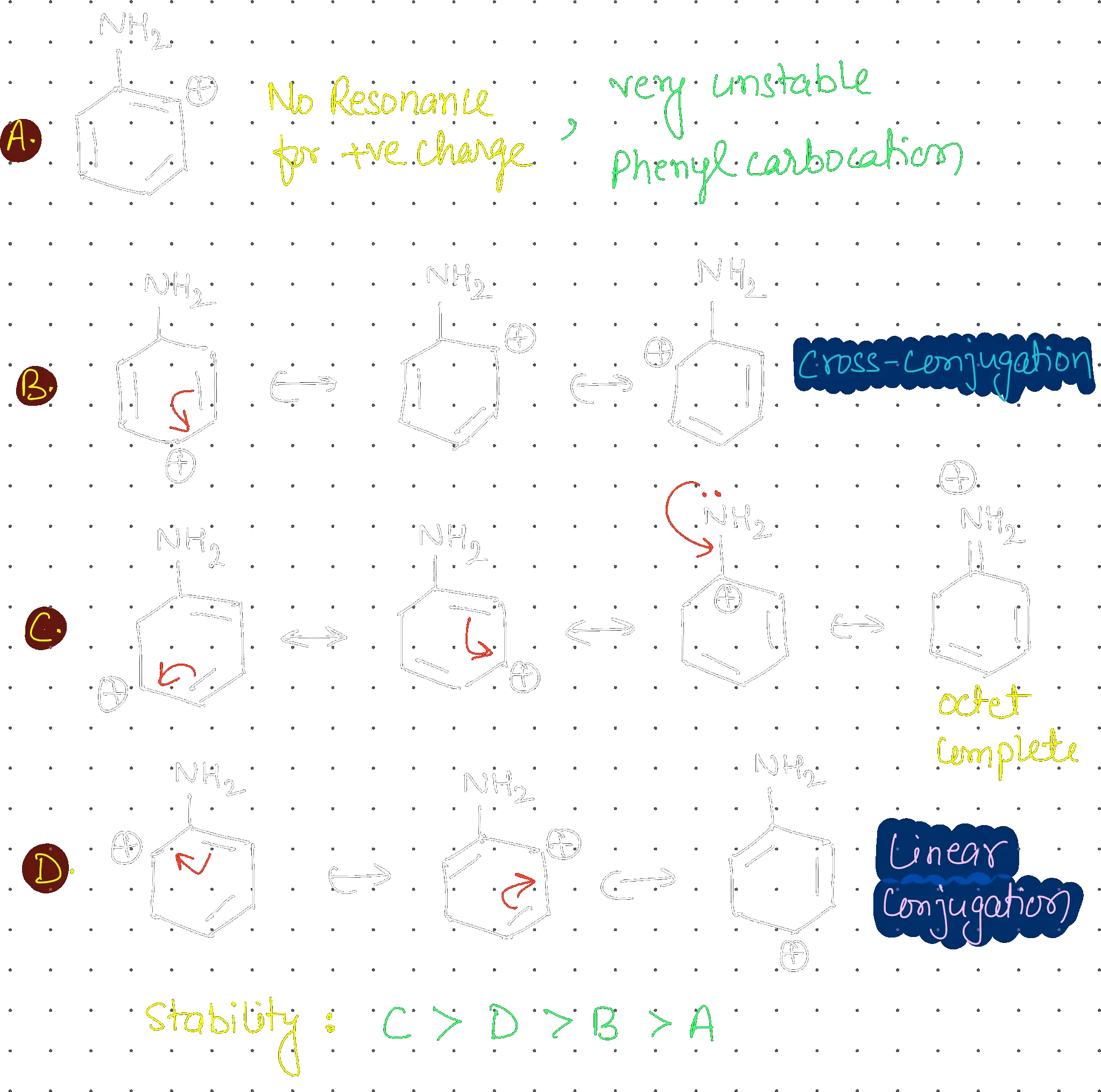
Most stable carbocation for the following is:
C
Linear conjugation (in C) is more stable than cross-conjugation (in B). Linear systems allow for continuous, uninterrupted delocalization of electrons across the entire molecule, maximizing resonance energy. In contrast, cross-conjugated systems have broken paths where the conjugation is split, leading to less efficient delocalization and lower stability.
Most stable carbocation:
Structure D
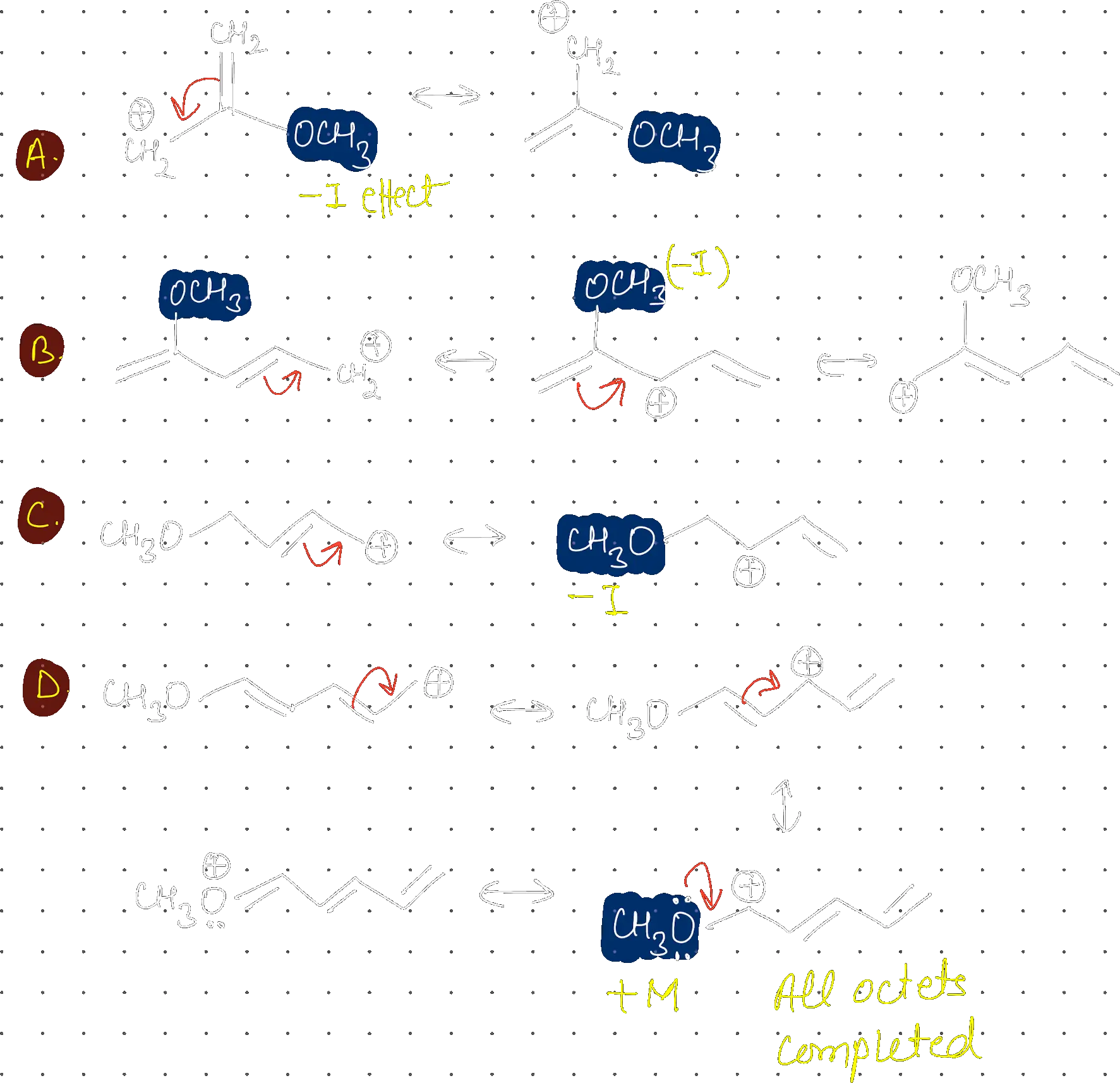
Compound with highest dipole moment is:
Structure A
Highest enol content will be shown by:
Compound (b)
Keto-Enol Tautomerism:
The enol content of a carbonyl compound depends on the stability of the enol form. Factors that stabilize the enol form include:
- Conjugation with other double bonds
- Intramolecular hydrogen bonding
- Aromaticity (if applicable)
Analysis of Options:
(a) Cyclohexane-1,3-dione: The two carbonyl groups are separated by one CH₂ group, so the enol form has extended conjugation and hydrogen bonding making enol form particularly stable.
(b) Cyclohexane-1,3,5-trione: This compound has three carbonyl groups alternating with CH₂ groups. The enol form is aromatic and has maximum conjugation and multiple intramolecular hydrogen bonds, making it the most stable enol form. Aromaticity alone is the biggest differentiator here.
(c) Cyclohexane-1,4-dione: Has two carbonyl groups with two CH₂ groups between them. The enol form has no conjugation and no hydrogen bonding due to both carbonyl groups positioned diagonally across.
(d) Cyclohexane-1,2,3-trione: Three adjacent carbonyl groups. The middle carbonyl group can't participate in tautomerism. Also three carbonyl groups repel each other strongly due to being on same side. The enol form has limited stabilization.
Conclusion: Cyclohexane-1,3,5-trione (option b) shows the highest enol content due to maximum stabilization through aromaticity and multiple intramolecular hydrogen bonding sites.
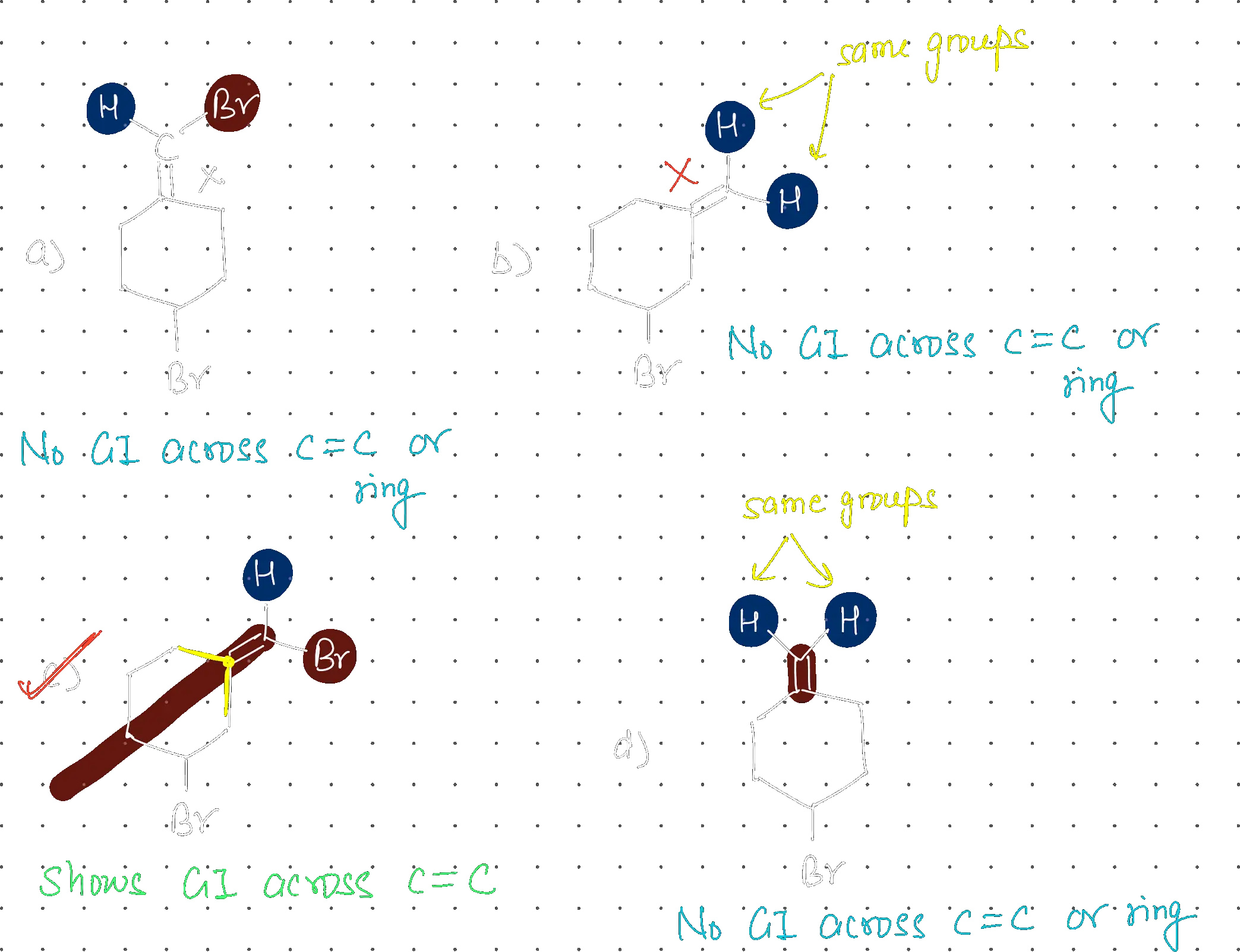
Which one of the following will show geometrical isomerism?
Structure C
Conditions for Geometrical Isomerism: For a compound to show geometrical (cis-trans or E/Z) isomerism:
- Must have a C=C double bond (restricted rotation)
- Each carbon of the double bond must have two different groups
- The double bond should allow for different spatial arrangements
Total number of stereo isomers possible for the given structure is:
8
Step 1: Identify Stereocenters
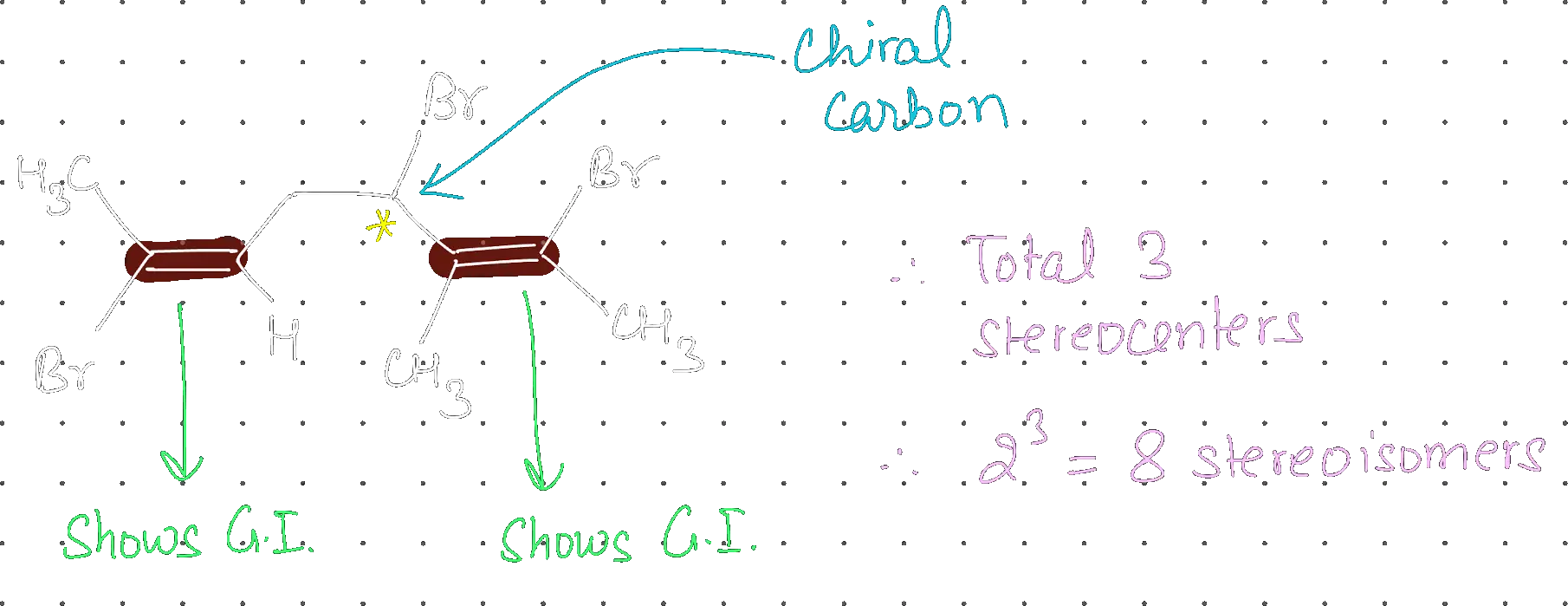 Step 2: Calculate Total Isomers
Since the molecule is unsymmetrical, the total number of stereoisomers is , where is the number of stereocenters ().
Step 2: Calculate Total Isomers
Since the molecule is unsymmetrical, the total number of stereoisomers is , where is the number of stereocenters ().
Given statements about Tropolone:
Statement-I: Tropolone is aromatic and has 8π electrons. Statement-II: π electrons of >C=O group in tropolone is involved in aromaticity.
Choose the correct option:
Statement-I true but Statement-II false
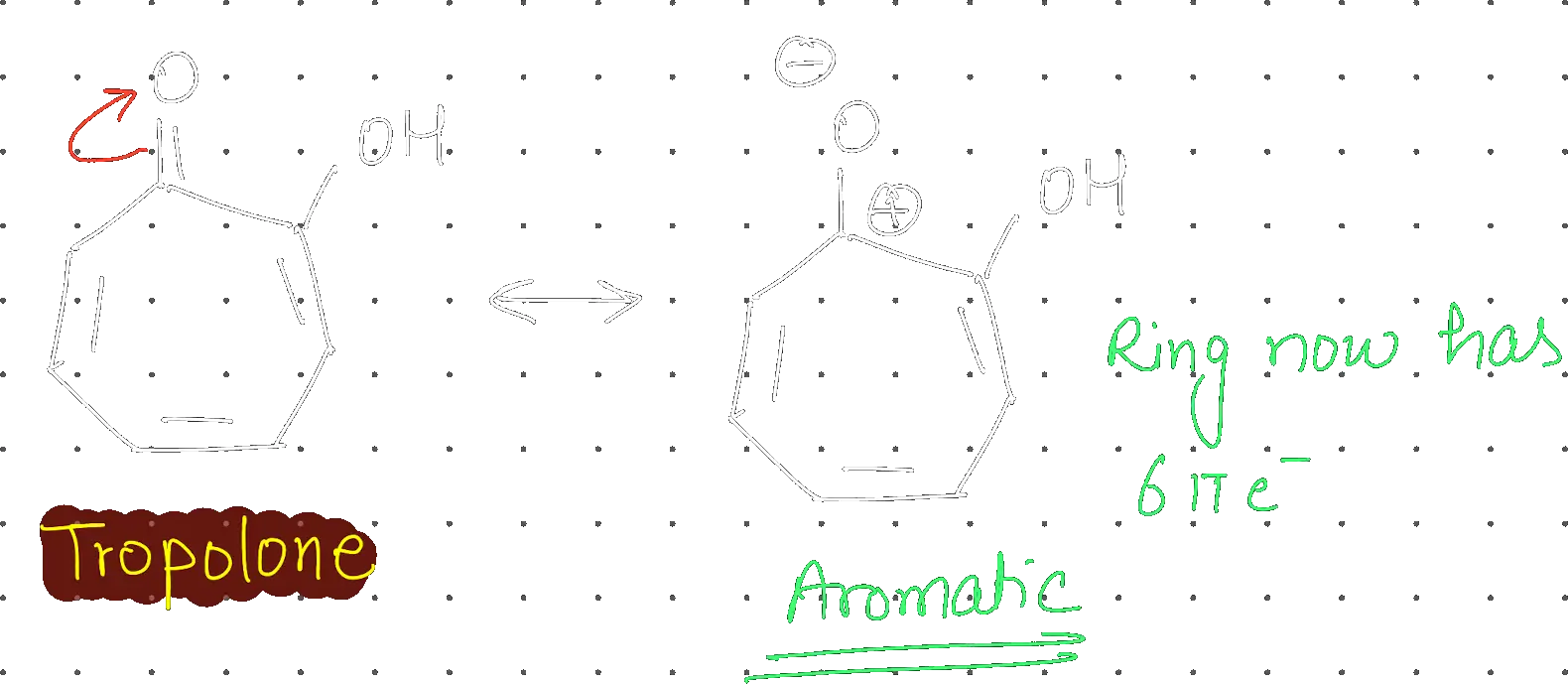 Tropolone has a total of 8 π electrons, but only 6 π electrons within the ring are involved in aromaticity. The first statement is correct that it has aromatic character and 8 π electrons.
Tropolone has a total of 8 π electrons, but only 6 π electrons within the ring are involved in aromaticity. The first statement is correct that it has aromatic character and 8 π electrons.
Second statement is incorrect because the pi electrons of the carbonyl group are not involved in aromaticity.
Correct statements for given reaction are:
(A) Compound B is aromatic
(B) Completion is very slow
(C) A shows tautomerism
(D) Bond lengths C-C in B are same
(A), (C) and (D) only
Step 1: Analyzing Squaric Acid Reaction Squaric acid reacts with 2 equivalents of to form the squarate dianion (Compound B).
- (A) Aromaticity: The squarate dianion is a 2π-electron system (resonance hybrid) which is aromatic and very stable. (True)
- (C) Tautomerism: Squaric acid (Compound A) exists in equilibrium with its tautomeric forms. (True)
- (D) Bond Lengths: Due to aromaticity and resonance, all bond lengths in the square dianion are identical. (True)
- Statement (B) says completion is very slow, which is typically false for acid-base neutralizations of such sharp acids.
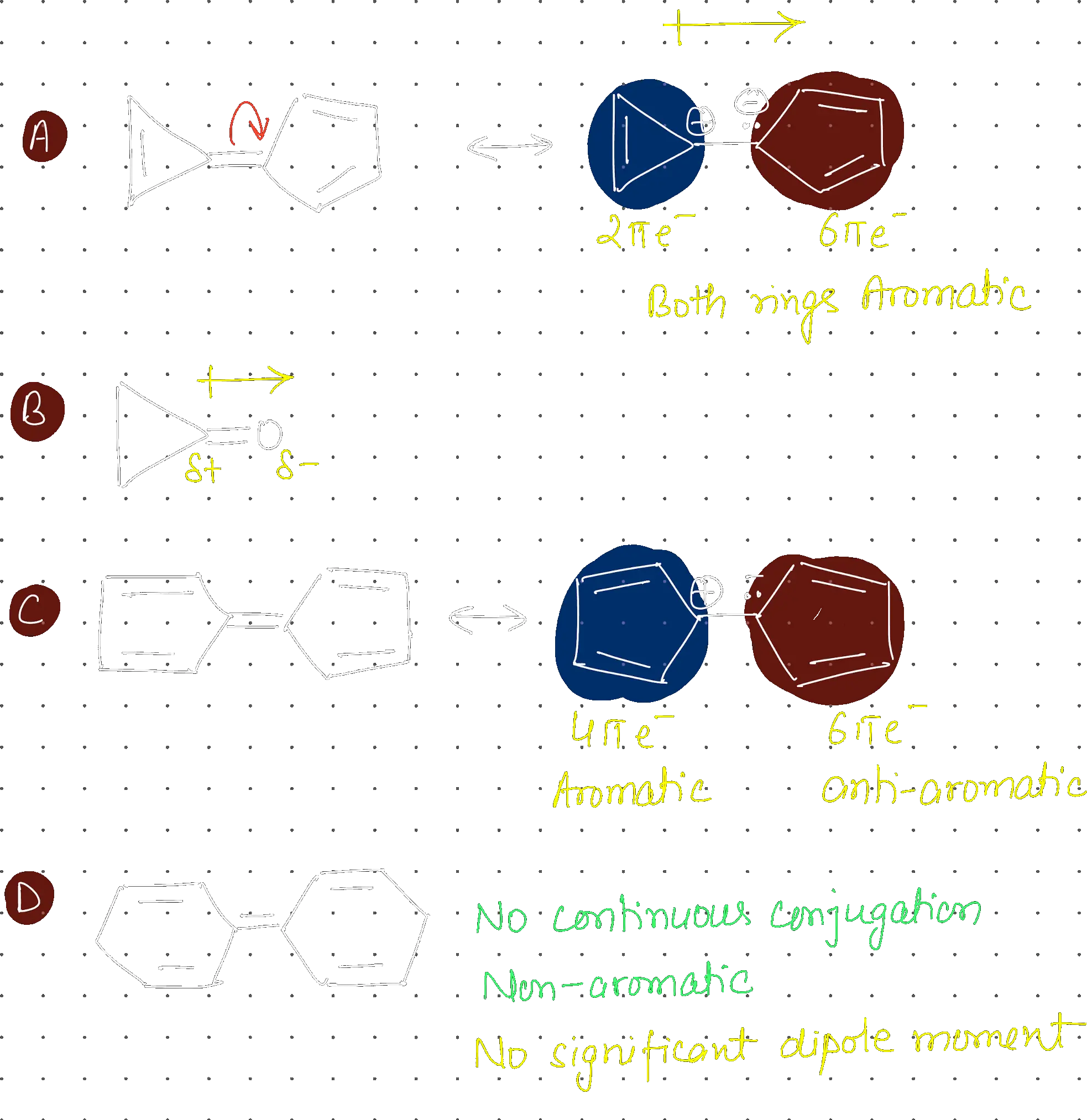
Which of the following compound is non aromatic:
Option a
Strategy: A compound is aromatic if it is cyclic, planar, fully conjugated, and has electrons. It is non-aromatic if it fails any of the first three criteria (usually conjugation is broken by an atom).
Step 1: Analyze each compound
- (a): Aziridinium cation-like system. The top nitrogen is and has no lone pair/p-orbital for conjugation. The system is not fully conjugated. Therefore, it is non-aromatic.
- (b): Pyrylium cation. electrons, fully conjugated. Aromatic.
- (c): Cyclopentadienyl anion. electrons, fully conjugated. Aromatic.
- (d): Furan-like system (1,3-dioxole derivative). electrons (including lone pairs from oxygens), fully conjugated. Aromatic.
Step 2: Conclusion Compound (a) is non-aromatic.
Frequently Asked Questions
What is in this General Organic Chemistry MCQ quiz?
+
Thirty multiple-choice questions sourced from star-marked JEE Main previous-year questions (PYQs) in the Canvas Classes Crucible question bank, balanced across the primary topics of GOC: nomenclature, electronic effects, acidity/basicity, reaction intermediates, electrophiles & nucleophiles, structural isomerism, geometrical isomerism, optical isomerism, conformational analysis, aromaticity, and atropisomerism / spiro compounds.
How are these questions different from the rest of the Crucible bank?
+
These are the GOC questions Paaras Sir's team has explicitly star-marked as high-yield JEE Main PYQs — every one of them has appeared in an official JEE Main paper. The full solution and explanation for each is the same one used inside The Crucible practice platform.
I'm preparing for NEET. Will these GOC questions still help me?
+
Yes. NEET's organic chemistry overlaps heavily with the JEE Main syllabus for GOC — concepts like inductive effect, hyperconjugation, carbocation stability and IUPAC naming appear in both. The questions are well-suited for NEET revision; the only caveat is that JEE Main questions are slightly more rigorous on stereochemistry and combined-effects reasoning.
Where can I practice more GOC questions?
+
Use The Crucible — Canvas Classes' adaptive practice platform — for the full GOC chapter with hundreds of additional questions, adaptive difficulty, and personalised recommendations based on your strengths and weaknesses.
Should I do this quiz before or after revising the GOC theory?
+
Either works. Practice-first surfaces the gaps in your understanding fast — the explanations then act as targeted revision. Read-first checks how solid your concepts are. If you score below 60%, prioritise re-reading electronic effects, intermediates, and acidity/basicity reasoning before attempting more PYQs.