Timeline of Atomic Models
2,400 years of human curiosity — from Greek philosophy to quantum mechanics
Around 400 BC, a Greek philosopher named Democritus proposed that if you kept cutting matter into smaller and smaller pieces, you'd eventually reach a particle that couldn't be cut any further. He called it atomos — Greek for indivisible.
He had no microscope, no instrument, no experimental data. Just pure reasoning. It took humanity another 2,200 years to prove he was on the right track — and another 100 years after that to realise the atom is actually divisible after all.
Before Science: The Philosophical Atom
Two ancient Greek thinkers had completely opposite ideas about matter:
Democritus (400 BC) argued that matter is made of tiny, discrete, indivisible particles moving through empty space. Different materials exist because their atoms have different shapes and sizes. He was largely right — but he was ignored.
Aristotle (350 BC) disagreed. He believed matter is continuous and made of four fundamental essences: Earth, Water, Air, and Fire. Every substance was a mixture of these four.
Aristotle was wrong — but he was more famous, more influential, and had the backing of the church. His idea dominated Western science for nearly 2,000 years, delaying the development of atomic theory until the 1800s. The lesson: authority can delay truth, but not indefinitely.
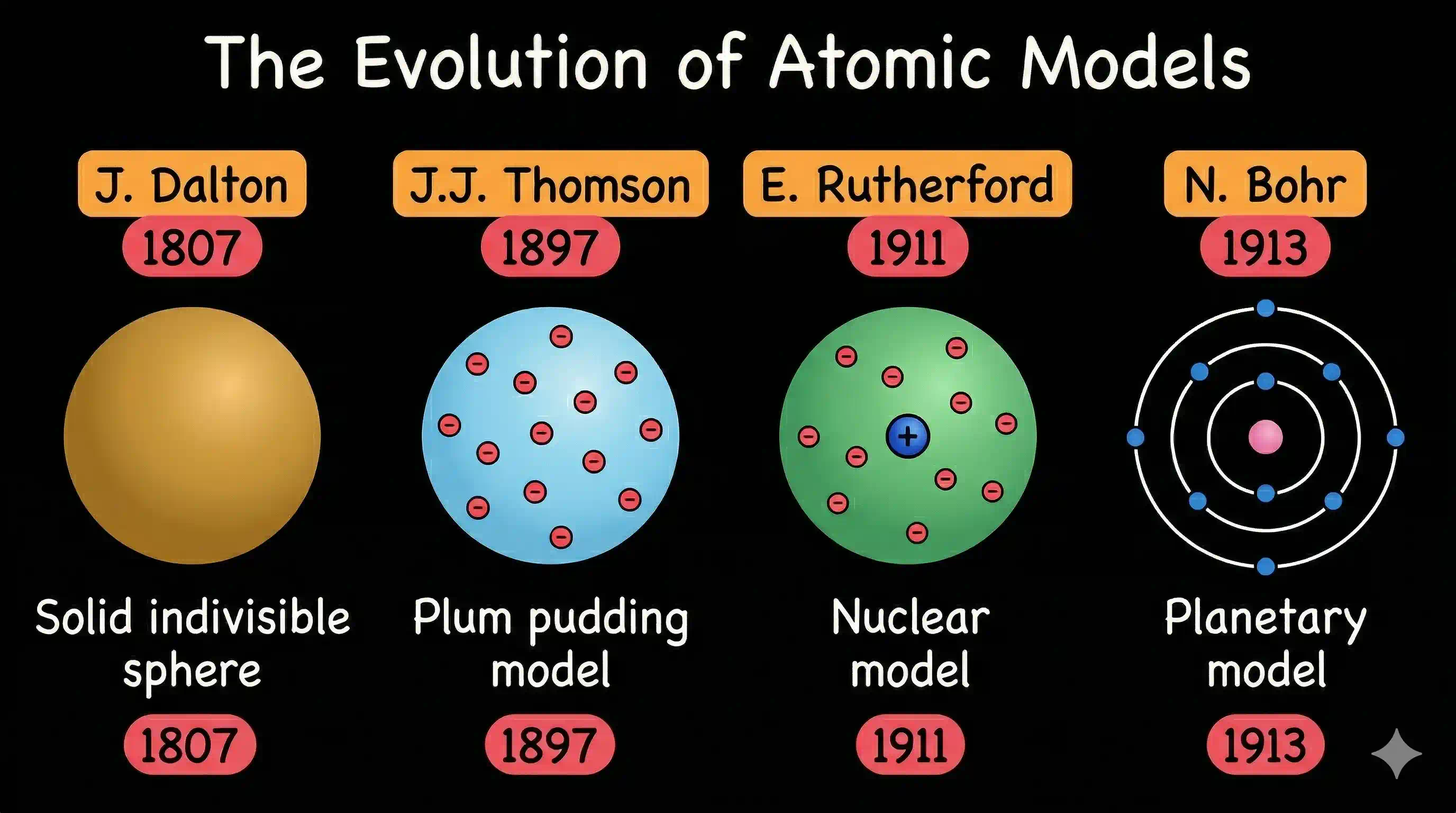
Dalton's Atomic Theory (1807) — When Science Finally Caught Up
English chemist John Dalton was the first to put the atom on a scientific footing — grounding it in experimental laws (conservation of mass, definite proportions, multiple proportions) rather than philosophy.
Dalton's key postulates:
- All matter is made of indivisible, indestructible atoms
- All atoms of a given element are identical in mass and size; atoms of different elements differ
- Compounds form when atoms of different elements combine in simple whole-number ratios
- Chemical reactions rearrange atoms — they don't create or destroy them
What turned out to be wrong: Atoms are divisible — into electrons, protons, and neutrons. And atoms of the same element can have different masses (isotopes). But Dalton's framework was revolutionary and drove chemistry forward for a century.
The reaction is a perfect illustration of postulate 4 — atoms just swap partners.
The Experimental Revolution (1897–1932)
In just 35 years, a burst of brilliant experiments shattered the 'solid indivisible sphere' picture and replaced it with the quantum mechanical model we use today. Each scientist below discovered something that made the previous model insufficient:
AI Generation Prompt
Horizontal scientific timeline diagram spanning 1897 to 1932. A thick horizontal grey line runs across the centre of the image. Eight coloured circular markers sit on the line at their respective years. Alternating above and below the line: Above the line — J.J. Thomson (1897, dark green marker): 'Discovery of electron'; R.K. Millikan (1910, yellow marker): 'Measurement of electronic charge'; Louis de Broglie (1924, purple marker): 'Wave-particle duality'; Werner Heisenberg (1927, dark red marker): 'Uncertainty principle'. Below the line — Ernest Rutherford (1909, red marker): 'Discovery of nucleus'; Niels Bohr (1913, blue marker): 'Bohr atomic model'; Erwin Schrödinger (1926, green marker): 'Schrödinger wave equation'; James Chadwick (1932, orange marker): 'Discovery of neutron'. Each entry has a vertical arrow connecting the marker to the label and year. Dark background, orange accent labels, clean technical illustration style.
Key discoveries in atomic structure (1897–1932)
| Scientist | Year | Contribution |
|---|---|---|
| J.J. Thomson | 1897 | Discovered the electron — proved atoms are divisible |
| Ernest Rutherford | 1909 | Gold foil experiment — discovered the nucleus |
| R.K. Millikan | 1910 | Oil drop experiment — measured the charge of one electron |
| Niels Bohr | 1913 | Proposed quantised electron orbits — explained hydrogen spectrum |
| Louis de Broglie | 1924 | Proposed wave-particle duality of electrons |
| Erwin Schrödinger | 1926 | Wave equation — electrons as probability clouds, not orbits |
| Werner Heisenberg | 1927 | Uncertainty principle — you cannot know position and momentum simultaneously |
| James Chadwick | 1932 | Discovered the neutron — completed the picture of atomic composition |
Q1.Which of Dalton's original postulates was later proved to be incorrect?
Around 400 BC, a Greek philosopher named Democritus proposed that if you kept cutting matter into smaller and smaller pieces, you'd eventually reach a particle that couldn't be cut any further. He called it atomos — Greek for indivisible.
He had no microscope, no instrument, no experimental data. Just pure reasoning. It took humanity another 2,200 years to prove he was on the right track — and another 100 years after that to realise the atom is actually divisible after all.

Before Science: The Philosophical Atom
Two ancient Greek thinkers had completely opposite ideas about matter:
Democritus (400 BC) argued that matter is made of tiny, discrete, indivisible particles moving through empty space. Different materials exist because their atoms have different shapes and sizes. He was largely right — but he was ignored.
Aristotle (350 BC) disagreed. He believed matter is continuous and made of four fundamental essences: Earth, Water, Air, and Fire. Every substance was a mixture of these four.
Aristotle was wrong — but he was more famous, more influential, and had the backing of the church. His idea dominated Western science for nearly 2,000 years, delaying the development of atomic theory until the 1800s. The lesson: authority can delay truth, but not indefinitely.
Dalton's Atomic Theory (1807) — When Science Finally Caught Up
English chemist John Dalton was the first to put the atom on a scientific footing — grounding it in experimental laws (conservation of mass, definite proportions, multiple proportions) rather than philosophy.
Dalton's key postulates:
- All matter is made of indivisible, indestructible atoms
- All atoms of a given element are identical in mass and size; atoms of different elements differ
- Compounds form when atoms of different elements combine in simple whole-number ratios
- Chemical reactions rearrange atoms — they don't create or destroy them
What turned out to be wrong: Atoms are divisible — into electrons, protons, and neutrons. And atoms of the same element can have different masses (isotopes). But Dalton's framework was revolutionary and drove chemistry forward for a century.
The reaction is a perfect illustration of postulate 4 — atoms just swap partners.
The Experimental Revolution (1897–1932)
In just 35 years, a burst of brilliant experiments shattered the 'solid indivisible sphere' picture and replaced it with the quantum mechanical model we use today. Each scientist below discovered something that made the previous model insufficient:
AI Generation Prompt
Horizontal scientific timeline diagram spanning 1897 to 1932. A thick horizontal grey line runs across the centre of the image. Eight coloured circular markers sit on the line at their respective years. Alternating above and below the line: Above the line — J.J. Thomson (1897, dark green marker): 'Discovery of electron'; R.K. Millikan (1910, yellow marker): 'Measurement of electronic charge'; Louis de Broglie (1924, purple marker): 'Wave-particle duality'; Werner Heisenberg (1927, dark red marker): 'Uncertainty principle'. Below the line — Ernest Rutherford (1909, red marker): 'Discovery of nucleus'; Niels Bohr (1913, blue marker): 'Bohr atomic model'; Erwin Schrödinger (1926, green marker): 'Schrödinger wave equation'; James Chadwick (1932, orange marker): 'Discovery of neutron'. Each entry has a vertical arrow connecting the marker to the label and year. Dark background, orange accent labels, clean technical illustration style.
Key discoveries in atomic structure (1897–1932)
| Scientist | Year | Contribution |
|---|---|---|
| J.J. Thomson | 1897 | Discovered the electron — proved atoms are divisible |
| Ernest Rutherford | 1909 | Gold foil experiment — discovered the nucleus |
| R.K. Millikan | 1910 | Oil drop experiment — measured the charge of one electron |
| Niels Bohr | 1913 | Proposed quantised electron orbits — explained hydrogen spectrum |
| Louis de Broglie | 1924 | Proposed wave-particle duality of electrons |
| Erwin Schrödinger | 1926 | Wave equation — electrons as probability clouds, not orbits |
| Werner Heisenberg | 1927 | Uncertainty principle — you cannot know position and momentum simultaneously |
| James Chadwick | 1932 | Discovered the neutron — completed the picture of atomic composition |
Q1.Which of Dalton's original postulates was later proved to be incorrect?