Atomic Spectra and the Hydrogen Spectrum
Why atoms emit only specific colours — the fingerprint that revealed quantised energy levels
During a solar eclipse in 1868, a French astronomer noticed a strange yellow line in the Sun's spectrum that matched no known element on Earth. Scientists named the mystery element helium after Helios, the Greek god of the Sun. It wasn't until 1895 that William Ramsay finally isolated helium from a uranium mineral in his London lab. A whole element, identified from 150 million kilometres away — purely by reading the light it emitted. That is the power of atomic spectra.


The speed of light depends upon the nature of the medium through which it passes. As a result, the beam of light is deviated or refracted from its original path as it passes from one medium to another.
It is observed that when a ray of white light is passed through a prism, the wave with shorter wavelength bends more than the one with a longer wavelength. Since ordinary white light consists of waves with all the wavelengths in the visible range, a ray of white light is spread out into a series of coloured bands called spectrum. The light of red colour which has longest wavelength is deviated the least while the violet light, which has shortest wavelength is deviated the most.

Now heat sodium vapour until it glows. Instead of a rainbow, you see just two sharp yellow lines sitting in complete darkness. That is a line spectrum — and those two lines are sodium's identity card. Every element has its own unique set of lines, and no two elements share the same pattern.
Continuous spectra and line spectra
Listen to the audio explanation
Understanding why atoms produce line spectra — and not continuous rainbows — turned out to be one of the most important clues that led scientists to quantum mechanics.
Two Types of Spectra: Continuous and Line
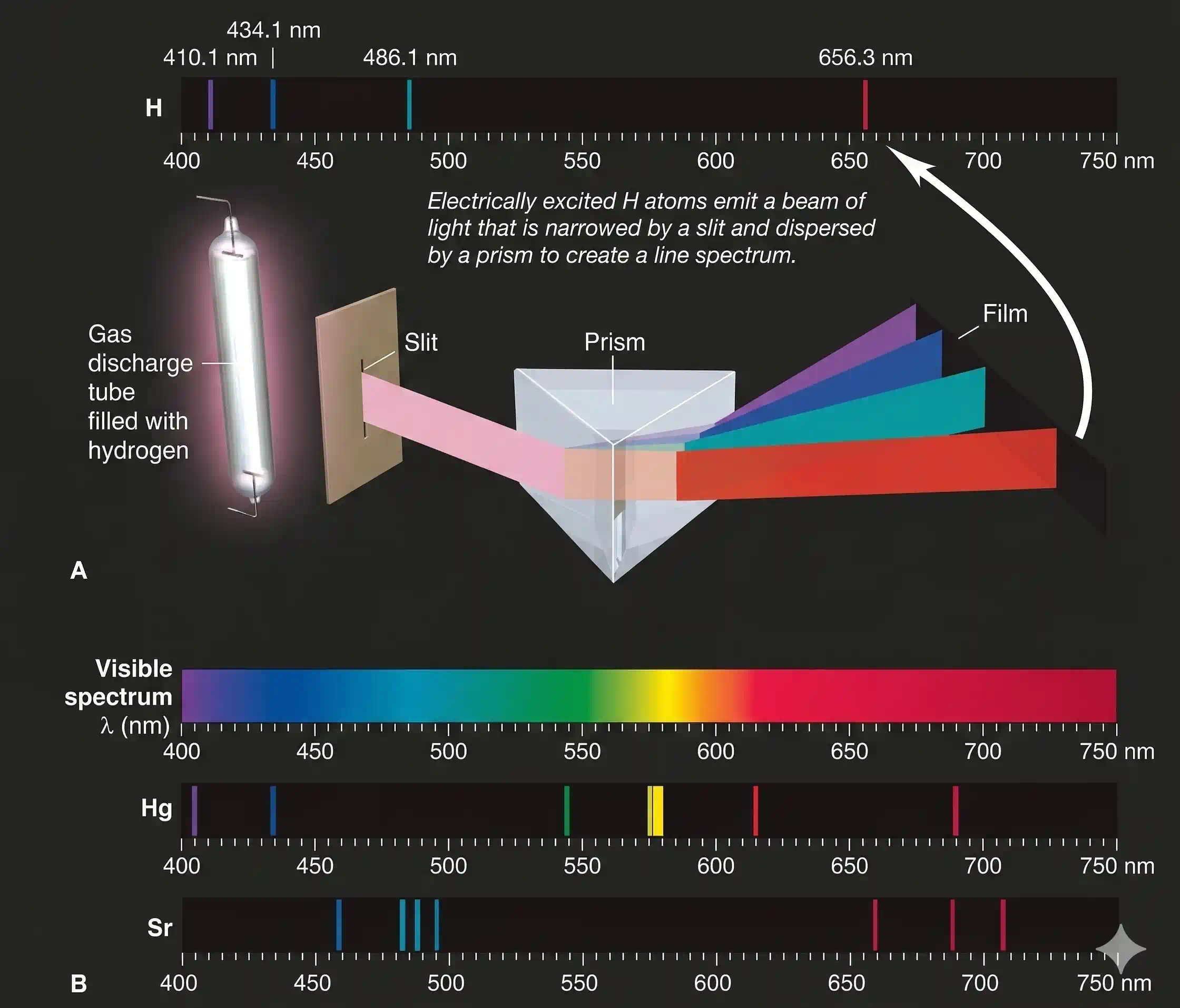
When light from electrically excited gaseous atoms passes through a slit and is refracted by a prism, it does not create a continuous spectrum, or rainbow, as sunlight does. Instead, it creates a line spectrum, a series of fine lines at specific frequencies separated by black spaces. Figure below shows the apparatus and the line spectrum of atomic hydrogen and line spectrum of mercury and strontium.
The emission spectrum shows only a handful of sharp bright lines at specific wavelengths. The rest is dark. These are called line spectra or atomic spectra, because the emitted radiation appears as discrete bright lines — not a rainbow.
Absorption spectra work the opposite way: pass a beam of white light through a cool gas, and that gas absorbs exactly the same specific wavelengths it would otherwise emit. You see a rainbow with dark gaps — a photographic negative of the emission spectrum. The study of both kinds is called spectroscopy.
Emission Spectrum vs Absorption Spectrum
Emission Spectrum
- Produced when excited atoms *release* energy as light
- Appears as bright coloured lines on a dark background
- Each line corresponds to a specific wavelength emitted
- Atoms must be energised first (heat, electric discharge, UV)
- Used to identify elements present in a glowing/hot sample
Absorption Spectrum
- Produced when atoms *absorb* specific wavelengths from white light
- Appears as dark lines (gaps) on a continuous rainbow background
- Dark lines occur at exactly the same wavelengths as emission lines
- The cool gas sample absorbs what it would otherwise emit
- Used to identify elements in cool interstellar clouds and stellar atmospheres
Emission Spectrum
- Produced when excited atoms *release* energy as light
- Appears as bright coloured lines on a dark background
- Each line corresponds to a specific wavelength emitted
- Atoms must be energised first (heat, electric discharge, UV)
- Used to identify elements present in a glowing/hot sample
Absorption Spectrum
- Produced when atoms *absorb* specific wavelengths from white light
- Appears as dark lines (gaps) on a continuous rainbow background
- Dark lines occur at exactly the same wavelengths as emission lines
- The cool gas sample absorbs what it would otherwise emit
- Used to identify elements in cool interstellar clouds and stellar atmospheres
Line Spectra: Every Element's Unique Fingerprint
The key insight that made spectroscopy revolutionary: each element has a unique set of spectral lines — no two elements share the same pattern. Just as no two people share the same fingerprint, no two elements share the same spectrum.
This means you can identify an unknown element by matching its spectral lines against a reference table — even from across the galaxy. The German chemist Robert Bunsen (1811–1899) — famous for the burner he co-developed — was among the first to use this systematically for chemical analysis.
Elements discovered entirely through spectroscopy:
- Rubidium (Rb) and Caesium (Cs) — Bunsen & Kirchhoff, 1860–61, from mineral spring residues
- Thallium (Tl), Indium (In), Gallium (Ga), Scandium (Sc) — all found by spotting previously unknown lines in mineral spectra
- Helium (He) — found in the Sun's spectrum in 1868, isolated on Earth in 1895
Spectroscopy remains one of the most powerful analytical tools in chemistry, astronomy, and forensic science today.
The Hydrogen Spectrum — Simplest, Yet Most Revealing
Hydrogen has only one electron, making its spectrum the simplest of all elements — and the easiest to analyse mathematically. When you pass an electric discharge through gaseous hydrogen, the molecules dissociate into individual atoms. The energetically excited hydrogen atoms then emit electromagnetic radiation, but only at discrete (specific) frequencies — not a continuous spread.
The hydrogen spectrum is organised into several distinct series of lines, each appearing in a different region of the electromagnetic spectrum, and each named after the scientist who first studied it:
- Lyman series — ultraviolet region
- Balmer series — visible region (the only one you can see with your eyes)
- Paschen series — infrared region
- Brackett series — infrared region
- Pfund series — infrared region
AI Generation Prompt
Hydrogen atom energy level diagram showing electron transitions for spectral series. Vertical layout with energy increasing upward. Horizontal lines represent energy levels from n=1 (bottom, ground state, labelled n=1) up through n=2, n=3, n=4, n=5, n=6 and n=infinity (top, ionisation limit, dashed line at 0 eV). Draw downward-pointing arrows for electronic transitions grouped by series: Lyman series — many blue/indigo arrows terminating at n=1 (labelled 'Lyman series (ultraviolet)'); Balmer series — four arrows terminating at n=2, coloured Red, Green, Blue, Violet from longest to shortest wavelength (labelled 'Balmer series (visible)', with colour labels on each arrow); Paschen series — arrows terminating at n=3 (labelled 'Paschen series (infrared)'). Energy axis on the left with an upward arrow and label 'Energy'. Show n=infinity dashed line at top with label 'n = infinity'. Label all quantum number levels: n=1 through n=6. Dark background, orange accent labels, clean technical illustration style.
Balmer's Formula — Finding the Pattern (1885)
In 1885, the Swiss mathematician Johann Balmer discovered that the four visible lines of the hydrogen spectrum obeyed a beautifully simple formula. He expressed the lines in terms of wavenumber — the number of waves per centimetre (unit: ) — which is the reciprocal of wavelength:
The Balmer formula for the visible lines:
Equation 2.8 — Balmer Series
where n is an integer ≥ 3 (i.e., n = 3, 4, 5, …). These lines lie in the visible region of the spectrum.
The Swedish spectroscopist Johannes Rydberg then showed that all the series of lines in the hydrogen spectrum — not just the visible Balmer series — could be described by a single generalised formula:
Equation 2.9 — Rydberg Formula
where n₁ = 1, 2, 3, … and n₂ = n₁ + 1, n₁ + 2, … The constant 109,677 cm⁻¹ is called the Rydberg constant (R∞). Each series corresponds to a fixed value of n₁ with n₂ taking successive higher integer values.
Table 2.3 — Spectral Line Series for Atomic Hydrogen
| Series | n₁ (lower level) | n₂ (upper levels) | Spectral Region |
|---|---|---|---|
| Lyman | 1 | 2, 3, 4, … | Ultraviolet |
| Balmer | 2 | 3, 4, 5, … | Visible |
| Paschen | 3 | 4, 5, 6, … | Infrared |
| Brackett | 4 | 5, 6, 7, … | Infrared |
| Pfund | 5 | 6, 7, 8, … | Infrared |
The Physical Meaning of n₁ and n₂
In the Rydberg formula, is the lower energy level (the shell the electron falls into) and is the higher energy level (where it starts). The photon is emitted because the electron loses energy dropping from down to .
The series name tells you :
- Lyman: (electron lands in ground state)
- Balmer: (visible — why we can see these)
- Paschen: , Brackett: , Pfund:
Why Does Hydrogen Emit Only Specific Lines?
NCERT poses this question directly at the end of the section: line spectra are unique to each element, and there is regularity in the pattern. Why? What does this tell us about electronic structure?
Q1.Which series of the hydrogen spectrum falls in the visible region?
During a solar eclipse in 1868, a French astronomer noticed a strange yellow line in the Sun's spectrum that matched no known element on Earth. Scientists named the mystery element helium after Helios, the Greek god of the Sun. It wasn't until 1895 that William Ramsay finally isolated helium from a uranium mineral in his London lab. A whole element, identified from 150 million kilometres away — purely by reading the light it emitted. That is the power of atomic spectra.


The speed of light depends upon the nature of the medium through which it passes. As a result, the beam of light is deviated or refracted from its original path as it passes from one medium to another.
It is observed that when a ray of white light is passed through a prism, the wave with shorter wavelength bends more than the one with a longer wavelength. Since ordinary white light consists of waves with all the wavelengths in the visible range, a ray of white light is spread out into a series of coloured bands called spectrum. The light of red colour which has longest wavelength is deviated the least while the violet light, which has shortest wavelength is deviated the most.

Now heat sodium vapour until it glows. Instead of a rainbow, you see just two sharp yellow lines sitting in complete darkness. That is a line spectrum — and those two lines are sodium's identity card. Every element has its own unique set of lines, and no two elements share the same pattern.
Continuous spectra and line spectra
Listen to the audio explanation
Understanding why atoms produce line spectra — and not continuous rainbows — turned out to be one of the most important clues that led scientists to quantum mechanics.
Two Types of Spectra: Continuous and Line
When light from electrically excited gaseous atoms passes through a slit and is refracted by a prism, it does not create a continuous spectrum, or rainbow, as sunlight does. Instead, it creates a line spectrum, a series of fine lines at specific frequencies separated by black spaces. Figure below shows the apparatus and the line spectrum of atomic hydrogen and line spectrum of mercury and strontium.
The emission spectrum shows only a handful of sharp bright lines at specific wavelengths. The rest is dark. These are called line spectra or atomic spectra, because the emitted radiation appears as discrete bright lines — not a rainbow.
Absorption spectra work the opposite way: pass a beam of white light through a cool gas, and that gas absorbs exactly the same specific wavelengths it would otherwise emit. You see a rainbow with dark gaps — a photographic negative of the emission spectrum. The study of both kinds is called spectroscopy.

Emission Spectrum vs Absorption Spectrum
Emission Spectrum
- Produced when excited atoms *release* energy as light
- Appears as bright coloured lines on a dark background
- Each line corresponds to a specific wavelength emitted
- Atoms must be energised first (heat, electric discharge, UV)
- Used to identify elements present in a glowing/hot sample
Absorption Spectrum
- Produced when atoms *absorb* specific wavelengths from white light
- Appears as dark lines (gaps) on a continuous rainbow background
- Dark lines occur at exactly the same wavelengths as emission lines
- The cool gas sample absorbs what it would otherwise emit
- Used to identify elements in cool interstellar clouds and stellar atmospheres
Emission Spectrum
- Produced when excited atoms *release* energy as light
- Appears as bright coloured lines on a dark background
- Each line corresponds to a specific wavelength emitted
- Atoms must be energised first (heat, electric discharge, UV)
- Used to identify elements present in a glowing/hot sample
Absorption Spectrum
- Produced when atoms *absorb* specific wavelengths from white light
- Appears as dark lines (gaps) on a continuous rainbow background
- Dark lines occur at exactly the same wavelengths as emission lines
- The cool gas sample absorbs what it would otherwise emit
- Used to identify elements in cool interstellar clouds and stellar atmospheres
Line Spectra: Every Element's Unique Fingerprint
The key insight that made spectroscopy revolutionary: each element has a unique set of spectral lines — no two elements share the same pattern. Just as no two people share the same fingerprint, no two elements share the same spectrum.
This means you can identify an unknown element by matching its spectral lines against a reference table — even from across the galaxy. The German chemist Robert Bunsen (1811–1899) — famous for the burner he co-developed — was among the first to use this systematically for chemical analysis.
Elements discovered entirely through spectroscopy:
- Rubidium (Rb) and Caesium (Cs) — Bunsen & Kirchhoff, 1860–61, from mineral spring residues
- Thallium (Tl), Indium (In), Gallium (Ga), Scandium (Sc) — all found by spotting previously unknown lines in mineral spectra
- Helium (He) — found in the Sun's spectrum in 1868, isolated on Earth in 1895
Spectroscopy remains one of the most powerful analytical tools in chemistry, astronomy, and forensic science today.
The Hydrogen Spectrum — Simplest, Yet Most Revealing
Hydrogen has only one electron, making its spectrum the simplest of all elements — and the easiest to analyse mathematically. When you pass an electric discharge through gaseous hydrogen, the molecules dissociate into individual atoms. The energetically excited hydrogen atoms then emit electromagnetic radiation, but only at discrete (specific) frequencies — not a continuous spread.
The hydrogen spectrum is organised into several distinct series of lines, each appearing in a different region of the electromagnetic spectrum, and each named after the scientist who first studied it:
- Lyman series — ultraviolet region
- Balmer series — visible region (the only one you can see with your eyes)
- Paschen series — infrared region
- Brackett series — infrared region
- Pfund series — infrared region
AI Generation Prompt
Hydrogen atom energy level diagram showing electron transitions for spectral series. Vertical layout with energy increasing upward. Horizontal lines represent energy levels from n=1 (bottom, ground state, labelled n=1) up through n=2, n=3, n=4, n=5, n=6 and n=infinity (top, ionisation limit, dashed line at 0 eV). Draw downward-pointing arrows for electronic transitions grouped by series: Lyman series — many blue/indigo arrows terminating at n=1 (labelled 'Lyman series (ultraviolet)'); Balmer series — four arrows terminating at n=2, coloured Red, Green, Blue, Violet from longest to shortest wavelength (labelled 'Balmer series (visible)', with colour labels on each arrow); Paschen series — arrows terminating at n=3 (labelled 'Paschen series (infrared)'). Energy axis on the left with an upward arrow and label 'Energy'. Show n=infinity dashed line at top with label 'n = infinity'. Label all quantum number levels: n=1 through n=6. Dark background, orange accent labels, clean technical illustration style.
Balmer's Formula — Finding the Pattern (1885)
In 1885, the Swiss mathematician Johann Balmer discovered that the four visible lines of the hydrogen spectrum obeyed a beautifully simple formula. He expressed the lines in terms of wavenumber — the number of waves per centimetre (unit: ) — which is the reciprocal of wavelength:
The Balmer formula for the visible lines:
Equation 2.8 — Balmer Series
where n is an integer ≥ 3 (i.e., n = 3, 4, 5, …). These lines lie in the visible region of the spectrum.
The Swedish spectroscopist Johannes Rydberg then showed that all the series of lines in the hydrogen spectrum — not just the visible Balmer series — could be described by a single generalised formula:
Equation 2.9 — Rydberg Formula
where n₁ = 1, 2, 3, … and n₂ = n₁ + 1, n₁ + 2, … The constant 109,677 cm⁻¹ is called the Rydberg constant (R∞). Each series corresponds to a fixed value of n₁ with n₂ taking successive higher integer values.
Table 2.3 — Spectral Line Series for Atomic Hydrogen
| Series | n₁ (lower level) | n₂ (upper levels) | Spectral Region |
|---|---|---|---|
| Lyman | 1 | 2, 3, 4, … | Ultraviolet |
| Balmer | 2 | 3, 4, 5, … | Visible |
| Paschen | 3 | 4, 5, 6, … | Infrared |
| Brackett | 4 | 5, 6, 7, … | Infrared |
| Pfund | 5 | 6, 7, 8, … | Infrared |
Why Does Hydrogen Emit Only Specific Lines?
NCERT poses this question directly at the end of the section: line spectra are unique to each element, and there is regularity in the pattern. Why? What does this tell us about electronic structure?
Q1.Which series of the hydrogen spectrum falls in the visible region?