Planck's Quantum Theory and the Photoelectric Effect
How four stubborn experiments broke classical physics — and why light delivers its energy in indivisible packets
On a cold winter evening in Berlin, a 42-year-old physicist named Max Planck stood before the German Physical Society and read out a paper that he himself did not believe. Buried inside it was a single mathematical trick — an assumption so strange that Planck called it "an act of desperation." He had no idea he had just split physics in half. Everything before that evening is called classical physics. Everything after is called quantum physics. And it all began because hot iron rods weren't behaving the way they should.
Four Cracks in the Classical Picture
By the late 1800s, physicists were quietly confident. Newton's mechanics explained the motion of planets and projectiles. Maxwell's equations explained electricity, magnetism, and light. Many leading scientists genuinely believed that physics was almost finished — all that remained was to add more decimal places to the constants.
Then four experiments began producing results that nothing in classical physics could explain. Each crack, on its own, seemed like a nuisance. Together, they demolished the entire house.
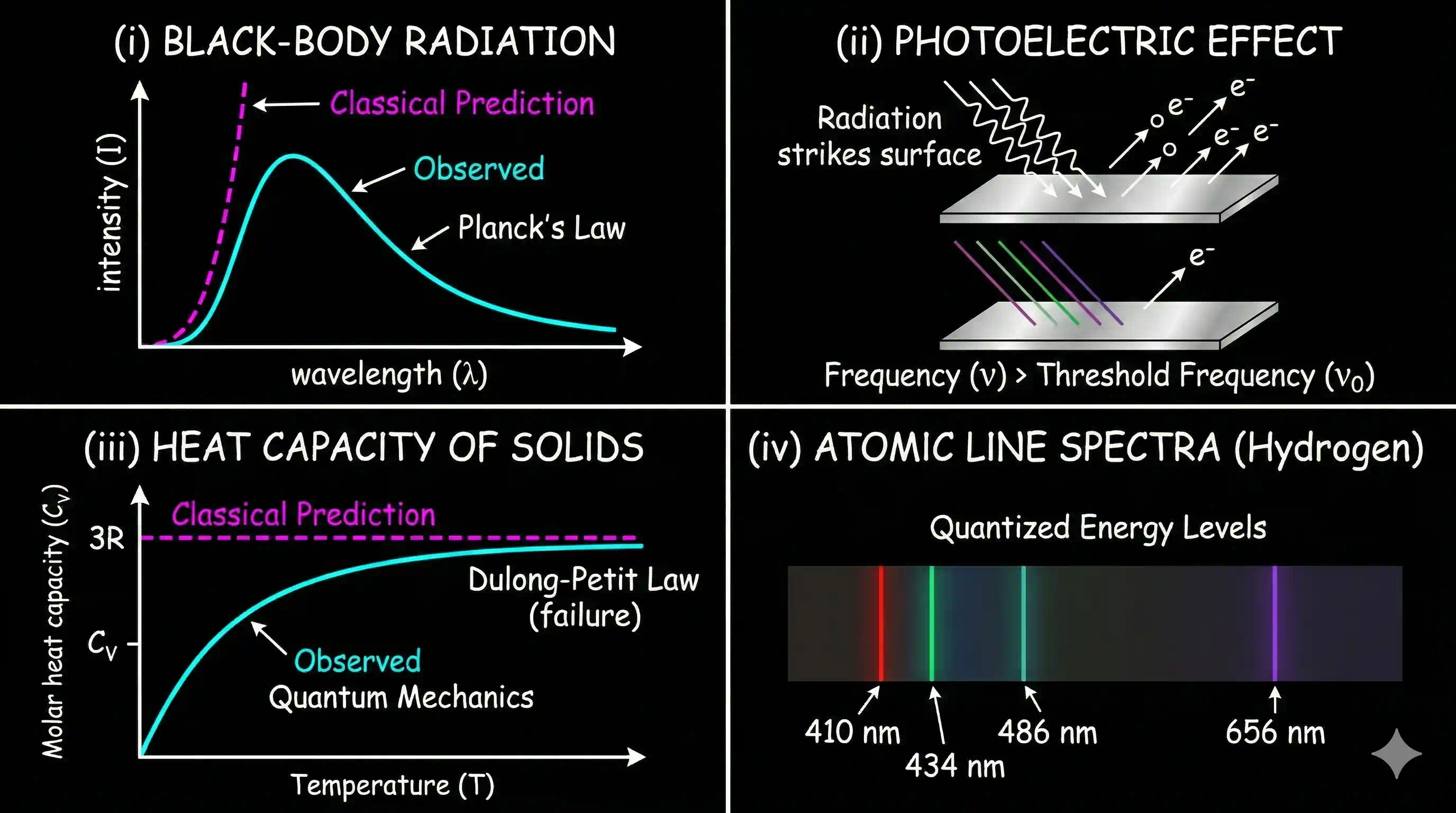
Crack 1 -Black Body Radiation
Understanding black body radiation
Listen to the audio explanation
Heat a piece of iron in a furnace. At first it stays dark. As it warms, it begins glowing a dull red. Push it hotter, and the red brightens into orange, then yellow. At extreme temperatures the same rod glows blue-white.
Why does the colour of emitted light depend entirely on temperature? This looks like a trivial question. It turned out to require inventing quantum mechanics to answer.
Hot objects emit electromagnetic radiations over a wide range of wavelengths. At high temperatures, an appreciable proportion of radiation is in the visible region of the spectrum. As the temperature is raised, a higher proportion of short wavelength (blue light) is generated. For example, when an iron rod is heated in a furnace, it first turns to dull red and then progressively becomes more and more red as the temperature increases. As this is heated further, the radiation emitted becomes white and then becomes blue as the temperature becomes very high.
This means that red radiation is most intense at a particular temperature and the blue radiation is more intense at another temperature. This means intensities of radiations of different wavelengths emitted by hot body depend upon its temperature.
By late 1850’s it was known that objects made of different material and kept at different temperatures emit different amount of radiation. Also, when the surface of an object is irradiated with light (electromagnetic radiation), a part of radiant energy is generally reflected as such, a part is absorbed and a part of it is transmitted. The reason for incomplete absorption is that ordinary objects are as a rule, imperfect absorbers of radiation. An ideal body, which emits and absorbs radiations of all frequencies uniformly, is called a black body and the radiation emitted by such a body is called black body radiation.
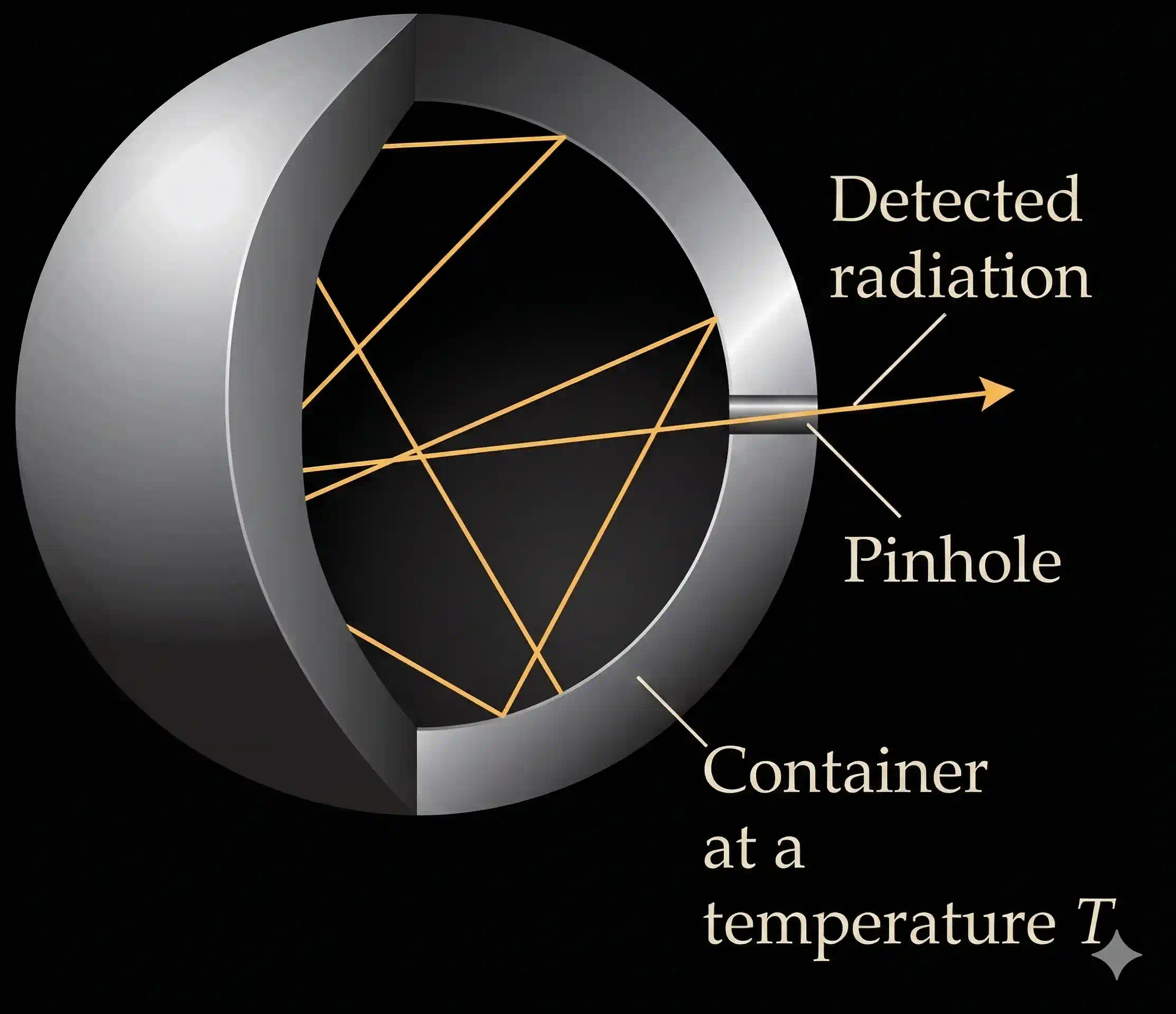
No real material is a perfect black body, but some come close. A surface of lamp-black (finely divided carbon) absorbs about 97 % of incident light. For laboratory work the standard trick is cleverer: take a hollow cavity with polished interior walls and a tiny hole on one side. Any radiation that enters the hole bounces around inside, losing a little to the walls on each reflection, and is almost certainly absorbed before it can find its way back out.
Furthermore, a black body is in thermal equilibrium with its surroundings. It radiates same amount of energy per unit area as it absorbs from its surrounding in any given time. The amount of light emitted (intensity of radiation) from a black body and its spectral distribution depends only on its temperature. At a given temperature, intensity of radiation emitted increases with the increase of wavelength, reaches a maximum value at a given wavelength and then starts decreasing with further increase of wavelength.
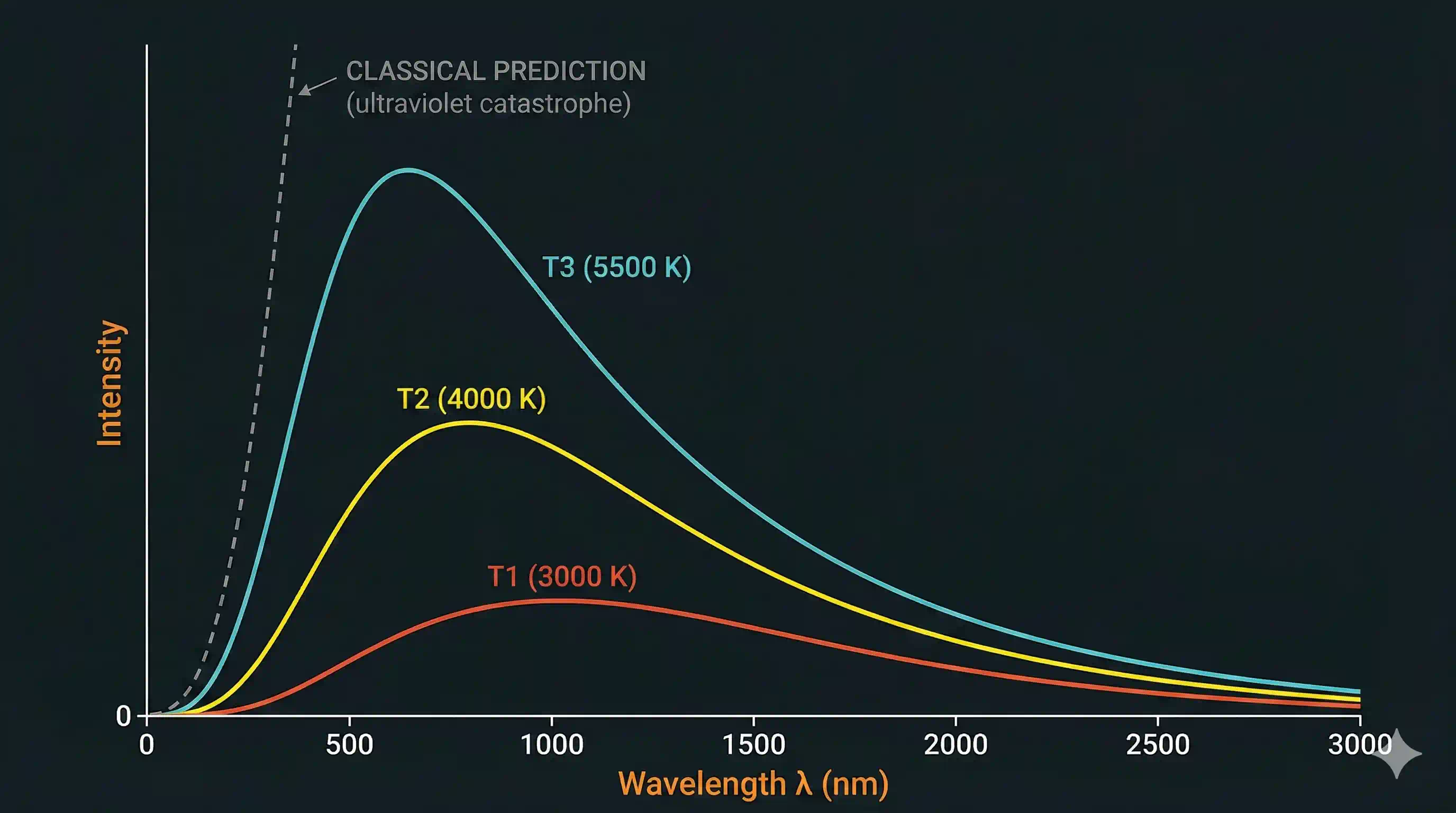
- The peak of the curve shifts to shorter wavelengths as rises. A filament bulb at about 2700 K peaks deep in the infrared. The Sun at 5800 K peaks in the visible green-yellow band. A blue giant star at 20 000 K peaks in the ultraviolet. This is why "red hot" is actually cooler than "white hot", and why truly hot objects look bluish. The precise rule is Wien's displacement law: m K.
- The total emitted power climbs explosively with temperature. Double the temperature and the total radiated power goes up by a factor of sixteen. Formally, power per unit area — the Stefan–Boltzmann law. This is why a small bump in a filament's temperature causes a huge leap in brightness, and in your electricity bill.
Classical physics — Maxwell's waves together with the equipartition theorem of statistical mechanics — made a definite prediction about what these curves should look like. The prediction was a disaster. According to classical theory, the intensity at short wavelengths should rise without limit, so that any heated object would radiate infinite energy at ultraviolet frequencies and beyond. This absurd result was nicknamed the "ultraviolet catastrophe". Of course nothing of the sort actually happens — a warm desk does not pour out lethal UV. The observed curves peak and fall cleanly. The gap between the real spectrum and the classical prediction (dashed line above) is exactly the size of the hole that classical physics had no way to plug.
From Filament to LED — a trillion-rupee consequence of the Planck curve
It is tempting to treat black-body radiation as an abstract physics curiosity. It is anything but. Every ordinary filament bulb is a miniature black body. Inside the glass envelope, a tungsten coil is heated by electric current to roughly 2700 K. At that temperature, Wien's law places the peak of its emission curve at about 1100 nm — deep in the infrared, far below the visible band. Only a sliver of the Planck curve — roughly 5 % of the electrical energy supplied — actually comes out as light your eyes can use. The other 95 % is radiated as heat. A 60 W "light" bulb is really a 3 W lamp bolted to a 57 W space-heater.
This is not a design flaw that clever engineering can fix. It is a direct consequence of the black-body curve. To make an incandescent bulb peak in the visible, you would have to push the filament to around 5800 K — the surface temperature of the Sun — at which point tungsten would have long since vaporised.
Compact fluorescent lamps (CFLs) sidestep the problem entirely. Instead of heating something until it glows thermally, a CFL passes an electric current through low-pressure mercury vapour. Excited mercury atoms emit sharp ultraviolet lines — not thermal black-body radiation. The UV then strikes a phosphor coating on the inside of the tube, which absorbs the UV and re-emits it as visible light. Because the emission is no longer constrained by the Planck curve, roughly 20 % of the input electricity becomes useful visible light. A 15 W CFL can replace a 60 W incandescent at the same brightness.
LEDs go further still. A light-emitting diode does not radiate thermally in any meaningful sense — it is not a hot object at all. When an electron drops across the band gap of a semiconductor, it releases a photon whose energy equals the band-gap energy, producing a narrow band of light centred on exactly the wavelength the chip was engineered for. A modern white LED reaches 40 – 50 % efficiency — eight to ten times better than a filament bulb — which is why every serious lighting installation in the world is switching to LEDs. The long arc of lighting technology is simple: we have spent a century escaping from the Planck curve.
Loading simulator…
Crack 2 — Light kicks electrons off metal, but only sometimes
In 1887, the German physicist Heinrich Hertz was experimenting with radio waves (ironically, trying to prove Maxwell's wave theory of light). He noticed something odd: sparks jumped more easily between metal electrodes when ultraviolet light was shining on them. Light was somehow knocking electrons out of the metal.
This phenomenon — called the photoelectric effect — was easy to describe but impossible to explain with wave theory:
- Brighter red light does nothing, no matter how long you shine it. But dim ultraviolet light ejects electrons instantly.
- Below a certain threshold frequency, no electrons come out. Period. Above it, they fly out immediately.
- Brighter light (of the right colour) ejects more electrons, but they all carry the same kinetic energy.
Classical wave theory said: light is a continuous wave of energy. An electron in the metal should just sit there and slowly absorb energy from the wave until it has enough to escape. Any colour should work given enough time. Brighter light should give more energy to each electron. Every single one of those predictions is wrong.
Crack 3 — Solids forget how to store heat near absolute zero
Classical thermodynamics predicted that every solid should have roughly the same heat capacity per mole — the Dulong-Petit law. At room temperature this works beautifully. But as experiments pushed toward cryogenic temperatures, the heat capacity of solids began dropping off sharply, approaching zero as . Classical physics had no mechanism for this. Something about the way atoms stored vibrational energy was changing at low temperatures — as if the "slots" for vibration were being switched off one by one.
Crack 4 — Hydrogen only emits a handful of colours
When you pass an electric discharge through hydrogen gas and split the emitted light through a prism, you don't get a continuous rainbow. You get a few razor-sharp coloured lines at very specific wavelengths — and nothing else in between. Every element has its own unique fingerprint of lines. Classical physics predicted a continuous smear. Reality delivered a barcode.
We will return to line spectra in the next page. For now, keep it in mind as the fourth mystery that classical physics couldn't touch.
What all four cracks have in common
Every one of these experiments seemed to suggest that energy wasn't smooth — that nature was allowing energy to be exchanged only in specific, discrete amounts, not as a continuous flow. It was as if a mountain climber could only stand on specific ledges, not anywhere on the slope.
Planck's Gamble: Energy Comes in Packets
Planck had been wrestling with the black body problem for six years. He was a deeply conservative physicist — he believed in classical mechanics and had no desire to overthrow it. He was simply trying to find any mathematical formula that would fit the experimental curves.
In October 1900, he found one. His formula worked perfectly. But to derive it, he had to make an assumption so bizarre that even he refused to accept it at first: he assumed that the atoms in the walls of the black body could only absorb or emit energy in discrete bundles — never as a continuous stream. The size of each bundle was proportional to the frequency of the radiation:
where is a fundamental constant of nature, now called Planck's constant:
Planck called these indivisible bundles quanta (singular: quantum). A quantum is the smallest possible packet of energy at a given frequency. You can have one quantum, or two, or ten — but never 1.5 quanta. Energy comes in whole-number multiples of .
Planck's Quantum Hypothesis
Energy cannot flow continuously. It can only be emitted or absorbed in discrete packets called quanta, each of size:
Planck's Quantum Relation
h is one of the most fundamental constants in all of physics, on par with c and G.
Here is the most striking thing about the story: Planck himself spent the next ten years trying to derive his formula from ordinary classical physics, without invoking quanta. He failed every time. Slowly, reluctantly, he came to accept that the universe really does hand out energy in tiny indivisible chunks. Later in life he wrote: "My futile attempts to fit the elementary quantum of action somehow into classical theory continued for a number of years and cost me a great deal of effort."
Quantum physics was born, and its own father was embarrassed by it.
See It: Wave vs. Photon
Use the slider to move through the electromagnetic spectrum from infrared to ultraviolet. On the left, you see light as a classical continuous wave — an unbroken stream of energy, where you could in principle "take off" any amount you want. On the right, you see the same frequency treated as Planck quanta — the same energy arrives chopped into discrete bouncing packets labelled "hν", each one indivisible.
Watch what happens as you raise the frequency: on the left the wave just oscillates faster. On the right, each individual packet gets bigger, because the energy of each quantum is . This visual difference is the whole story — and it is the foundation of the photoelectric effect that follows.
Loading simulator…
Einstein Takes Planck Seriously (1905)
1905 was Albert Einstein's annus mirabilis — his "miracle year". He was 26 years old, working as a technical assistant at the Swiss patent office in Bern, and in that single year he published four papers that each rewrote a branch of physics:
- A paper on Brownian motion, providing direct proof that atoms exist.
- A paper on special relativity, showing that space and time are one fabric.
- A paper deriving .
- A paper on the photoelectric effect — which, ironically, is the one that would later win him the Nobel Prize in 1921.
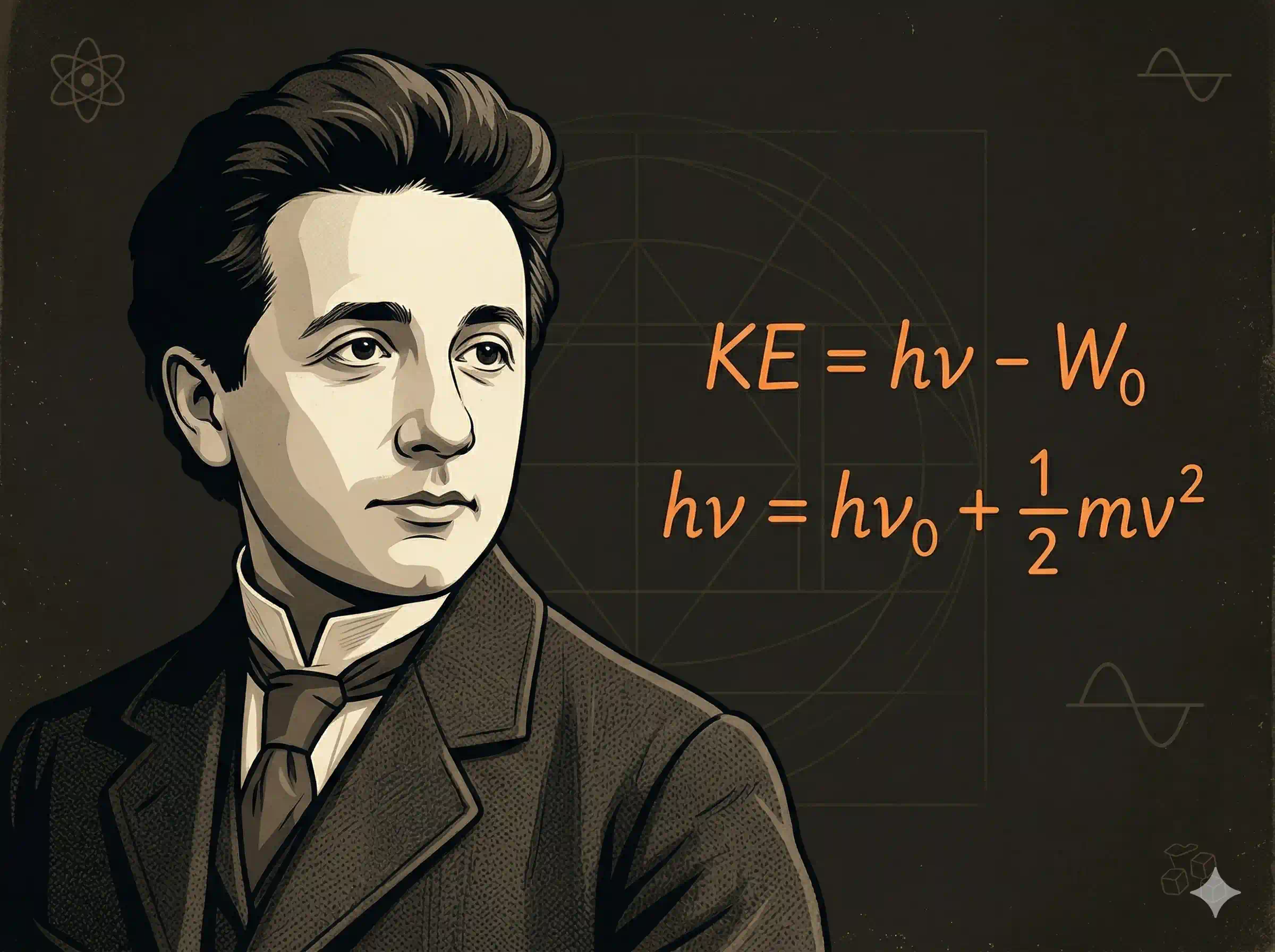
Einstein's insight in that fourth paper was audacious. Planck had assumed that atoms emit and absorb energy in packets. Einstein went further: he proposed that light itself is made of packets. The photoelectric effect, he argued, was simple once you accepted this.
The Photoelectric Effect, Unpacked
Here is the experimental setup. A clean metal surface (potassium, rubidium, or caesium works well — they hold their outer electrons loosely) is placed inside a vacuum chamber. A beam of light of a specific frequency is shone on the metal. If electrons are ejected, a detector counts them and measures their kinetic energy.
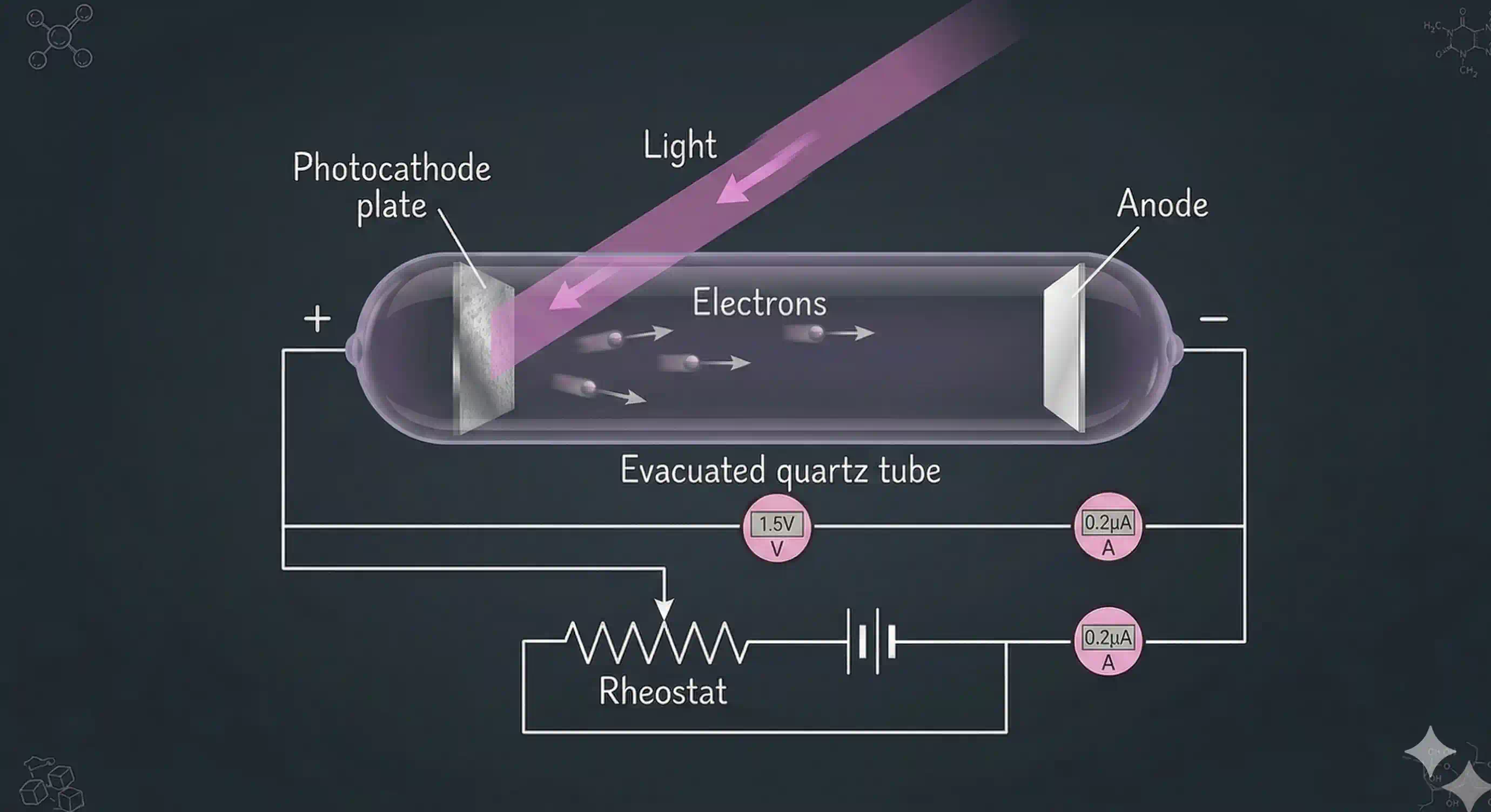
What the experiments revealed
Careful measurements — first by Hertz in 1887, then refined by Lenard, Millikan, and others over two decades — produced four rock-solid observations:
- Instant ejection. Electrons are ejected the moment light of the right frequency hits the surface. There is no detectable time lag — less than a billionth of a second.
- Brightness controls count, not speed. Doubling the intensity ejects twice as many electrons, but each one carries the same kinetic energy.
- Frequency controls speed, not count. Increasing the frequency of the light gives the ejected electrons more kinetic energy — but does not change how many are ejected.
- Sharp threshold. For every metal there is a minimum frequency — the threshold frequency — below which no electrons are ejected at all, no matter how long or how intensely you shine the light.
Why classical wave theory fails spectacularly
Classical wave theory treats light as a continuous flow of energy. An electron buried in the metal is held there by a binding energy called the work function () — think of it as the depth of a well the electron has to climb out of.
If energy arrives continuously, classical physics predicts four things:
- Prediction: Any colour should eventually work — just keep the light on long enough, let the electron slowly accumulate energy from the wave, and it will escape. Reality: Below , electrons never come out. You can leave the lamp on for days. Nothing happens.
- Prediction: Brighter light should give each electron more energy, so ejected electrons should be faster. Reality: Brighter light ejects more electrons, but each one has the same speed. Faster electrons only come from higher frequency light, not brighter light.
- Prediction: There should be a measurable time lag as the electron absorbs wave energy bit by bit. Reality: Ejection is instantaneous.
- Prediction: Energy should flow into the whole metal uniformly. Reality: Individual electrons pop out at discrete points.
Four wrong predictions, zero right ones. Wave theory is shredded.
The raindrop and the bucket
Imagine an electron as a bucket sitting on a ledge at the top of a wall. The bucket holds water (energy). If the bucket contains enough water to pour over the lip, it tips and the water (electron) escapes.

Einstein's Equation
Einstein wrote down conservation of energy for one photon hitting one electron: the incoming photon's energy equals the energy needed to escape the metal plus whatever is left over as kinetic energy of the freed electron.
Einstein's Photoelectric Equation
ν is the frequency of incident light, ν₀ is the metal's threshold frequency, W₀ is the work function.
Three consequences fall out immediately:
- Below threshold: if , then — the photon doesn't carry enough energy to lift one electron out of its well, and nothing happens. No matter how bright the light is, each individual raindrop is still too small.
- At threshold: , and electrons just barely escape with zero kinetic energy.
- Above threshold: the excess energy becomes kinetic energy of the ejected electron. Kinetic energy therefore depends only on frequency, not intensity.
Every puzzling observation is now obvious. Einstein's prediction — a perfectly straight line when you plot KE against frequency, with slope equal to Planck's constant — was verified experimentally by Robert Millikan over 1914–1916. Millikan initially tried to disprove Einstein's theory, spent years on it, and ended up confirming it so thoroughly that he won the 1923 Nobel Prize for his trouble.
Work functions of selected metals. Lower W₀ means the metal gives up electrons more easily.
| Metal | Work Function W₀ (eV) | Threshold Wavelength (nm) | Region |
|---|---|---|---|
| Caesium (Cs) | 2.14 | 579 | Visible (yellow) |
| Potassium (K) | 2.25 | 551 | Visible (green) |
| Sodium (Na) | 2.3 | 539 | Visible (green) |
| Lithium (Li) | 2.42 | 512 | Visible (green) |
| Magnesium (Mg) | 3.7 | 335 | Ultraviolet |
| Silver (Ag) | 4.3 | 288 | Ultraviolet |
| Copper (Cu) | 4.8 | 258 | Ultraviolet |
See It: The Photoelectric Effect
Numbers in a table are one thing — watching photons actually kick electrons out of real metals is another. In the simulator below, you can pick a metal, change the frequency of incoming light, and change the intensity (how bright the beam is). Two live graphs plot kinetic energy against frequency and against intensity so you can see the relationships Einstein predicted.
Things worth trying:
- Start with Cs and crank the frequency down until electrons stop ejecting. Note the threshold.
- Now swap to Cu at the same frequency — nothing happens. Its work function is too high.
- Hold the frequency above threshold and drag the intensity from 1 → 10. More electrons fly out, but each one still carries the same KE.
- Watch the KE-vs-ν graph: the curve is flat at zero until ν = ν₀, then rises as a perfectly straight line — exactly as Einstein predicted.
Loading simulator…
Where you meet the photoelectric effect every day
- Solar panels — photons from the sun knock electrons loose inside a silicon cell. Those freed electrons become the current that powers your phone charger.
- Digital camera sensors (CCD / CMOS) — each pixel is a tiny photoelectric cell. Photons create electrons; the camera counts them.
- Night-vision goggles — IR photons are amplified into visible-range electrons that illuminate a phosphor screen.
- Photomultiplier tubes — the devices inside medical PET scanners and particle physics detectors, sensitive enough to see a single photon.
- Automatic door sensors and smoke alarms — a beam of light crossing a photocell; break the beam, and the photocurrent drops.
Every time you use any of these, you are personally witnessing Planck and Einstein's quanta at work.

The Dual Nature of Light
There is now a serious problem. Diffraction and interference (the previous page) proved that light is a wave. The photoelectric effect proves that light is a particle. Both sets of experiments are rock-solid. Both conclusions are right.
The resolution, which took decades to accept, is this: light has a dual nature. It propagates through space as a wave — that is how it diffracts, interferes, and travels at . But it interacts with matter as a particle — a photon — that delivers its energy all at once, in a single indivisible chunk.
Which face light shows you depends entirely on the experiment you perform. Pass it through two slits and you see waves. Point it at a metal plate and you see particles. Nature is not being inconsistent — we just don't have a single everyday word that describes something that is both. In the next chapter, you will discover that electrons do exactly the same thing. Wave-particle duality isn't a quirk of light. It's a basic property of reality at the atomic scale.
Worked Examples
Problem
Calculate energy of one mole of photons of radiation whose frequency is Hz.
Problem
A 100 W bulb emits monochromatic light of wavelength 400 nm. Calculate the number of photons emitted per second by the bulb.
Problem
When electromagnetic radiation of wavelength 300 nm falls on the surface of sodium, electrons are emitted with a kinetic energy of J mol. What is the minimum energy needed to remove an electron from sodium? What is the maximum wavelength that will cause a photoelectron to be emitted?
Problem
The threshold frequency for a metal is s. Calculate the kinetic energy of an electron emitted when radiation of frequency s hits the metal.
Q1.Which observation could NOT be explained by the classical wave theory of light?
On a cold winter evening in Berlin, a 42-year-old physicist named Max Planck stood before the German Physical Society and read out a paper that he himself did not believe. Buried inside it was a single mathematical trick — an assumption so strange that Planck called it "an act of desperation." He had no idea he had just split physics in half. Everything before that evening is called classical physics. Everything after is called quantum physics. And it all began because hot iron rods weren't behaving the way they should.
Four Cracks in the Classical Picture
By the late 1800s, physicists were quietly confident. Newton's mechanics explained the motion of planets and projectiles. Maxwell's equations explained electricity, magnetism, and light. Many leading scientists genuinely believed that physics was almost finished — all that remained was to add more decimal places to the constants.
Then four experiments began producing results that nothing in classical physics could explain. Each crack, on its own, seemed like a nuisance. Together, they demolished the entire house.

Crack 1 -Black Body Radiation
Understanding black body radiation
Listen to the audio explanation
Heat a piece of iron in a furnace. At first it stays dark. As it warms, it begins glowing a dull red. Push it hotter, and the red brightens into orange, then yellow. At extreme temperatures the same rod glows blue-white.
Why does the colour of emitted light depend entirely on temperature? This looks like a trivial question. It turned out to require inventing quantum mechanics to answer.
Hot objects emit electromagnetic radiations over a wide range of wavelengths. At high temperatures, an appreciable proportion of radiation is in the visible region of the spectrum. As the temperature is raised, a higher proportion of short wavelength (blue light) is generated. For example, when an iron rod is heated in a furnace, it first turns to dull red and then progressively becomes more and more red as the temperature increases. As this is heated further, the radiation emitted becomes white and then becomes blue as the temperature becomes very high.
This means that red radiation is most intense at a particular temperature and the blue radiation is more intense at another temperature. This means intensities of radiations of different wavelengths emitted by hot body depend upon its temperature.
By late 1850’s it was known that objects made of different material and kept at different temperatures emit different amount of radiation. Also, when the surface of an object is irradiated with light (electromagnetic radiation), a part of radiant energy is generally reflected as such, a part is absorbed and a part of it is transmitted. The reason for incomplete absorption is that ordinary objects are as a rule, imperfect absorbers of radiation. An ideal body, which emits and absorbs radiations of all frequencies uniformly, is called a black body and the radiation emitted by such a body is called black body radiation.

No real material is a perfect black body, but some come close. A surface of lamp-black (finely divided carbon) absorbs about 97 % of incident light. For laboratory work the standard trick is cleverer: take a hollow cavity with polished interior walls and a tiny hole on one side. Any radiation that enters the hole bounces around inside, losing a little to the walls on each reflection, and is almost certainly absorbed before it can find its way back out.
Furthermore, a black body is in thermal equilibrium with its surroundings. It radiates same amount of energy per unit area as it absorbs from its surrounding in any given time. The amount of light emitted (intensity of radiation) from a black body and its spectral distribution depends only on its temperature. At a given temperature, intensity of radiation emitted increases with the increase of wavelength, reaches a maximum value at a given wavelength and then starts decreasing with further increase of wavelength.

- The peak of the curve shifts to shorter wavelengths as rises. A filament bulb at about 2700 K peaks deep in the infrared. The Sun at 5800 K peaks in the visible green-yellow band. A blue giant star at 20 000 K peaks in the ultraviolet. This is why "red hot" is actually cooler than "white hot", and why truly hot objects look bluish. The precise rule is Wien's displacement law: m K.
- The total emitted power climbs explosively with temperature. Double the temperature and the total radiated power goes up by a factor of sixteen. Formally, power per unit area — the Stefan–Boltzmann law. This is why a small bump in a filament's temperature causes a huge leap in brightness, and in your electricity bill.
Classical physics — Maxwell's waves together with the equipartition theorem of statistical mechanics — made a definite prediction about what these curves should look like. The prediction was a disaster. According to classical theory, the intensity at short wavelengths should rise without limit, so that any heated object would radiate infinite energy at ultraviolet frequencies and beyond. This absurd result was nicknamed the "ultraviolet catastrophe". Of course nothing of the sort actually happens — a warm desk does not pour out lethal UV. The observed curves peak and fall cleanly. The gap between the real spectrum and the classical prediction (dashed line above) is exactly the size of the hole that classical physics had no way to plug.
From Filament to LED — a trillion-rupee consequence of the Planck curve
It is tempting to treat black-body radiation as an abstract physics curiosity. It is anything but. Every ordinary filament bulb is a miniature black body. Inside the glass envelope, a tungsten coil is heated by electric current to roughly 2700 K. At that temperature, Wien's law places the peak of its emission curve at about 1100 nm — deep in the infrared, far below the visible band. Only a sliver of the Planck curve — roughly 5 % of the electrical energy supplied — actually comes out as light your eyes can use. The other 95 % is radiated as heat. A 60 W "light" bulb is really a 3 W lamp bolted to a 57 W space-heater.
This is not a design flaw that clever engineering can fix. It is a direct consequence of the black-body curve. To make an incandescent bulb peak in the visible, you would have to push the filament to around 5800 K — the surface temperature of the Sun — at which point tungsten would have long since vaporised.
Compact fluorescent lamps (CFLs) sidestep the problem entirely. Instead of heating something until it glows thermally, a CFL passes an electric current through low-pressure mercury vapour. Excited mercury atoms emit sharp ultraviolet lines — not thermal black-body radiation. The UV then strikes a phosphor coating on the inside of the tube, which absorbs the UV and re-emits it as visible light. Because the emission is no longer constrained by the Planck curve, roughly 20 % of the input electricity becomes useful visible light. A 15 W CFL can replace a 60 W incandescent at the same brightness.
LEDs go further still. A light-emitting diode does not radiate thermally in any meaningful sense — it is not a hot object at all. When an electron drops across the band gap of a semiconductor, it releases a photon whose energy equals the band-gap energy, producing a narrow band of light centred on exactly the wavelength the chip was engineered for. A modern white LED reaches 40 – 50 % efficiency — eight to ten times better than a filament bulb — which is why every serious lighting installation in the world is switching to LEDs. The long arc of lighting technology is simple: we have spent a century escaping from the Planck curve.
Loading simulator…
Crack 2 — Light kicks electrons off metal, but only sometimes
In 1887, the German physicist Heinrich Hertz was experimenting with radio waves (ironically, trying to prove Maxwell's wave theory of light). He noticed something odd: sparks jumped more easily between metal electrodes when ultraviolet light was shining on them. Light was somehow knocking electrons out of the metal.
This phenomenon — called the photoelectric effect — was easy to describe but impossible to explain with wave theory:
- Brighter red light does nothing, no matter how long you shine it. But dim ultraviolet light ejects electrons instantly.
- Below a certain threshold frequency, no electrons come out. Period. Above it, they fly out immediately.
- Brighter light (of the right colour) ejects more electrons, but they all carry the same kinetic energy.
Classical wave theory said: light is a continuous wave of energy. An electron in the metal should just sit there and slowly absorb energy from the wave until it has enough to escape. Any colour should work given enough time. Brighter light should give more energy to each electron. Every single one of those predictions is wrong.
Crack 3 — Solids forget how to store heat near absolute zero
Classical thermodynamics predicted that every solid should have roughly the same heat capacity per mole — the Dulong-Petit law. At room temperature this works beautifully. But as experiments pushed toward cryogenic temperatures, the heat capacity of solids began dropping off sharply, approaching zero as . Classical physics had no mechanism for this. Something about the way atoms stored vibrational energy was changing at low temperatures — as if the "slots" for vibration were being switched off one by one.
Crack 4 — Hydrogen only emits a handful of colours
When you pass an electric discharge through hydrogen gas and split the emitted light through a prism, you don't get a continuous rainbow. You get a few razor-sharp coloured lines at very specific wavelengths — and nothing else in between. Every element has its own unique fingerprint of lines. Classical physics predicted a continuous smear. Reality delivered a barcode.
We will return to line spectra in the next page. For now, keep it in mind as the fourth mystery that classical physics couldn't touch.
What all four cracks have in common
Every one of these experiments seemed to suggest that energy wasn't smooth — that nature was allowing energy to be exchanged only in specific, discrete amounts, not as a continuous flow. It was as if a mountain climber could only stand on specific ledges, not anywhere on the slope.
Planck's Gamble: Energy Comes in Packets
Planck had been wrestling with the black body problem for six years. He was a deeply conservative physicist — he believed in classical mechanics and had no desire to overthrow it. He was simply trying to find any mathematical formula that would fit the experimental curves.
In October 1900, he found one. His formula worked perfectly. But to derive it, he had to make an assumption so bizarre that even he refused to accept it at first: he assumed that the atoms in the walls of the black body could only absorb or emit energy in discrete bundles — never as a continuous stream. The size of each bundle was proportional to the frequency of the radiation:
where is a fundamental constant of nature, now called Planck's constant:
Planck called these indivisible bundles quanta (singular: quantum). A quantum is the smallest possible packet of energy at a given frequency. You can have one quantum, or two, or ten — but never 1.5 quanta. Energy comes in whole-number multiples of .
Planck's Quantum Hypothesis
Energy cannot flow continuously. It can only be emitted or absorbed in discrete packets called quanta, each of size:
Planck's Quantum Relation
h is one of the most fundamental constants in all of physics, on par with c and G.
Here is the most striking thing about the story: Planck himself spent the next ten years trying to derive his formula from ordinary classical physics, without invoking quanta. He failed every time. Slowly, reluctantly, he came to accept that the universe really does hand out energy in tiny indivisible chunks. Later in life he wrote: "My futile attempts to fit the elementary quantum of action somehow into classical theory continued for a number of years and cost me a great deal of effort."
Quantum physics was born, and its own father was embarrassed by it.
See It: Wave vs. Photon
Use the slider to move through the electromagnetic spectrum from infrared to ultraviolet. On the left, you see light as a classical continuous wave — an unbroken stream of energy, where you could in principle "take off" any amount you want. On the right, you see the same frequency treated as Planck quanta — the same energy arrives chopped into discrete bouncing packets labelled "hν", each one indivisible.
Watch what happens as you raise the frequency: on the left the wave just oscillates faster. On the right, each individual packet gets bigger, because the energy of each quantum is . This visual difference is the whole story — and it is the foundation of the photoelectric effect that follows.
Loading simulator…
Einstein Takes Planck Seriously (1905)
1905 was Albert Einstein's annus mirabilis — his "miracle year". He was 26 years old, working as a technical assistant at the Swiss patent office in Bern, and in that single year he published four papers that each rewrote a branch of physics:
- A paper on Brownian motion, providing direct proof that atoms exist.
- A paper on special relativity, showing that space and time are one fabric.
- A paper deriving .
- A paper on the photoelectric effect — which, ironically, is the one that would later win him the Nobel Prize in 1921.
Einstein's insight in that fourth paper was audacious. Planck had assumed that atoms emit and absorb energy in packets. Einstein went further: he proposed that light itself is made of packets. The photoelectric effect, he argued, was simple once you accepted this.
The Photoelectric Effect, Unpacked
Here is the experimental setup. A clean metal surface (potassium, rubidium, or caesium works well — they hold their outer electrons loosely) is placed inside a vacuum chamber. A beam of light of a specific frequency is shone on the metal. If electrons are ejected, a detector counts them and measures their kinetic energy.

What the experiments revealed
Careful measurements — first by Hertz in 1887, then refined by Lenard, Millikan, and others over two decades — produced four rock-solid observations:
- Instant ejection. Electrons are ejected the moment light of the right frequency hits the surface. There is no detectable time lag — less than a billionth of a second.
- Brightness controls count, not speed. Doubling the intensity ejects twice as many electrons, but each one carries the same kinetic energy.
- Frequency controls speed, not count. Increasing the frequency of the light gives the ejected electrons more kinetic energy — but does not change how many are ejected.
- Sharp threshold. For every metal there is a minimum frequency — the threshold frequency — below which no electrons are ejected at all, no matter how long or how intensely you shine the light.
Why classical wave theory fails spectacularly
Classical wave theory treats light as a continuous flow of energy. An electron buried in the metal is held there by a binding energy called the work function () — think of it as the depth of a well the electron has to climb out of.
If energy arrives continuously, classical physics predicts four things:
- Prediction: Any colour should eventually work — just keep the light on long enough, let the electron slowly accumulate energy from the wave, and it will escape. Reality: Below , electrons never come out. You can leave the lamp on for days. Nothing happens.
- Prediction: Brighter light should give each electron more energy, so ejected electrons should be faster. Reality: Brighter light ejects more electrons, but each one has the same speed. Faster electrons only come from higher frequency light, not brighter light.
- Prediction: There should be a measurable time lag as the electron absorbs wave energy bit by bit. Reality: Ejection is instantaneous.
- Prediction: Energy should flow into the whole metal uniformly. Reality: Individual electrons pop out at discrete points.
Four wrong predictions, zero right ones. Wave theory is shredded.
The raindrop and the bucket
Imagine an electron as a bucket sitting on a ledge at the top of a wall. The bucket holds water (energy). If the bucket contains enough water to pour over the lip, it tips and the water (electron) escapes.

Einstein's Equation
Einstein wrote down conservation of energy for one photon hitting one electron: the incoming photon's energy equals the energy needed to escape the metal plus whatever is left over as kinetic energy of the freed electron.
Einstein's Photoelectric Equation
ν is the frequency of incident light, ν₀ is the metal's threshold frequency, W₀ is the work function.
Three consequences fall out immediately:
- Below threshold: if , then — the photon doesn't carry enough energy to lift one electron out of its well, and nothing happens. No matter how bright the light is, each individual raindrop is still too small.
- At threshold: , and electrons just barely escape with zero kinetic energy.
- Above threshold: the excess energy becomes kinetic energy of the ejected electron. Kinetic energy therefore depends only on frequency, not intensity.
Every puzzling observation is now obvious. Einstein's prediction — a perfectly straight line when you plot KE against frequency, with slope equal to Planck's constant — was verified experimentally by Robert Millikan over 1914–1916. Millikan initially tried to disprove Einstein's theory, spent years on it, and ended up confirming it so thoroughly that he won the 1923 Nobel Prize for his trouble.
Work functions of selected metals. Lower W₀ means the metal gives up electrons more easily.
| Metal | Work Function W₀ (eV) | Threshold Wavelength (nm) | Region |
|---|---|---|---|
| Caesium (Cs) | 2.14 | 579 | Visible (yellow) |
| Potassium (K) | 2.25 | 551 | Visible (green) |
| Sodium (Na) | 2.3 | 539 | Visible (green) |
| Lithium (Li) | 2.42 | 512 | Visible (green) |
| Magnesium (Mg) | 3.7 | 335 | Ultraviolet |
| Silver (Ag) | 4.3 | 288 | Ultraviolet |
| Copper (Cu) | 4.8 | 258 | Ultraviolet |
See It: The Photoelectric Effect
Numbers in a table are one thing — watching photons actually kick electrons out of real metals is another. In the simulator below, you can pick a metal, change the frequency of incoming light, and change the intensity (how bright the beam is). Two live graphs plot kinetic energy against frequency and against intensity so you can see the relationships Einstein predicted.
Things worth trying:
- Start with Cs and crank the frequency down until electrons stop ejecting. Note the threshold.
- Now swap to Cu at the same frequency — nothing happens. Its work function is too high.
- Hold the frequency above threshold and drag the intensity from 1 → 10. More electrons fly out, but each one still carries the same KE.
- Watch the KE-vs-ν graph: the curve is flat at zero until ν = ν₀, then rises as a perfectly straight line — exactly as Einstein predicted.
Loading simulator…
Where you meet the photoelectric effect every day
- Solar panels — photons from the sun knock electrons loose inside a silicon cell. Those freed electrons become the current that powers your phone charger.
- Digital camera sensors (CCD / CMOS) — each pixel is a tiny photoelectric cell. Photons create electrons; the camera counts them.
- Night-vision goggles — IR photons are amplified into visible-range electrons that illuminate a phosphor screen.
- Photomultiplier tubes — the devices inside medical PET scanners and particle physics detectors, sensitive enough to see a single photon.
- Automatic door sensors and smoke alarms — a beam of light crossing a photocell; break the beam, and the photocurrent drops.
Every time you use any of these, you are personally witnessing Planck and Einstein's quanta at work.

The Dual Nature of Light
There is now a serious problem. Diffraction and interference (the previous page) proved that light is a wave. The photoelectric effect proves that light is a particle. Both sets of experiments are rock-solid. Both conclusions are right.
The resolution, which took decades to accept, is this: light has a dual nature. It propagates through space as a wave — that is how it diffracts, interferes, and travels at . But it interacts with matter as a particle — a photon — that delivers its energy all at once, in a single indivisible chunk.
Which face light shows you depends entirely on the experiment you perform. Pass it through two slits and you see waves. Point it at a metal plate and you see particles. Nature is not being inconsistent — we just don't have a single everyday word that describes something that is both. In the next chapter, you will discover that electrons do exactly the same thing. Wave-particle duality isn't a quirk of light. It's a basic property of reality at the atomic scale.
Worked Examples
Problem
Calculate energy of one mole of photons of radiation whose frequency is Hz.
Problem
A 100 W bulb emits monochromatic light of wavelength 400 nm. Calculate the number of photons emitted per second by the bulb.
Problem
When electromagnetic radiation of wavelength 300 nm falls on the surface of sodium, electrons are emitted with a kinetic energy of J mol. What is the minimum energy needed to remove an electron from sodium? What is the maximum wavelength that will cause a photoelectron to be emitted?
Problem
The threshold frequency for a metal is s. Calculate the kinetic energy of an electron emitted when radiation of frequency s hits the metal.
Q1.Which observation could NOT be explained by the classical wave theory of light?