Sub-Atomic Particles
How scientists cracked open the atom and found electrons, protons, and neutrons inside
You're reading this on a screen that works because electrons flow through circuits. The TV, the MRI machine, the X-ray — all of these were made possible by one fundamental discovery: atoms are not solid, indivisible balls. They're made of smaller parts. And the story of how we found those parts is one of the most dramatic detective stories in the history of science.
Dalton's atomic theory (1808) described atoms as indivisible, indestructible solid spheres. It explained the law of conservation of mass and definite proportions beautifully. But by the 1850s, cracks were appearing. When you rub glass with silk, charge builds up. Pass electricity through a gas and strange glowing rays appear. Something was clearly inside the atom.
Three particles were discovered in quick succession:
- Electron — 1897 (J.J. Thomson)
- Proton — 1919 (Rutherford)
- Neutron — 1932 (Chadwick)
Faraday's Clue: Electricity and Atoms Are Connected
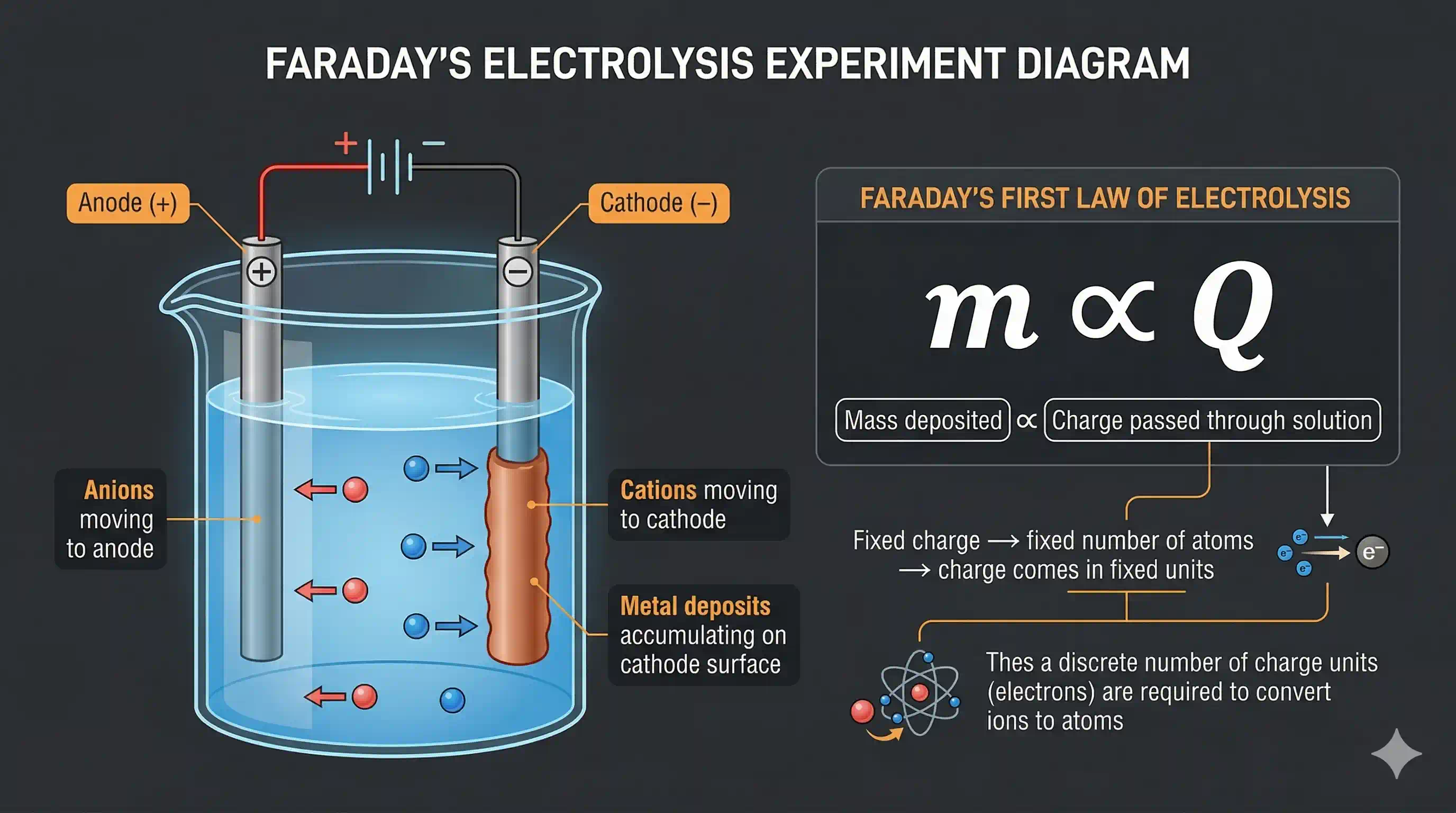
Around 1830–1860, Michael Faraday was studying what happens when electricity passes through solutions — a process called electrolysis. His experiments were startling:
- When he passed electricity through a copper sulphate solution, copper deposited exactly on the negative electrode.
- The amount deposited was directly proportional to the amount of electricity passed.
- Different substances needed different amounts of electricity per gram — but always in neat whole-number ratios.
Faraday's laws of electrolysis suggested that electricity itself comes in discrete chunks — and that these chunks are linked to atoms. If you need a fixed amount of charge to deposit one atom of copper, then maybe charge and atoms are connected at a fundamental level.
Faraday didn't have an explanation. But he planted the seed: electricity might be particle-like, not a continuous fluid. It took another 40 years — and a glass tube — to find the particle.
Inside the Discharge Tube: A Controlled Lightning

You've seen lightning. The atmosphere is full of gas molecules — nitrogen, oxygen, argon. Normally they don't conduct electricity. But when the voltage difference between a cloud and the ground becomes enormous (millions of volts), something dramatic happens: the electric field rips electrons off the gas molecules, turning them into ions. These ions and free electrons rush through the air, colliding and glowing — that's the lightning bolt.
Scientists in the 1850s realised they could create a miniature, controllable version of this in a laboratory. The key ingredients:
- A sealed glass tube with metal electrodes at both ends
- Very low pressure inside (pumped out with a vacuum pump — around 0.001 mmHg or less)
- High voltage across the electrodes (several thousand volts)
At atmospheric pressure, the gas is too dense — molecules collide constantly and block charge flow. But as you pump the gas out:
- The remaining molecules become far apart
- Free electrons can accelerate over long distances without hitting anything
- They gain enough energy to ionise the gas molecules they do hit, creating an avalanche of charge
The result? A glowing beam shoots from the cathode (negative electrode) toward the anode (positive electrode). These beams were called cathode rays.
Cathode Rays vs Canal Rays — A Critical Distinction
When scientists drilled a hole in the cathode, they discovered something moving in the opposite direction — from anode toward cathode:
| Property | Cathode Rays | Canal Rays (Anode Rays) |
|---|---|---|
| Charge | Negative | Positive |
| Direction | Cathode → Anode | Anode → Cathode |
| Charge-to-mass ratio | Constant (same always) | Varies (depends on gas) |
| Identity | Electrons (universal) | Positive ions (gas-specific) |
| Discovered by | Crookes / Thomson | Goldstein (1886) |
This table is the most important thing on this page for exams. The fact that cathode rays have a constant e/m ratio regardless of cathode material or gas proved that electrons are a universal constituent of all matter — the same particle in every atom.
Loading simulator…
Thomson's e/m Ratio
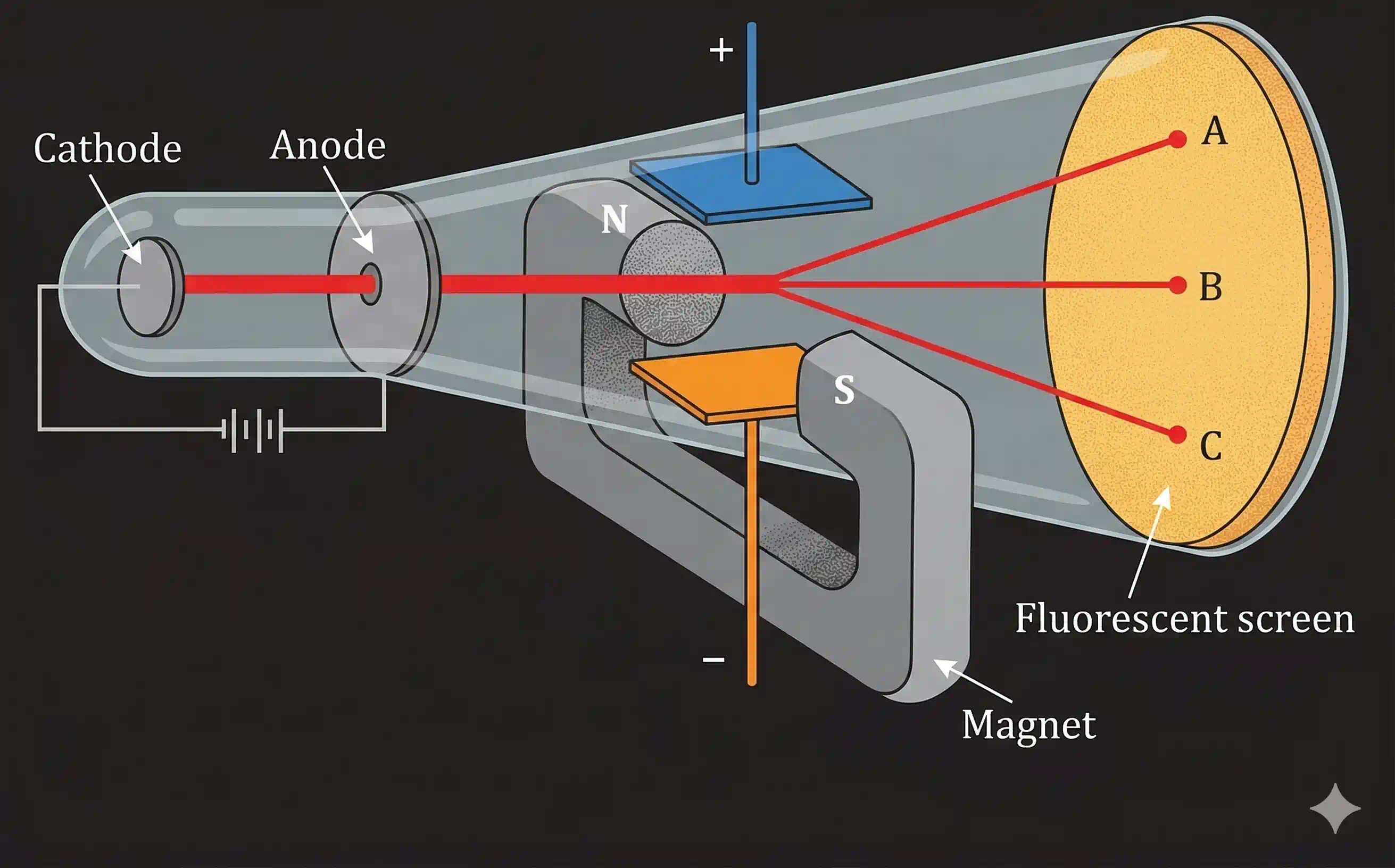
The deviation of cathode rays from a straight line depends on the strength of the applied electric and magnetic forces. When only electric field is applied, the electrons deviate from their path and hit the cathode ray tube at point A. Similarly when only magnetic field is applied, electron strikes the cathode ray tube at point C. By carefully balancing the electrical and magnetic field strength, it is possible to bring back the electron to the path followed as in the absence of electric or magnetic field and they hit the screen at point B.
In 1897, J.J. Thomson measured the charge-to-mass ratio of the electron by balancing electric and magnetic fields acting on the cathode rays.
Method: Apply an electric field (deflects beam one way) and a magnetic field (deflects beam the other way). When the beam travels straight, the two forces balance:
Knowing , Thomson then used just the magnetic field to measure the radius of curvature, giving:
This value was identical regardless of the cathode material — confirming electrons are universal.
But Thomson only knew the ratio. He couldn't determine charge and mass individually. That required Millikan.
Millikan's Oil Drop Experiment — Charge on Electron
Thomson's discovery in 1897 had left everyone stunned — and a little confused. He had shown that cathode rays were made of particles far smaller than any atom. How much smaller? His measurements of the ratio suggested these particles weighed less than of a hydrogen atom — the lightest atom known. Fellow scientists were openly sceptical. Some thought he was joking. Nobody had seriously entertained the idea that atoms — supposedly the smallest things in existence — could have even smaller pieces inside them.
But Thomson's measurement had a gap. The ratio told you how the charge and mass related to each other, but not what either of them actually was. The charge on a single electron was still completely unknown. Without that, you couldn't calculate the electron's mass either.
That's where Robert Millikan came in. An American physicist working at the University of Chicago, Millikan spent nearly a decade — from 1906 to 1914 — designing a clever experiment to pin down the exact charge on one electron. He knew that if he could measure the charge, Thomson's ratio would immediately give him the mass too.
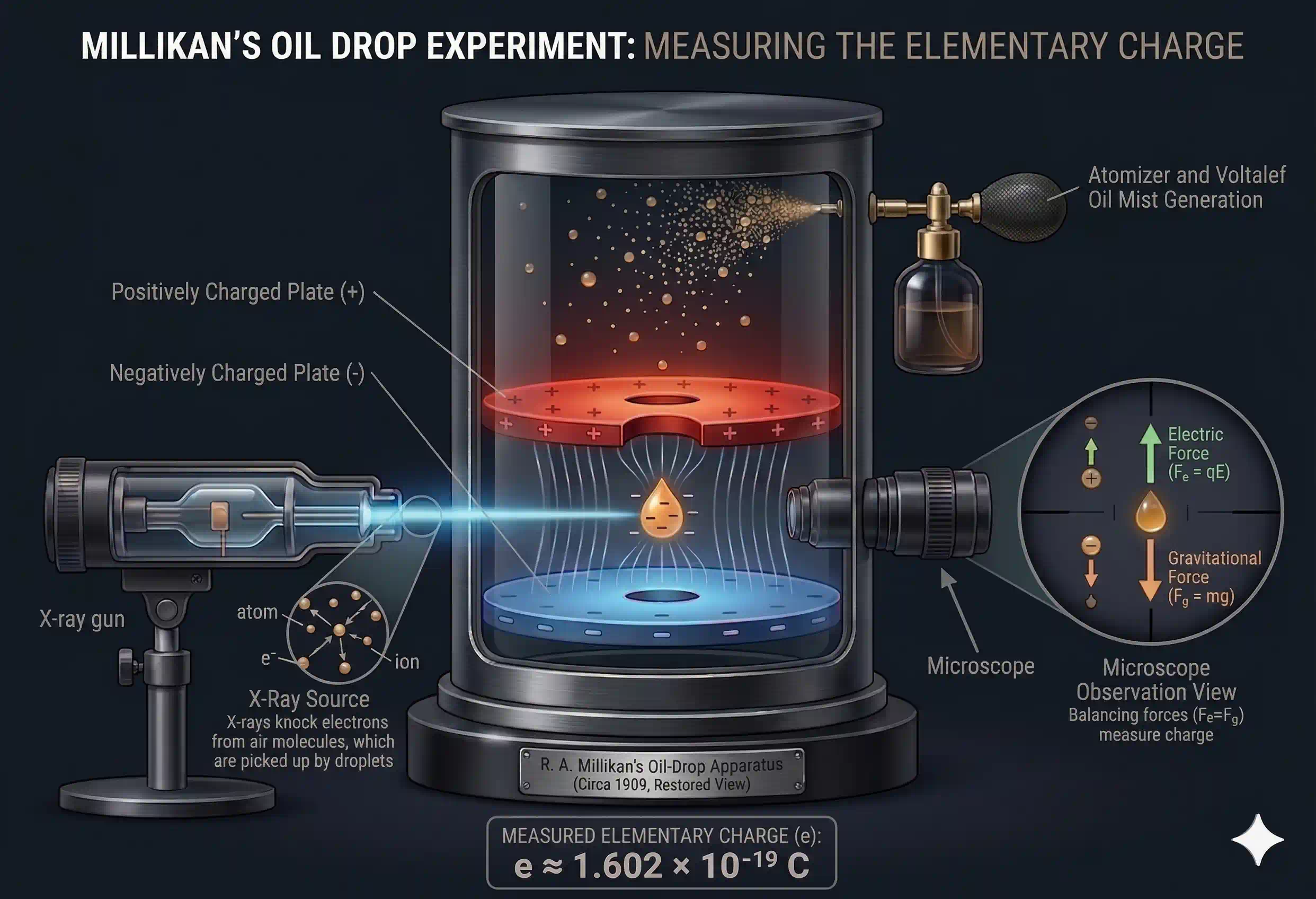
How it worked:
- Fine oil droplets were sprayed into a chamber between two charged plates
- X-rays ionised the air, giving some droplets a negative charge by knocking electrons off nearby gas molecules — those electrons stuck to the falling oil droplet
- With the electric field switched off, Millikan measured the droplet's mass from how fast it fell under gravity
- He then switched the field on and adjusted its strength until the charged droplet hovered motionless — that meant the upward electric force exactly balanced the downward pull of gravity
- From the field strength needed to suspend the droplet, he could calculate the total charge it was carrying
After doing this hundreds of times with droplets carrying different numbers of electrons, Millikan noticed something that made everything click: the charge on any droplet was always a whole-number multiple of the same tiny value. It didn't matter how many electrons were stuck to the drop — one electron, three, seven — the total charge was always , , . There were no fractional values. That smallest possible unit had to be the charge of a single electron.
The value Millikan calculated for that fundamental unit:
Remarkably, his result — worked out over a century ago — is within 1% of the value we accept today. Once the charge was known, the mass followed directly by plugging into Thomson's ratio:
Discovery of Protons and Neutrons
Proton: Remember canal rays? When different gases were used in the discharge tube, the positive ions had different e/m ratios. The lightest positive ion — with the highest e/m — came from hydrogen gas. This was named the proton (characterised by Rutherford in 1919).
- Charge: C (equal and opposite to electron)
- Mass: kg = 1836 times heavier than electron
Neutron: In 1932, James Chadwick bombarded a thin sheet of beryllium with -particles:
Electrically neutral particles with mass slightly greater than a proton were emitted — neutrons.
| Particle | Symbol | Charge | Mass (kg) | Location in Atom |
|---|---|---|---|---|
| Electron | C | Outside nucleus | ||
| Proton | C | Nucleus | ||
| Neutron | 0 | Nucleus |
Q1.The charge-to-mass ratio of cathode rays is constant regardless of the cathode material and gas used. This proves that:
You're reading this on a screen that works because electrons flow through circuits. The TV, the MRI machine, the X-ray — all of these were made possible by one fundamental discovery: atoms are not solid, indivisible balls. They're made of smaller parts. And the story of how we found those parts is one of the most dramatic detective stories in the history of science.
Dalton's atomic theory (1808) described atoms as indivisible, indestructible solid spheres. It explained the law of conservation of mass and definite proportions beautifully. But by the 1850s, cracks were appearing. When you rub glass with silk, charge builds up. Pass electricity through a gas and strange glowing rays appear. Something was clearly inside the atom.
Three particles were discovered in quick succession:
- Electron — 1897 (J.J. Thomson)
- Proton — 1919 (Rutherford)
- Neutron — 1932 (Chadwick)
Faraday's Clue: Electricity and Atoms Are Connected
Around 1830–1860, Michael Faraday was studying what happens when electricity passes through solutions — a process called electrolysis. His experiments were startling:
- When he passed electricity through a copper sulphate solution, copper deposited exactly on the negative electrode.
- The amount deposited was directly proportional to the amount of electricity passed.
- Different substances needed different amounts of electricity per gram — but always in neat whole-number ratios.
Faraday's laws of electrolysis suggested that electricity itself comes in discrete chunks — and that these chunks are linked to atoms. If you need a fixed amount of charge to deposit one atom of copper, then maybe charge and atoms are connected at a fundamental level.
Faraday didn't have an explanation. But he planted the seed: electricity might be particle-like, not a continuous fluid. It took another 40 years — and a glass tube — to find the particle.

Inside the Discharge Tube: A Controlled Lightning

You've seen lightning. The atmosphere is full of gas molecules — nitrogen, oxygen, argon. Normally they don't conduct electricity. But when the voltage difference between a cloud and the ground becomes enormous (millions of volts), something dramatic happens: the electric field rips electrons off the gas molecules, turning them into ions. These ions and free electrons rush through the air, colliding and glowing — that's the lightning bolt.
Scientists in the 1850s realised they could create a miniature, controllable version of this in a laboratory. The key ingredients:
- A sealed glass tube with metal electrodes at both ends
- Very low pressure inside (pumped out with a vacuum pump — around 0.001 mmHg or less)
- High voltage across the electrodes (several thousand volts)
At atmospheric pressure, the gas is too dense — molecules collide constantly and block charge flow. But as you pump the gas out:
- The remaining molecules become far apart
- Free electrons can accelerate over long distances without hitting anything
- They gain enough energy to ionise the gas molecules they do hit, creating an avalanche of charge
The result? A glowing beam shoots from the cathode (negative electrode) toward the anode (positive electrode). These beams were called cathode rays.
Cathode Rays vs Canal Rays — A Critical Distinction
When scientists drilled a hole in the cathode, they discovered something moving in the opposite direction — from anode toward cathode:
| Property | Cathode Rays | Canal Rays (Anode Rays) |
|---|---|---|
| Charge | Negative | Positive |
| Direction | Cathode → Anode | Anode → Cathode |
| Charge-to-mass ratio | Constant (same always) | Varies (depends on gas) |
| Identity | Electrons (universal) | Positive ions (gas-specific) |
| Discovered by | Crookes / Thomson | Goldstein (1886) |
This table is the most important thing on this page for exams. The fact that cathode rays have a constant e/m ratio regardless of cathode material or gas proved that electrons are a universal constituent of all matter — the same particle in every atom.
Loading simulator…
Thomson's e/m Ratio

The deviation of cathode rays from a straight line depends on the strength of the applied electric and magnetic forces. When only electric field is applied, the electrons deviate from their path and hit the cathode ray tube at point A. Similarly when only magnetic field is applied, electron strikes the cathode ray tube at point C. By carefully balancing the electrical and magnetic field strength, it is possible to bring back the electron to the path followed as in the absence of electric or magnetic field and they hit the screen at point B.
In 1897, J.J. Thomson measured the charge-to-mass ratio of the electron by balancing electric and magnetic fields acting on the cathode rays.
Method: Apply an electric field (deflects beam one way) and a magnetic field (deflects beam the other way). When the beam travels straight, the two forces balance:
Knowing , Thomson then used just the magnetic field to measure the radius of curvature, giving:
This value was identical regardless of the cathode material — confirming electrons are universal.
But Thomson only knew the ratio. He couldn't determine charge and mass individually. That required Millikan.
Millikan's Oil Drop Experiment — Charge on Electron
Thomson's discovery in 1897 had left everyone stunned — and a little confused. He had shown that cathode rays were made of particles far smaller than any atom. How much smaller? His measurements of the ratio suggested these particles weighed less than of a hydrogen atom — the lightest atom known. Fellow scientists were openly sceptical. Some thought he was joking. Nobody had seriously entertained the idea that atoms — supposedly the smallest things in existence — could have even smaller pieces inside them.
But Thomson's measurement had a gap. The ratio told you how the charge and mass related to each other, but not what either of them actually was. The charge on a single electron was still completely unknown. Without that, you couldn't calculate the electron's mass either.
That's where Robert Millikan came in. An American physicist working at the University of Chicago, Millikan spent nearly a decade — from 1906 to 1914 — designing a clever experiment to pin down the exact charge on one electron. He knew that if he could measure the charge, Thomson's ratio would immediately give him the mass too.
How it worked:
- Fine oil droplets were sprayed into a chamber between two charged plates
- X-rays ionised the air, giving some droplets a negative charge by knocking electrons off nearby gas molecules — those electrons stuck to the falling oil droplet
- With the electric field switched off, Millikan measured the droplet's mass from how fast it fell under gravity
- He then switched the field on and adjusted its strength until the charged droplet hovered motionless — that meant the upward electric force exactly balanced the downward pull of gravity
- From the field strength needed to suspend the droplet, he could calculate the total charge it was carrying
After doing this hundreds of times with droplets carrying different numbers of electrons, Millikan noticed something that made everything click: the charge on any droplet was always a whole-number multiple of the same tiny value. It didn't matter how many electrons were stuck to the drop — one electron, three, seven — the total charge was always , , . There were no fractional values. That smallest possible unit had to be the charge of a single electron.
The value Millikan calculated for that fundamental unit:
Remarkably, his result — worked out over a century ago — is within 1% of the value we accept today. Once the charge was known, the mass followed directly by plugging into Thomson's ratio:

Discovery of Protons and Neutrons
Proton: Remember canal rays? When different gases were used in the discharge tube, the positive ions had different e/m ratios. The lightest positive ion — with the highest e/m — came from hydrogen gas. This was named the proton (characterised by Rutherford in 1919).
- Charge: C (equal and opposite to electron)
- Mass: kg = 1836 times heavier than electron
Neutron: In 1932, James Chadwick bombarded a thin sheet of beryllium with -particles:
Electrically neutral particles with mass slightly greater than a proton were emitted — neutrons.
| Particle | Symbol | Charge | Mass (kg) | Location in Atom |
|---|---|---|---|---|
| Electron | C | Outside nucleus | ||
| Proton | C | Nucleus | ||
| Neutron | 0 | Nucleus |
Q1.The charge-to-mass ratio of cathode rays is constant regardless of the cathode material and gas used. This proves that: