Estimation of Phosphorus and Oxygen
Completing the picture — the last two elements, and how oxygen is usually found by subtraction
Every element in an organic compound must account for some percentage of its mass, and all percentages must add up to exactly 100. So once you know %C, %H, %N, %S, and %halogen — you're done. %O = 100 − (sum of everything else). Oxygen is the only element routinely estimated "by difference". That's not laziness — it's elegant accounting.
Estimation of Phosphorus
Principle: A known mass of the organic compound is heated with fuming nitric acid. Phosphorus in the compound is oxidised to phosphoric acid ( ).
The is then precipitated in two alternative ways:
Method A — Ammonium phosphomolybdate:
Add ammonia and ammonium molybdate. A canary yellow precipitate of ammonium phosphomolybdate forms:
Molar mass = 1877 g/mol
Method B — Magnesium pyrophosphate:
Add magnesia mixture → forms → ignite → gives
Molar mass of = 222 g/mol; contains 2 × 31 = 62 g of P
Where = mass of compound, = mass of precipitate formed.
Two Phosphorus Formulas
As ammonium phosphomolybdate :
As magnesium pyrophosphate :
The 62 in the second formula = 2 phosphorus atoms (31 × 2) because contains 2 phosphorus atoms per formula unit.
Estimation of Oxygen — By Difference
The percentage of oxygen in an organic compound is usually calculated by difference:
This works because all elemental percentages must sum to 100%.
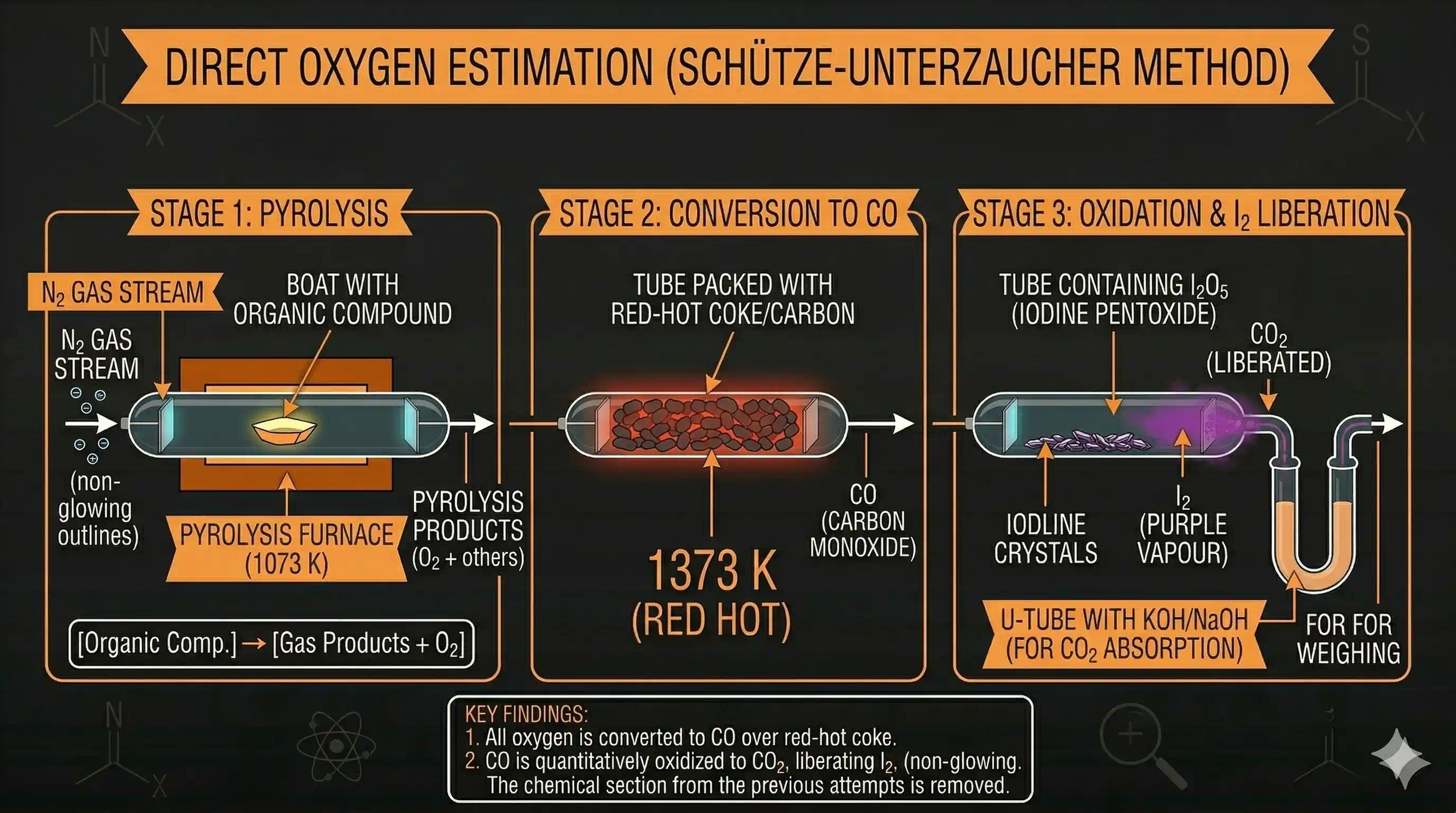
Direct Estimation of Oxygen
When a direct method is needed, the following process is used:
-
The organic compound is decomposed by heating in a stream of nitrogen gas. This releases oxygen and other gaseous products from the compound.
-
The gaseous products (containing ) are passed over red-hot coke (carbon) at high temperature. All oxygen is converted to carbon monoxide:
-
This gas stream is then passed through warm iodine pentoxide ( ). CO reduces the iodine pentoxide:
-
The iodine liberated is measured (or the produced is weighed).
Calculation: Each mole of from the compound gives 2 moles of CO (from the coke reaction), which gives 2 moles of (from the reaction):
where = mass of produced, = mass of compound
(since 88 g = 2 mol corresponds to 32 g O)
Q1.An organic compound contains 40% C, 6.67% H, and 53.33% O (by mass). The % of oxygen was found by:
Every element in an organic compound must account for some percentage of its mass, and all percentages must add up to exactly 100. So once you know %C, %H, %N, %S, and %halogen — you're done. %O = 100 − (sum of everything else). Oxygen is the only element routinely estimated "by difference". That's not laziness — it's elegant accounting.
Estimation of Phosphorus
Principle: A known mass of the organic compound is heated with fuming nitric acid. Phosphorus in the compound is oxidised to phosphoric acid ( ).
The is then precipitated in two alternative ways:
Method A — Ammonium phosphomolybdate:
Add ammonia and ammonium molybdate. A canary yellow precipitate of ammonium phosphomolybdate forms:
Molar mass = 1877 g/mol
Method B — Magnesium pyrophosphate:
Add magnesia mixture → forms → ignite → gives
Molar mass of = 222 g/mol; contains 2 × 31 = 62 g of P
Where = mass of compound, = mass of precipitate formed.

Estimation of Oxygen — By Difference
The percentage of oxygen in an organic compound is usually calculated by difference:
This works because all elemental percentages must sum to 100%.
Direct Estimation of Oxygen
When a direct method is needed, the following process is used:
-
The organic compound is decomposed by heating in a stream of nitrogen gas. This releases oxygen and other gaseous products from the compound.
-
The gaseous products (containing ) are passed over red-hot coke (carbon) at high temperature. All oxygen is converted to carbon monoxide:
-
This gas stream is then passed through warm iodine pentoxide ( ). CO reduces the iodine pentoxide:
-
The iodine liberated is measured (or the produced is weighed).
Calculation: Each mole of from the compound gives 2 moles of CO (from the coke reaction), which gives 2 moles of (from the reaction):
where = mass of produced, = mass of compound
(since 88 g = 2 mol corresponds to 32 g O)
Q1.An organic compound contains 40% C, 6.67% H, and 53.33% O (by mass). The % of oxygen was found by: