Crystallisation
Using hot and cold solubility to pull pure crystals out of an impure mixture

Think about salt — it comes out of the sea as a grey, gritty mess. But what you buy in a packet is perfectly white, clean crystals. That transformation happens through crystallisation, and it's one of the most elegant purification tricks in all of chemistry.
The technique works for any solid organic compound that needs to be purified from traces of other solids mixed in with it.
The Core Idea: Solubility Changes with Temperature
Most solid compounds dissolve much more freely in hot solvent than in cold solvent. Crystallisation exploits this gap.
Here's the logic:
- In hot solvent → both your compound and the impurities dissolve completely
- You cool the solution slowly → your compound, being present in large quantity, can no longer stay dissolved and forms neat, ordered crystals
- The impurities, present in tiny amounts, remain dissolved in the cooled liquid — they never reach a concentration high enough to crystallise out
Result: pure crystals of your compound, separated from everything else.
The Step-by-Step Process
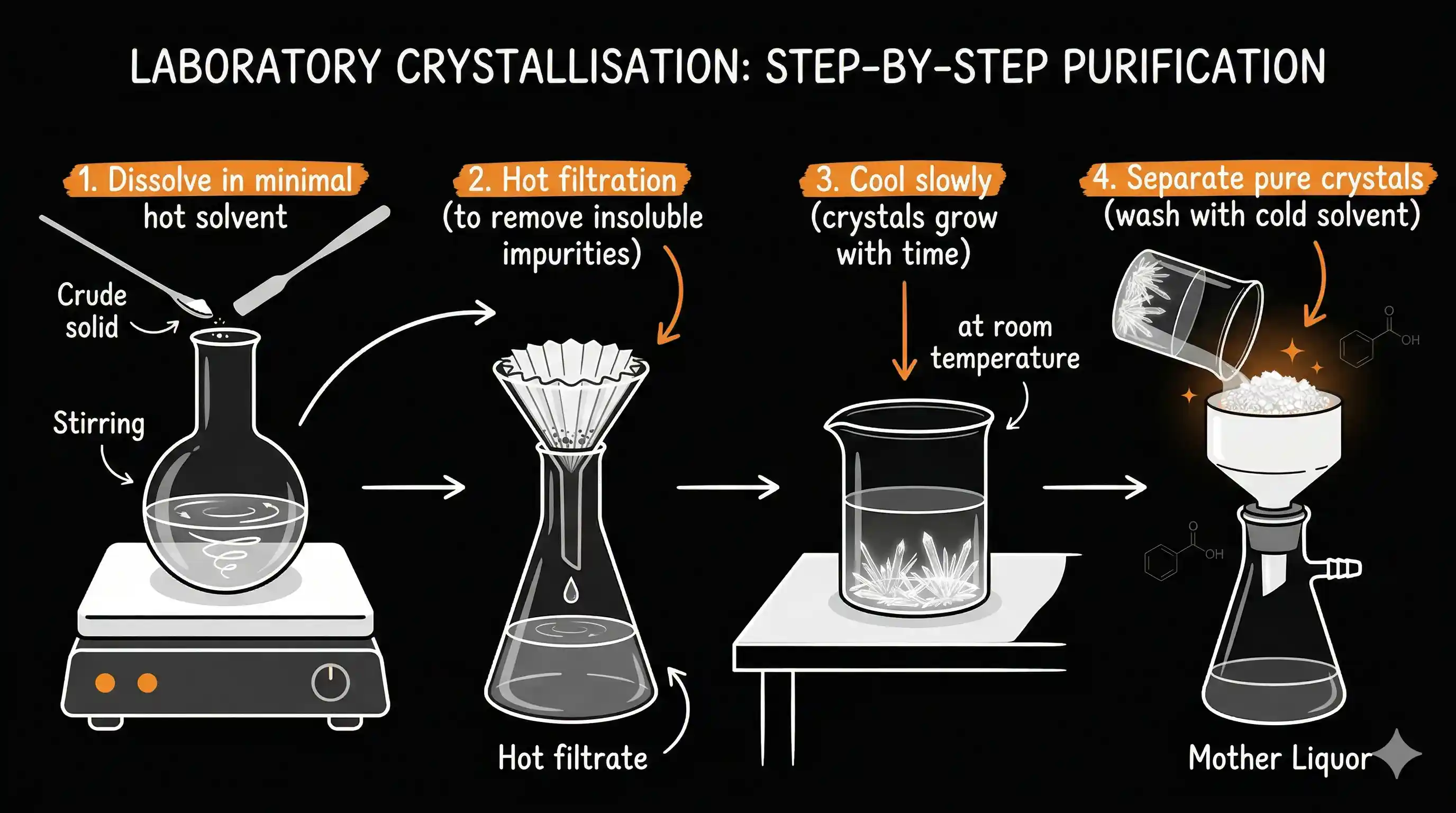
- Choose the right solvent — one in which the compound is sparingly soluble when cold, but freely soluble when hot.
- Dissolve in minimum hot solvent — heat the solvent, add the impure compound, dissolve with just enough solvent (not too much).
- Hot filtration — filter the hot solution through filter paper to remove any insoluble impurities (like sand or dust). Do this quickly before it cools.
- Concentrate if needed — gently evaporate some solvent to get a nearly saturated solution.
- Cool slowly — set the flask aside and let it cool at room temperature. Slow cooling gives large, well-formed crystals that trap fewer impurities. Rushing (ice bath immediately) gives fine powder that is harder to filter and less pure.
- Filter and dry — collect the crystals using vacuum filtration. Wash with a small amount of cold solvent to remove any mother liquor clinging to the surface. Dry in air or a warm oven.
The liquid left in the flask after removing the crystals is called the mother liquor. It contains the impurities plus a small amount of the compound that didn't crystallise.
When One Solvent Isn't Enough: Mixed Solvent Crystallisation
When a compound is too soluble in one solvent but too insoluble in another, use a mixed solvent pair. Dissolve in the minimum hot Solvent A, then add Solvent B drop by drop until the solution just turns cloudy — then cool slowly. The compound crystallises out as Solvent B reduces overall solubility below saturation. Classic example: ethanol + water.
Removing Coloured Impurities: Activated Charcoal
If the hot solution is coloured, add a small amount of activated charcoal (decolourising charcoal) and stir for a few minutes. Its enormous surface area adsorbs the coloured impurities, which are carried away when you filter out the charcoal during hot filtration — leaving a colourless filtrate and white crystals.
Repeated Crystallisation (Recrystallisation)
When impurities have a similar solubility to the compound, a single round may not give sufficient purity. Simply dissolve the crystals again in fresh hot solvent and repeat — each cycle pushes purity higher. Pharmaceutical compounds like aspirin are typically recrystallised 3–5 times before they meet purity standards.
Real-World Applications
Crystallisation is one of the most economically valuable separation and purification techniques in industry:
-
Pharmaceutical Manufacturing — Almost every solid drug you take (aspirin, paracetamol, ibuprofen, statins) is crystallised at least once during manufacture to achieve the legally required purity (>99.5 %). Industrial crystallisers are jacketed stainless steel vessels capable of processing tonnes of compound per batch. The crystal habit (shape) and size distribution directly affect how fast the tablet dissolves in the body.
-
Salt Production — India is the world's third-largest salt producer. The Rann of Kutch and Sambhar Lake produce salt by solar evaporation of brine — a natural crystallisation process driven by temperature and concentration. The Tata Salt and Nirma plants in Gujarat refine this using repeated recrystallisation to achieve food-grade purity.
-
Sugar Refining — Sucrose from sugarcane juice is crystallised by concentrating the syrup in vacuum pans (vacuum prevents caramelisation). Seed crystals are added to initiate uniform nucleation — exactly the principle you studied.
-
Semiconductor Industry — Silicon wafers for computer chips are grown by the Czochralski crystallisation process — pulling a single crystal of silicon from a melt at 1414 °C. The entire global semiconductor industry depends on the purity achievable by crystallisation.


Q1.Crystallisation is based on the difference in:

Think about salt — it comes out of the sea as a grey, gritty mess. But what you buy in a packet is perfectly white, clean crystals. That transformation happens through crystallisation, and it's one of the most elegant purification tricks in all of chemistry.
The technique works for any solid organic compound that needs to be purified from traces of other solids mixed in with it.
The Core Idea: Solubility Changes with Temperature
Most solid compounds dissolve much more freely in hot solvent than in cold solvent. Crystallisation exploits this gap.
Here's the logic:
- In hot solvent → both your compound and the impurities dissolve completely
- You cool the solution slowly → your compound, being present in large quantity, can no longer stay dissolved and forms neat, ordered crystals
- The impurities, present in tiny amounts, remain dissolved in the cooled liquid — they never reach a concentration high enough to crystallise out
Result: pure crystals of your compound, separated from everything else.
The Step-by-Step Process

- Choose the right solvent — one in which the compound is sparingly soluble when cold, but freely soluble when hot.
- Dissolve in minimum hot solvent — heat the solvent, add the impure compound, dissolve with just enough solvent (not too much).
- Hot filtration — filter the hot solution through filter paper to remove any insoluble impurities (like sand or dust). Do this quickly before it cools.
- Concentrate if needed — gently evaporate some solvent to get a nearly saturated solution.
- Cool slowly — set the flask aside and let it cool at room temperature. Slow cooling gives large, well-formed crystals that trap fewer impurities. Rushing (ice bath immediately) gives fine powder that is harder to filter and less pure.
- Filter and dry — collect the crystals using vacuum filtration. Wash with a small amount of cold solvent to remove any mother liquor clinging to the surface. Dry in air or a warm oven.
The liquid left in the flask after removing the crystals is called the mother liquor. It contains the impurities plus a small amount of the compound that didn't crystallise.
When One Solvent Isn't Enough: Mixed Solvent Crystallisation
When a compound is too soluble in one solvent but too insoluble in another, use a mixed solvent pair. Dissolve in the minimum hot Solvent A, then add Solvent B drop by drop until the solution just turns cloudy — then cool slowly. The compound crystallises out as Solvent B reduces overall solubility below saturation. Classic example: ethanol + water.
Removing Coloured Impurities: Activated Charcoal
If the hot solution is coloured, add a small amount of activated charcoal (decolourising charcoal) and stir for a few minutes. Its enormous surface area adsorbs the coloured impurities, which are carried away when you filter out the charcoal during hot filtration — leaving a colourless filtrate and white crystals.
Repeated Crystallisation (Recrystallisation)
When impurities have a similar solubility to the compound, a single round may not give sufficient purity. Simply dissolve the crystals again in fresh hot solvent and repeat — each cycle pushes purity higher. Pharmaceutical compounds like aspirin are typically recrystallised 3–5 times before they meet purity standards.
Real-World Applications
Crystallisation is one of the most economically valuable separation and purification techniques in industry:
-
Pharmaceutical Manufacturing — Almost every solid drug you take (aspirin, paracetamol, ibuprofen, statins) is crystallised at least once during manufacture to achieve the legally required purity (>99.5 %). Industrial crystallisers are jacketed stainless steel vessels capable of processing tonnes of compound per batch. The crystal habit (shape) and size distribution directly affect how fast the tablet dissolves in the body.
-
Salt Production — India is the world's third-largest salt producer. The Rann of Kutch and Sambhar Lake produce salt by solar evaporation of brine — a natural crystallisation process driven by temperature and concentration. The Tata Salt and Nirma plants in Gujarat refine this using repeated recrystallisation to achieve food-grade purity.
-
Sugar Refining — Sucrose from sugarcane juice is crystallised by concentrating the syrup in vacuum pans (vacuum prevents caramelisation). Seed crystals are added to initiate uniform nucleation — exactly the principle you studied.
-
Semiconductor Industry — Silicon wafers for computer chips are grown by the Czochralski crystallisation process — pulling a single crystal of silicon from a melt at 1414 °C. The entire global semiconductor industry depends on the purity achievable by crystallisation.


Q1.Crystallisation is based on the difference in: