Why Study the Structure of the Atom?
Same protons, neutrons, electrons — yet wildly different behaviour. The answer is always in the arrangement.

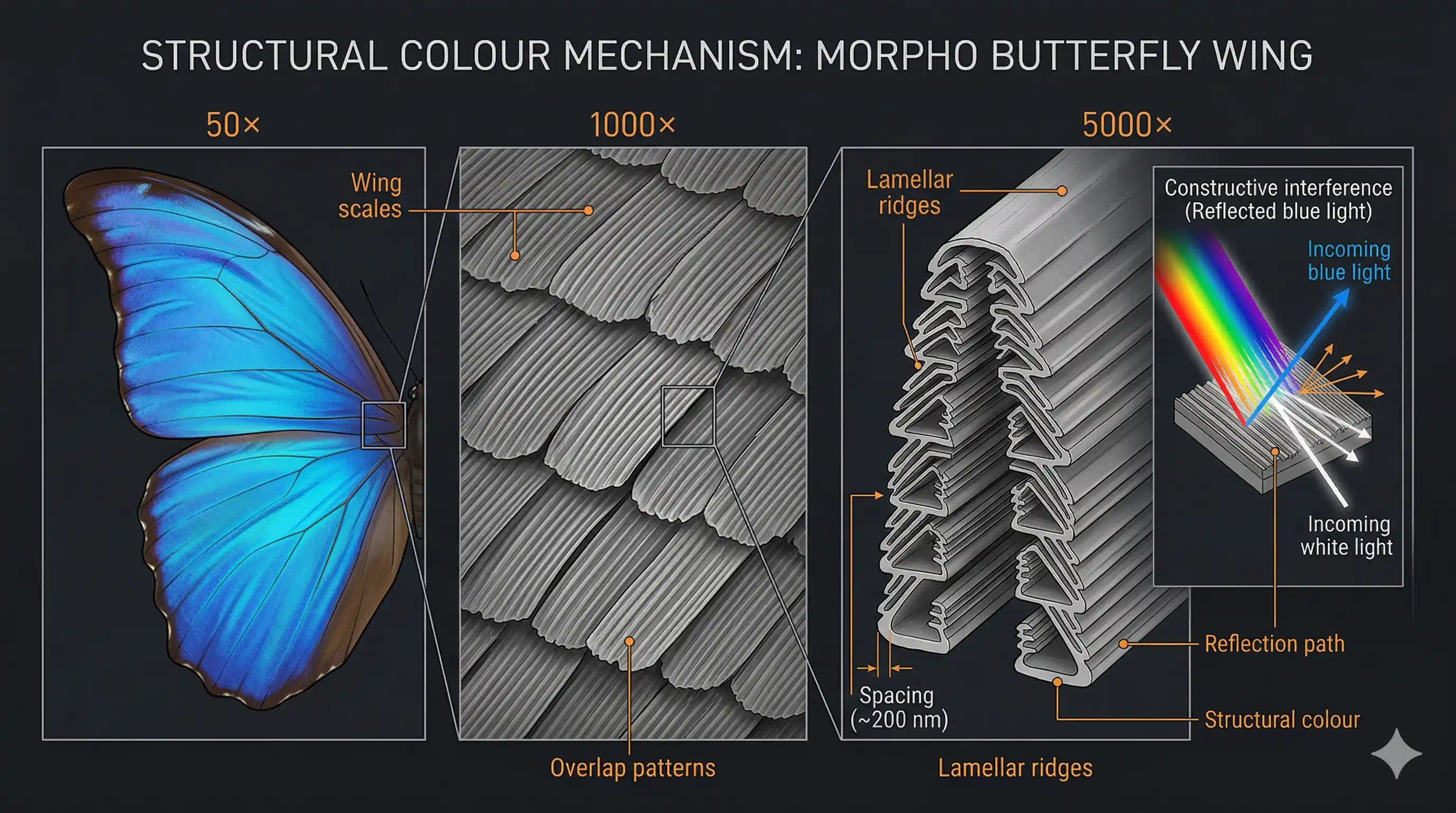
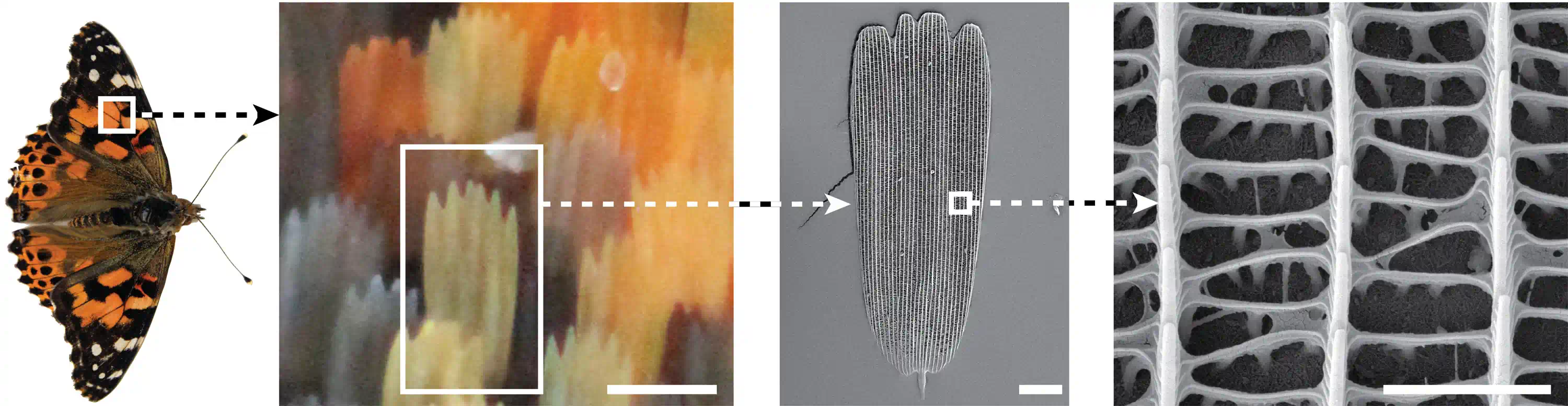
The Morpho butterfly has wings that shift from deep blue to violet to almost black — depending on the angle you look from. No dye, no pigment change, nothing chemical. The colour comes entirely from the structure of the wing: microscopic ridges just 200 nm apart that scatter light like a diffraction grating.
The lesson: to understand a property, you must understand the structure that produces it. The same idea drives all of atomic chemistry.
Consider three metals — sodium (), iron (), and gold (). Drop each one in water and watch what happens:
- Sodium reacts so violently it catches fire and skips across the surface, producing hydrogen gas
- Iron sits quietly but corrodes into rust over days or weeks in the presence of moisture
- Gold doesn't react at all — not with water, not with most acids, not with anything you'll find in everyday life
Here's the puzzle: every single atom of sodium, iron, and gold is built from the same three fundamental particles — protons, neutrons, and electrons. Nothing else. If all matter is made from these three identical building blocks, why do sodium and gold behave so differently?
The answer is not what the particles are — the answer is how they are arranged inside the atom.
The Same Logic — From Butterfly Wings to Atoms
A butterfly's wing looks like it's painted blue. But if you zoom in 1000× under an electron microscope, there's no blue pigment — only a forest of microscopic ridges and scales in a precise repeating pattern.
Colour in butterfly wings comes from two completely different sources:
- Ordinary (pigment) colours: A chemical absorbs certain wavelengths and reflects others. The chemical's composition determines the colour.
- Structural colours: No pigment at all. The structure of the wing — nanoscale ridges spaced exactly right — causes light waves to interfere with each other. The colour you see depends on the viewing angle, not the chemistry. This is called iridescence.
To explain why the Morpho butterfly appears blue from one angle and black from another, you cannot study the chemistry of the wing. You must study the structure of the wing.
Atoms work exactly the same way. To explain why sodium is reactive and gold is not, you cannot stop at 'both have protons and electrons.' You must go deeper — into how those electrons are arranged inside the atom.
Chemistry Is Everywhere Around You
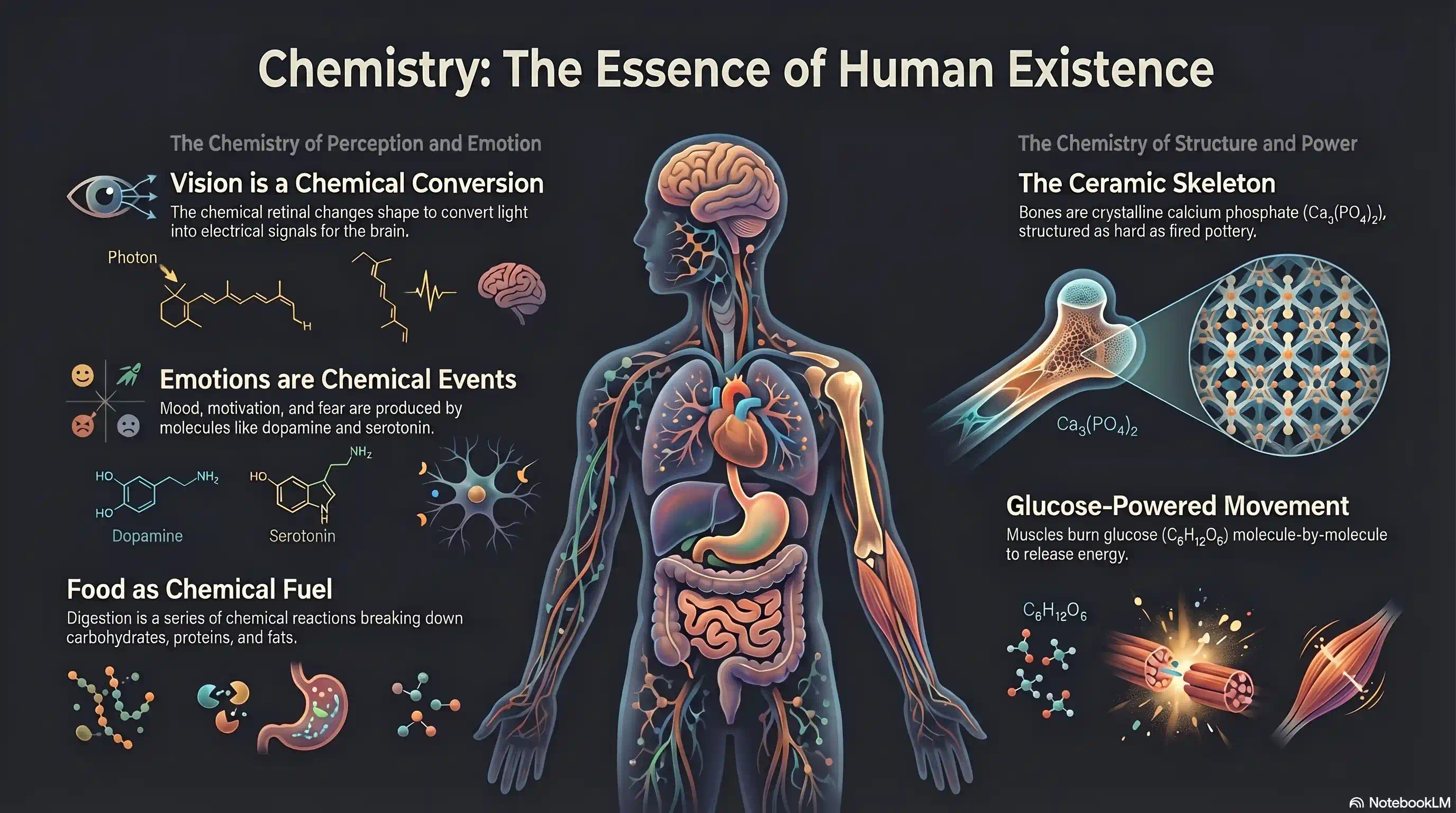
Before we dive into the atom's internal structure, here's one more reason to care. Chemistry isn't just a subject — it's what you are:
- What you see — Your eyes are biological photodetectors. The retina converts photons into electrical signals using a chemical called retinal, which changes shape when it absorbs light.
- What you feel — Every emotion is a chemical event. Dopamine, serotonin, adrenaline — mood, motivation, and fear are all produced by molecules in your brain.
- What you eat — Food is a mixture of chemicals: carbohydrates, proteins, fats, vitamins. Digestion is a series of chemical reactions.
- What supports you — Your skeleton is a ceramic: calcium phosphate () arranged in a crystalline structure, as hard as fired pottery.
- How you move — Your muscles run on glucose (), burning it in a controlled chemical reaction that releases energy molecule by molecule.
Chemistry is the study of matter and its transformations. And since everything is matter, chemistry is the study of everything.
Q1.Sodium, iron, and gold all contain protons, neutrons, and electrons. Why do they have such different chemical reactivities?

The Morpho butterfly has wings that shift from deep blue to violet to almost black — depending on the angle you look from. No dye, no pigment change, nothing chemical. The colour comes entirely from the structure of the wing: microscopic ridges just 200 nm apart that scatter light like a diffraction grating.
The lesson: to understand a property, you must understand the structure that produces it. The same idea drives all of atomic chemistry.
Consider three metals — sodium (), iron (), and gold (). Drop each one in water and watch what happens:
- Sodium reacts so violently it catches fire and skips across the surface, producing hydrogen gas
- Iron sits quietly but corrodes into rust over days or weeks in the presence of moisture
- Gold doesn't react at all — not with water, not with most acids, not with anything you'll find in everyday life
Here's the puzzle: every single atom of sodium, iron, and gold is built from the same three fundamental particles — protons, neutrons, and electrons. Nothing else. If all matter is made from these three identical building blocks, why do sodium and gold behave so differently?
The answer is not what the particles are — the answer is how they are arranged inside the atom.
The Same Logic — From Butterfly Wings to Atoms
A butterfly's wing looks like it's painted blue. But if you zoom in 1000× under an electron microscope, there's no blue pigment — only a forest of microscopic ridges and scales in a precise repeating pattern.
Colour in butterfly wings comes from two completely different sources:
- Ordinary (pigment) colours: A chemical absorbs certain wavelengths and reflects others. The chemical's composition determines the colour.
- Structural colours: No pigment at all. The structure of the wing — nanoscale ridges spaced exactly right — causes light waves to interfere with each other. The colour you see depends on the viewing angle, not the chemistry. This is called iridescence.
To explain why the Morpho butterfly appears blue from one angle and black from another, you cannot study the chemistry of the wing. You must study the structure of the wing.
Atoms work exactly the same way. To explain why sodium is reactive and gold is not, you cannot stop at 'both have protons and electrons.' You must go deeper — into how those electrons are arranged inside the atom.


Chemistry Is Everywhere Around You
Before we dive into the atom's internal structure, here's one more reason to care. Chemistry isn't just a subject — it's what you are:
- What you see — Your eyes are biological photodetectors. The retina converts photons into electrical signals using a chemical called retinal, which changes shape when it absorbs light.
- What you feel — Every emotion is a chemical event. Dopamine, serotonin, adrenaline — mood, motivation, and fear are all produced by molecules in your brain.
- What you eat — Food is a mixture of chemicals: carbohydrates, proteins, fats, vitamins. Digestion is a series of chemical reactions.
- What supports you — Your skeleton is a ceramic: calcium phosphate () arranged in a crystalline structure, as hard as fired pottery.
- How you move — Your muscles run on glucose (), burning it in a controlled chemical reaction that releases energy molecule by molecule.
Chemistry is the study of matter and its transformations. And since everything is matter, chemistry is the study of everything.

Q1.Sodium, iron, and gold all contain protons, neutrons, and electrons. Why do they have such different chemical reactivities?