Wave Nature of Electromagnetic Radiation
From Maxwell's unified theory to the full EM spectrum — and why waves diffract while particles don't
At this exact moment, radio waves from hundreds of broadcast towers, WiFi routers, mobile base stations, and satellites are passing through your body. Your TV remote fires invisible infrared pulses. The sun is bombarding you with ultraviolet rays. Your own body emits infrared heat radiation. All of these — along with visible light, X-rays, and gamma rays — are the same type of wave, just at wildly different frequencies.
What is a Wave?
A wave is a disturbance that travels through space or matter, carrying energy from one place to another — without carrying matter itself. Think of ripples on a pond: the water molecules don't travel toward the shore, but the energy of the disturbance does.
Waves come in two fundamental types:
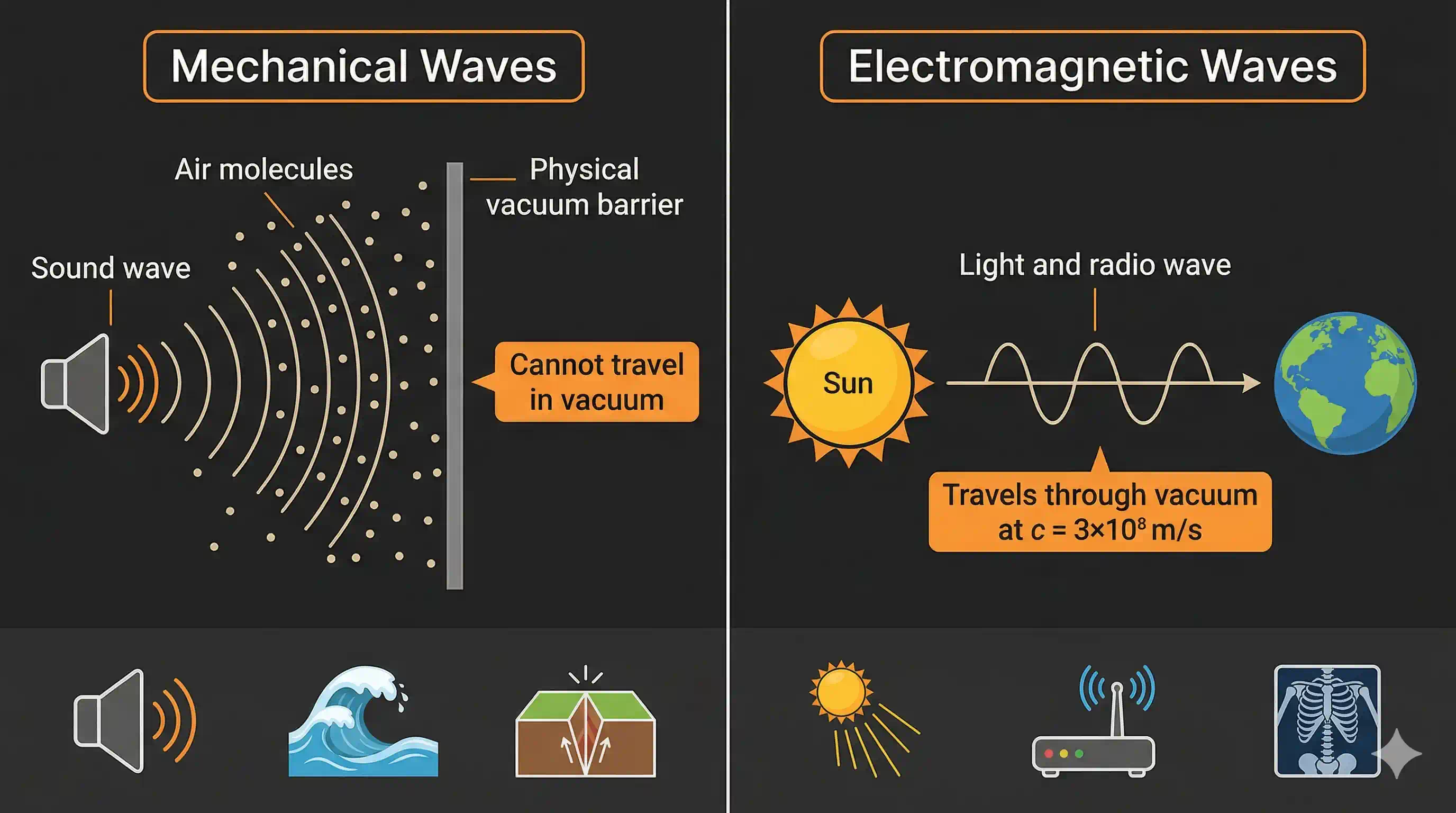
- Mechanical waves — need a physical medium to travel. Sound waves, water waves, and earthquake waves are mechanical. They cannot travel through a vacuum (empty space). That's why space is completely silent.
- Electromagnetic (EM) waves — need no medium at all. They can travel through the vacuum of space. That's how sunlight reaches us across 150 million kilometres of empty space.
Maxwell's Discovery: Light is an Electromagnetic Wave
In 1870, James Clerk Maxwell published equations showing that a charged particle undergoing acceleration creates oscillating electric and magnetic fields that propagate outward as a wave. These waves travel at exactly the speed of light — leading Maxwell to conclude that light itself is an electromagnetic wave.
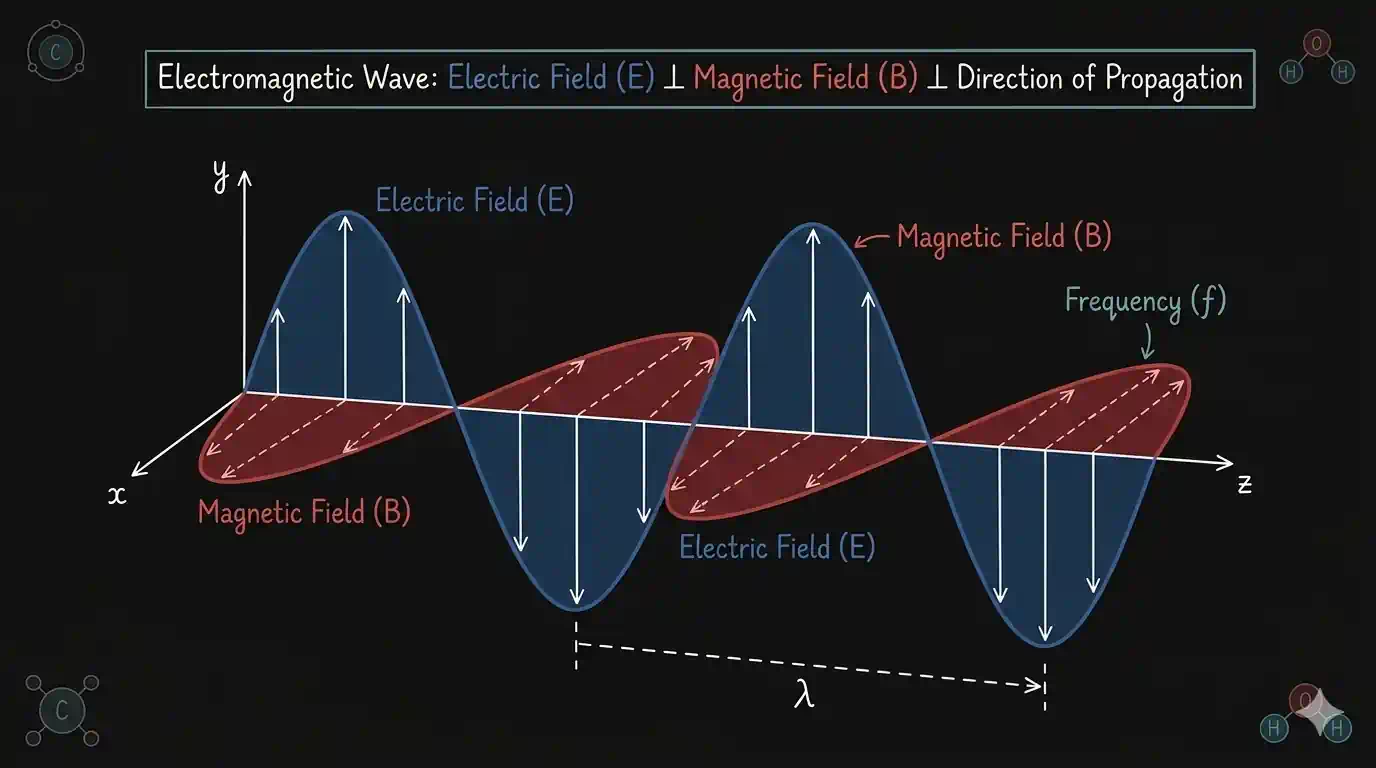
An electromagnetic wave has two key features:
- Perpendicular fields: The electric field (E) and the magnetic field (B) oscillate at right angles to each other, and both are perpendicular to the direction the wave travels.
- Self-sustaining: A changing E field creates a B field, which creates an E field — the wave sustains itself through empty space with no medium needed.
Key Properties of Electromagnetic Waves
- No medium required — travel through vacuum; sound cannot.
- Constant speed in vacuum — all EM waves travel at m s⁻¹, regardless of wavelength or frequency. (They slow slightly inside materials like glass or water.)
- Transverse waves — the oscillations are perpendicular to the direction of travel.
- Characterised by frequency (ν) and wavelength (λ) — related by:
- Carry energy — the energy of each photon is , where J s (Planck's constant). Higher frequency = more energy per photon.
Frequency, Wavelength, and the Speed of Light
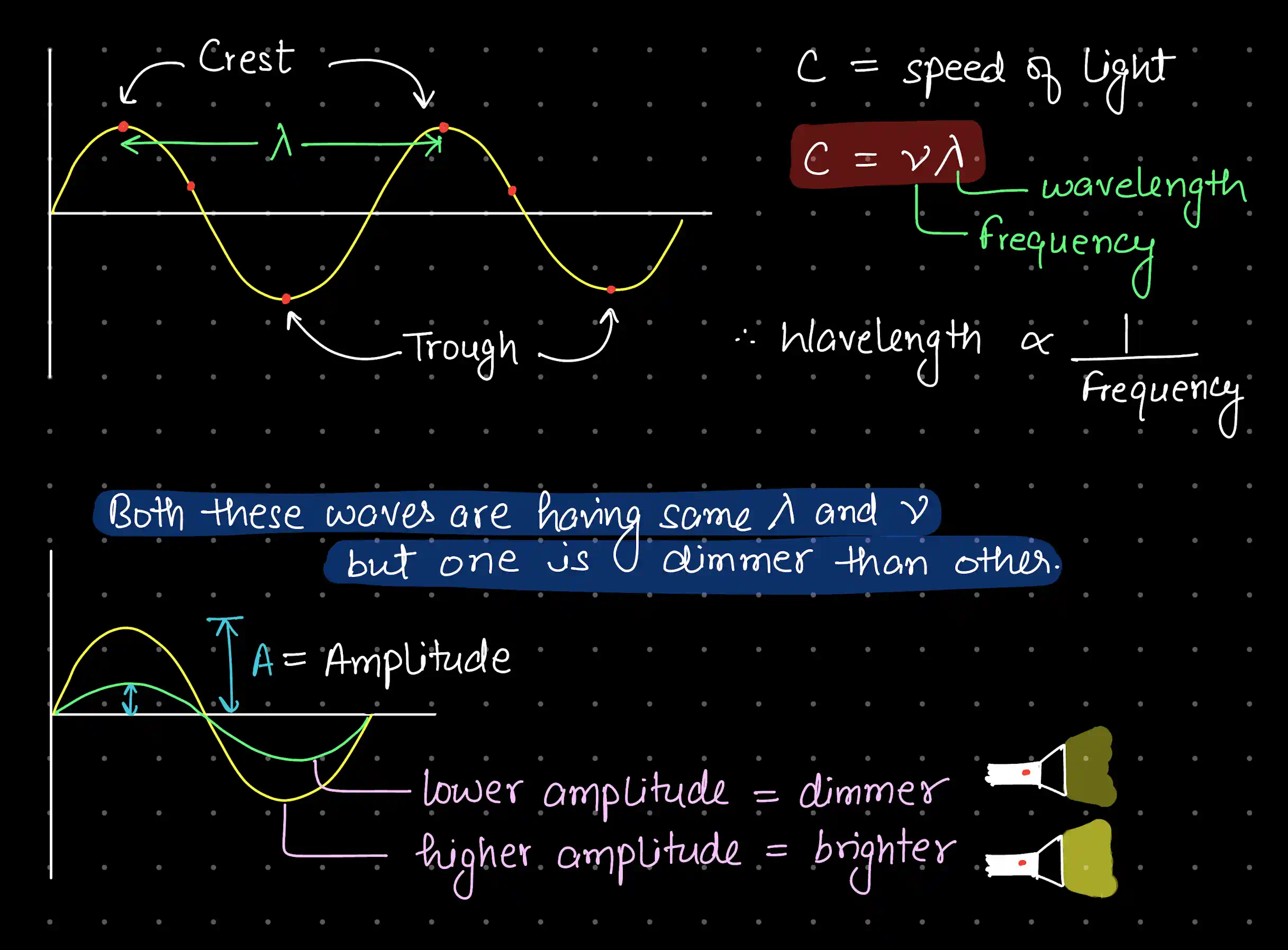
Wavelength (λ) is the distance between two successive crests (or troughs) of the wave. It is easy to visualise — you can literally measure it on a wave diagram.
Frequency (ν) is harder to picture because it describes time, not space. It is the number of complete wave cycles that pass a fixed point per second. Its SI unit is the hertz (Hz), named after Heinrich Hertz, where Hz s.
For all EM waves in vacuum:
where m s (often rounded to m s).
Wavenumber () is another useful quantity in spectroscopy. It is the number of wavelengths per unit length:
Its SI unit is m, but the commonly used unit in spectroscopy is cm.
Equations 2.5 & 2.6
c = 3.0 × 10⁸ m s⁻¹ · h = 6.626 × 10⁻³⁴ J s · ν in Hz · λ in m · ν̄ in m⁻¹ (or cm⁻¹)
See It: The EM Wave Explorer
Drag the slider to move through the electromagnetic spectrum from radio waves to gamma rays. Watch how the wavelength visible in the animation shrinks as frequency rises. Click any region label to jump straight to it.
Loading simulator…
The Electromagnetic Spectrum at a Glance
All electromagnetic radiation is the same type of wave — but the frequency range spans more than 20 orders of magnitude. Different regions of the spectrum interact with matter in completely different ways, which is why radio waves pass harmlessly through walls while gamma rays can kill cancer cells.
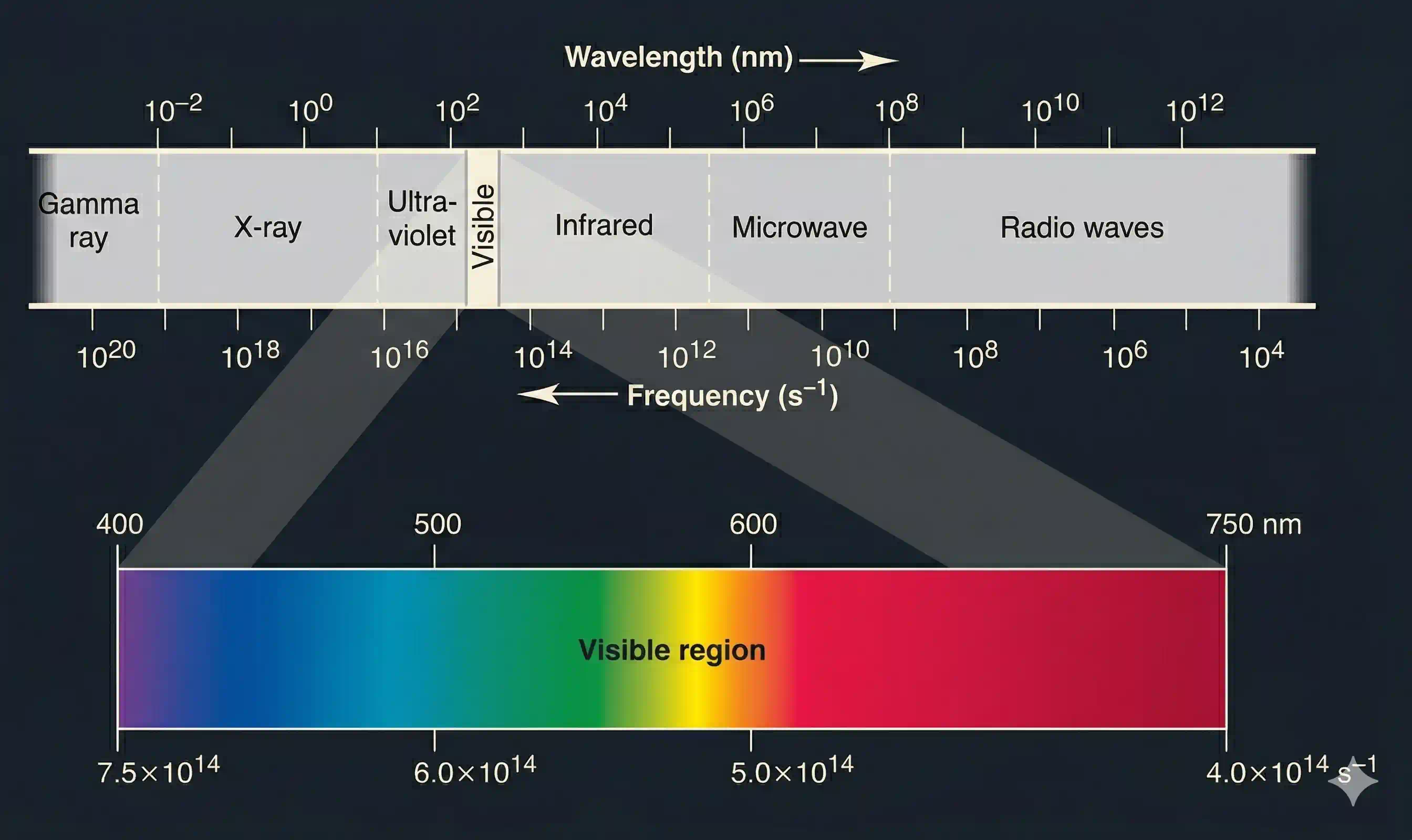
What is the electromagnetic spectrum?
Listen to the audio explanation
Wave Behaviour: Diffraction and Interference
The strongest evidence that light is a wave — and not a stream of particles (as Newton believed) — comes from two behaviours that only waves can show:
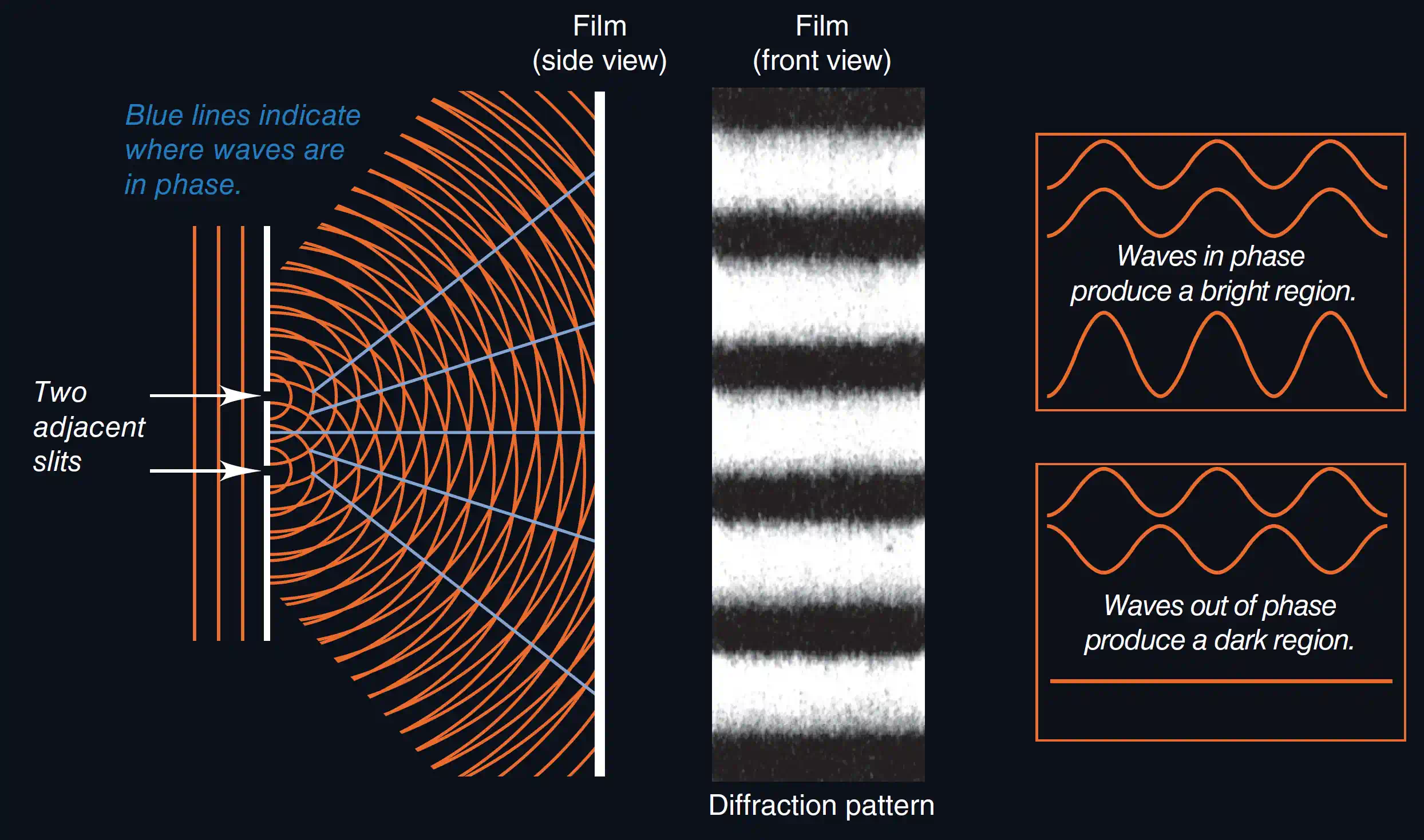
Diffraction is the bending of a wave around an obstacle or through a narrow opening. You hear someone talking around a corner because sound waves (wavelength ~1 m) diffract around walls. Light also diffracts, but the wavelength is so tiny (~500 nm) that you need a very fine grating to observe it.
Interference occurs when two waves meet. If they arrive in phase (crest meets crest), they reinforce each other — constructive interference, producing a brighter light. If they arrive out of phase (crest meets trough), they cancel — destructive interference, producing darkness.
These two phenomena are the hallmark of wave behaviour. Particles hitting each other produce markedly different patterns, as you will see in the simulation below.
Loading simulator…
Problem
The Vividh Bharati station of All India Radio, Delhi, broadcasts on a frequency of 1368 kHz (kilohertz). Calculate the wavelength of the electromagnetic radiation emitted by the transmitter. Which part of the electromagnetic spectrum does it belong to?
Problem
The wavelength range of the visible spectrum extends from violet (400 nm) to red (750 nm). Express these wavelengths in terms of frequencies (Hz). (1 nm = 10⁻⁹ m)
Problem
Calculate (a) wavenumber and (b) frequency of yellow radiation having wavelength 5800 Å. (1 Å = 10⁻¹⁰ m)
Q1.A microwave oven operates at a frequency of 2.45 GHz. What is the wavelength of this radiation?
At this exact moment, radio waves from hundreds of broadcast towers, WiFi routers, mobile base stations, and satellites are passing through your body. Your TV remote fires invisible infrared pulses. The sun is bombarding you with ultraviolet rays. Your own body emits infrared heat radiation. All of these — along with visible light, X-rays, and gamma rays — are the same type of wave, just at wildly different frequencies.
What is a Wave?
A wave is a disturbance that travels through space or matter, carrying energy from one place to another — without carrying matter itself. Think of ripples on a pond: the water molecules don't travel toward the shore, but the energy of the disturbance does.
Waves come in two fundamental types:
- Mechanical waves — need a physical medium to travel. Sound waves, water waves, and earthquake waves are mechanical. They cannot travel through a vacuum (empty space). That's why space is completely silent.
- Electromagnetic (EM) waves — need no medium at all. They can travel through the vacuum of space. That's how sunlight reaches us across 150 million kilometres of empty space.

Maxwell's Discovery: Light is an Electromagnetic Wave
In 1870, James Clerk Maxwell published equations showing that a charged particle undergoing acceleration creates oscillating electric and magnetic fields that propagate outward as a wave. These waves travel at exactly the speed of light — leading Maxwell to conclude that light itself is an electromagnetic wave.
An electromagnetic wave has two key features:
- Perpendicular fields: The electric field (E) and the magnetic field (B) oscillate at right angles to each other, and both are perpendicular to the direction the wave travels.
- Self-sustaining: A changing E field creates a B field, which creates an E field — the wave sustains itself through empty space with no medium needed.

Key Properties of Electromagnetic Waves
- No medium required — travel through vacuum; sound cannot.
- Constant speed in vacuum — all EM waves travel at m s⁻¹, regardless of wavelength or frequency. (They slow slightly inside materials like glass or water.)
- Transverse waves — the oscillations are perpendicular to the direction of travel.
- Characterised by frequency (ν) and wavelength (λ) — related by:
- Carry energy — the energy of each photon is , where J s (Planck's constant). Higher frequency = more energy per photon.
Frequency, Wavelength, and the Speed of Light

Wavelength (λ) is the distance between two successive crests (or troughs) of the wave. It is easy to visualise — you can literally measure it on a wave diagram.
Frequency (ν) is harder to picture because it describes time, not space. It is the number of complete wave cycles that pass a fixed point per second. Its SI unit is the hertz (Hz), named after Heinrich Hertz, where Hz s.
For all EM waves in vacuum:
where m s (often rounded to m s).
Wavenumber () is another useful quantity in spectroscopy. It is the number of wavelengths per unit length:
Its SI unit is m, but the commonly used unit in spectroscopy is cm.
Equations 2.5 & 2.6
c = 3.0 × 10⁸ m s⁻¹ · h = 6.626 × 10⁻³⁴ J s · ν in Hz · λ in m · ν̄ in m⁻¹ (or cm⁻¹)
See It: The EM Wave Explorer
Drag the slider to move through the electromagnetic spectrum from radio waves to gamma rays. Watch how the wavelength visible in the animation shrinks as frequency rises. Click any region label to jump straight to it.
Loading simulator…
The Electromagnetic Spectrum at a Glance
All electromagnetic radiation is the same type of wave — but the frequency range spans more than 20 orders of magnitude. Different regions of the spectrum interact with matter in completely different ways, which is why radio waves pass harmlessly through walls while gamma rays can kill cancer cells.

What is the electromagnetic spectrum?
Listen to the audio explanation
Wave Behaviour: Diffraction and Interference
The strongest evidence that light is a wave — and not a stream of particles (as Newton believed) — comes from two behaviours that only waves can show:
Diffraction is the bending of a wave around an obstacle or through a narrow opening. You hear someone talking around a corner because sound waves (wavelength ~1 m) diffract around walls. Light also diffracts, but the wavelength is so tiny (~500 nm) that you need a very fine grating to observe it.
Interference occurs when two waves meet. If they arrive in phase (crest meets crest), they reinforce each other — constructive interference, producing a brighter light. If they arrive out of phase (crest meets trough), they cancel — destructive interference, producing darkness.
These two phenomena are the hallmark of wave behaviour. Particles hitting each other produce markedly different patterns, as you will see in the simulation below.

Loading simulator…
Problem
The Vividh Bharati station of All India Radio, Delhi, broadcasts on a frequency of 1368 kHz (kilohertz). Calculate the wavelength of the electromagnetic radiation emitted by the transmitter. Which part of the electromagnetic spectrum does it belong to?
Problem
The wavelength range of the visible spectrum extends from violet (400 nm) to red (750 nm). Express these wavelengths in terms of frequencies (Hz). (1 nm = 10⁻⁹ m)
Problem
Calculate (a) wavenumber and (b) frequency of yellow radiation having wavelength 5800 Å. (1 Å = 10⁻¹⁰ m)
Q1.A microwave oven operates at a frequency of 2.45 GHz. What is the wavelength of this radiation?