Thomson's Model of the Atom
The first picture of the atom — elegant, simple, and completely wrong
Imagine a British dessert: a soft, fluffy plum pudding with raisins embedded throughout. That was the first serious picture scientists had of the atom — and it came from the man who discovered the electron itself. For almost fifteen years, it was the best answer humanity had to the question: what does an atom actually look like inside?
The Puzzle Before Thomson
Most scientists of the late nineteenth century accepted that all matter was built from atoms — but beyond that, they knew almost nothing about what atoms actually were. Were they tiny billiard balls? Clouds? Gears? Nobody could say.
Then came a huge clue. In 1897, J. J. Thomson — working at the Cavendish Laboratory in Cambridge — discovered through his discharge tube experiments that every atom contains negatively charged particles, which were eventually called electrons. These electrons carried a negative charge. But here was the catch: atoms themselves are electrically neutral. You can touch an iron bar without getting a shock.
So if atoms contain negative electrons but have no overall charge, the conclusion was inescapable: there must be positive charge hiding somewhere inside each atom, exactly balancing the electrons.
This raised two urgent questions:
- What is this positive matter?
- How is it arranged — and where do the electrons sit inside it?
Thomson's Answer: The Plum Pudding Model (1898)
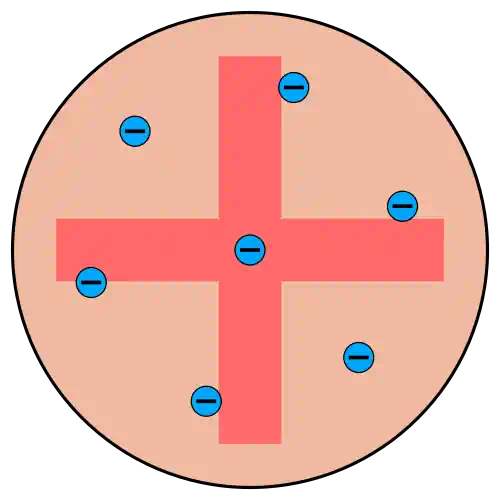
One year after discovering the electron, in 1898, Thomson proposed a simple and visually intuitive answer.
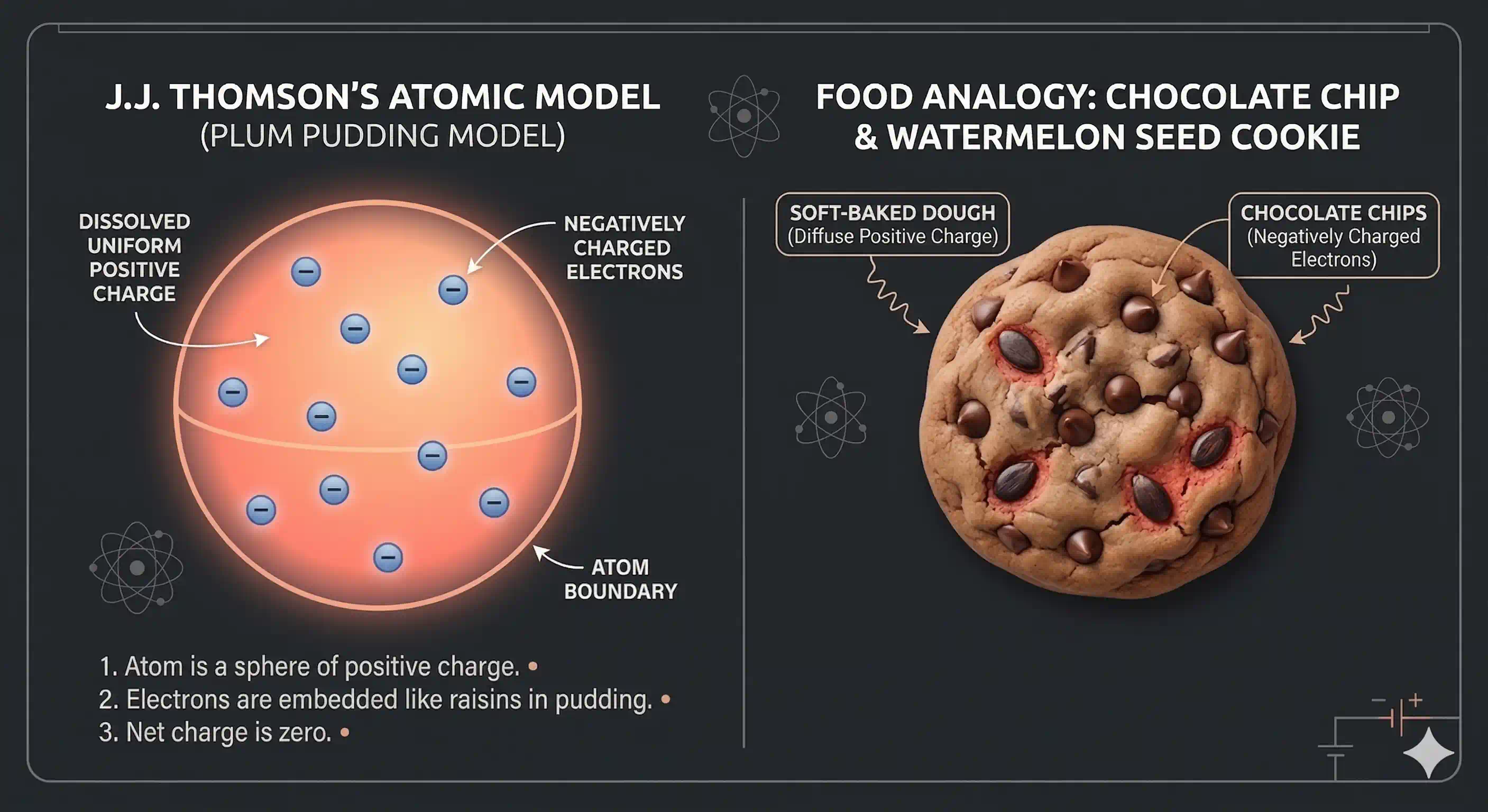
He pictured the atom as a sphere of uniformly distributed positive charge — roughly m across — with the tiny negative electrons embedded inside it, like raisins pressed into a ball of dough.
The electrons were arranged in the positions that made the whole system most stable electrostatically — each one balanced against the push of its neighbours and the pull of the surrounding positive 'pudding'.
Different textbooks use different food analogies for exactly the same model:
- Plum pudding (British dessert, the original name)
- Raisin pudding
- Watermelon — red flesh = positive charge, black seeds = electrons
What the Model Got Right
For 1898, this was a genuinely clever proposal. It explained several facts about atoms that had puzzled scientists for decades:
- Electrical neutrality. The total positive charge of the 'pudding' exactly cancels the total negative charge of all the electrons, so the atom as a whole is neutral — just as observed.
- Mass distribution. Thomson assumed the atom's mass was spread uniformly through the sphere. This made sense with what was known at the time — nobody had yet detected a concentrated core.
- Stability. The electrons, pushed by their mutual repulsion and pulled by the positive background, could settle into a stable geometric pattern — explaining why atoms don't spontaneously fly apart.
- Emission of electrons. Since electrons were stuck inside the atom like raisins, it was easy to see why energetic processes (heating, electric discharge) could knock them out — producing cathode rays, photoelectrons, and β-rays.
Thomson's work was so influential that he was awarded the Nobel Prize in Physics in 1906 for his theoretical and experimental investigations on the conduction of electricity by gases — the very work that led to his model.
Why the Model Failed
Elegant as it was, the plum pudding model did not survive its first serious test. Within just 13 years, experiments destroyed it completely. Here is exactly where it went wrong:
1. It could not explain Rutherford's α-particle scattering experiment (1909–1911).
If the positive charge and mass were spread uniformly throughout the atom, then fast-moving α-particles fired at a thin gold foil should have passed straight through with at most tiny deflections. But Rutherford's team saw something shocking: a small fraction of α-particles bounced straight back, as if they had hit something solid and concentrated. In Rutherford's words, it was 'as if you had fired a 15-inch shell at a piece of tissue paper and it came back and hit you.' A uniform pudding could never produce that — there had to be a dense, concentrated nucleus.
2. It could not explain atomic spectra.
When atoms are heated or excited, they give off light at very specific wavelengths — sharp, discrete lines (the hydrogen spectrum, for example). Thomson's model predicted that electrons vibrating inside the positive cloud should emit a smooth, continuous range of frequencies. The observed line spectra made no sense in his picture at all.
3. It gave no mechanism for the positive charge.
Thomson simply assumed the positive charge was a featureless cloud. He had no particles, no structure, no explanation. Once the proton was discovered (Rutherford, 1919) and later the neutron (Chadwick, 1932), it became clear that positive charge lives in discrete particles inside a tiny nucleus — not smeared across the whole atom.
4. It could not explain the stability of atoms quantitatively.
Even within its own assumptions, the equations didn't quite close. Electrons sitting inside a positive cloud would either radiate away energy and spiral in, or arrange themselves in ways inconsistent with how real elements actually behave.
5. It said nothing about isotopes or atomic number.
Why do hydrogen, helium, and carbon behave completely differently? Thomson's model had no way to distinguish them beyond number of electrons. It couldn't predict why the periodic table has the shape it does, or why isotopes of the same element exist.
Thomson's Legacy
Thomson's model was wrong, but it was wrong in the most productive possible way. By making a clear, testable prediction (α-particles should pass through easily), it invited the experiment that disproved it. Science moves forward when a model is specific enough to fail.
Q1.In Thomson's plum pudding model, how is the positive charge distributed inside the atom?
Imagine a British dessert: a soft, fluffy plum pudding with raisins embedded throughout. That was the first serious picture scientists had of the atom — and it came from the man who discovered the electron itself. For almost fifteen years, it was the best answer humanity had to the question: what does an atom actually look like inside?
The Puzzle Before Thomson
Most scientists of the late nineteenth century accepted that all matter was built from atoms — but beyond that, they knew almost nothing about what atoms actually were. Were they tiny billiard balls? Clouds? Gears? Nobody could say.
Then came a huge clue. In 1897, J. J. Thomson — working at the Cavendish Laboratory in Cambridge — discovered through his discharge tube experiments that every atom contains negatively charged particles, which were eventually called electrons. These electrons carried a negative charge. But here was the catch: atoms themselves are electrically neutral. You can touch an iron bar without getting a shock.
So if atoms contain negative electrons but have no overall charge, the conclusion was inescapable: there must be positive charge hiding somewhere inside each atom, exactly balancing the electrons.
This raised two urgent questions:
- What is this positive matter?
- How is it arranged — and where do the electrons sit inside it?
Thomson's Answer: The Plum Pudding Model (1898)

One year after discovering the electron, in 1898, Thomson proposed a simple and visually intuitive answer.
He pictured the atom as a sphere of uniformly distributed positive charge — roughly m across — with the tiny negative electrons embedded inside it, like raisins pressed into a ball of dough.
The electrons were arranged in the positions that made the whole system most stable electrostatically — each one balanced against the push of its neighbours and the pull of the surrounding positive 'pudding'.
Different textbooks use different food analogies for exactly the same model:
- Plum pudding (British dessert, the original name)
- Raisin pudding
- Watermelon — red flesh = positive charge, black seeds = electrons

What the Model Got Right
For 1898, this was a genuinely clever proposal. It explained several facts about atoms that had puzzled scientists for decades:
- Electrical neutrality. The total positive charge of the 'pudding' exactly cancels the total negative charge of all the electrons, so the atom as a whole is neutral — just as observed.
- Mass distribution. Thomson assumed the atom's mass was spread uniformly through the sphere. This made sense with what was known at the time — nobody had yet detected a concentrated core.
- Stability. The electrons, pushed by their mutual repulsion and pulled by the positive background, could settle into a stable geometric pattern — explaining why atoms don't spontaneously fly apart.
- Emission of electrons. Since electrons were stuck inside the atom like raisins, it was easy to see why energetic processes (heating, electric discharge) could knock them out — producing cathode rays, photoelectrons, and β-rays.
Thomson's work was so influential that he was awarded the Nobel Prize in Physics in 1906 for his theoretical and experimental investigations on the conduction of electricity by gases — the very work that led to his model.
Why the Model Failed
Elegant as it was, the plum pudding model did not survive its first serious test. Within just 13 years, experiments destroyed it completely. Here is exactly where it went wrong:
1. It could not explain Rutherford's α-particle scattering experiment (1909–1911).
If the positive charge and mass were spread uniformly throughout the atom, then fast-moving α-particles fired at a thin gold foil should have passed straight through with at most tiny deflections. But Rutherford's team saw something shocking: a small fraction of α-particles bounced straight back, as if they had hit something solid and concentrated. In Rutherford's words, it was 'as if you had fired a 15-inch shell at a piece of tissue paper and it came back and hit you.' A uniform pudding could never produce that — there had to be a dense, concentrated nucleus.
2. It could not explain atomic spectra.
When atoms are heated or excited, they give off light at very specific wavelengths — sharp, discrete lines (the hydrogen spectrum, for example). Thomson's model predicted that electrons vibrating inside the positive cloud should emit a smooth, continuous range of frequencies. The observed line spectra made no sense in his picture at all.
3. It gave no mechanism for the positive charge.
Thomson simply assumed the positive charge was a featureless cloud. He had no particles, no structure, no explanation. Once the proton was discovered (Rutherford, 1919) and later the neutron (Chadwick, 1932), it became clear that positive charge lives in discrete particles inside a tiny nucleus — not smeared across the whole atom.
4. It could not explain the stability of atoms quantitatively.
Even within its own assumptions, the equations didn't quite close. Electrons sitting inside a positive cloud would either radiate away energy and spiral in, or arrange themselves in ways inconsistent with how real elements actually behave.
5. It said nothing about isotopes or atomic number.
Why do hydrogen, helium, and carbon behave completely differently? Thomson's model had no way to distinguish them beyond number of electrons. It couldn't predict why the periodic table has the shape it does, or why isotopes of the same element exist.
Q1.In Thomson's plum pudding model, how is the positive charge distributed inside the atom?