Differential Extraction
Pulling a compound out of water and into an organic solvent using solubility differences
Imagine a compound dissolved in water — maybe it was synthesised in an aqueous reaction medium — and you need to isolate it. Evaporating all the water would destroy many compounds. Distillation won't work if the compound has a very high boiling point.
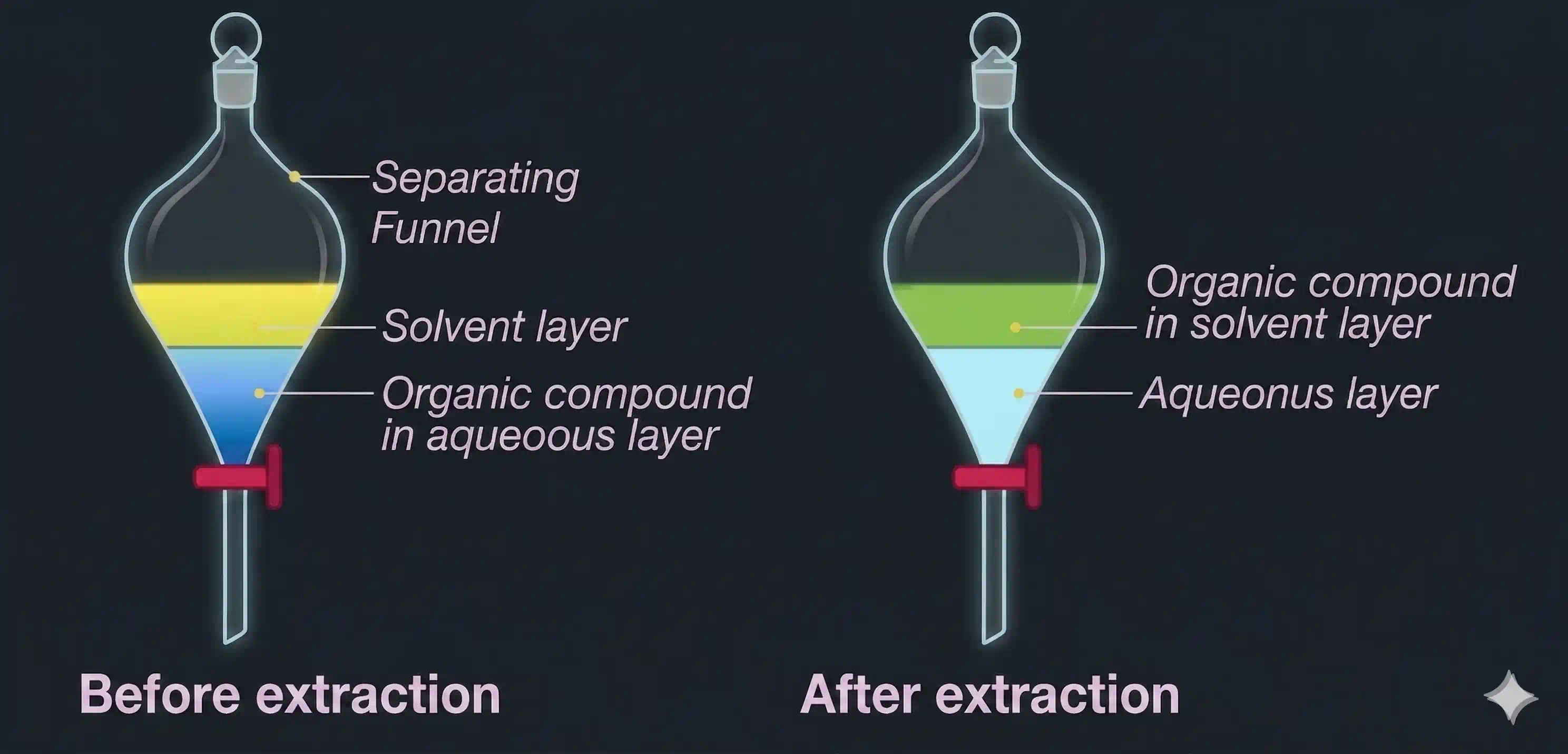
The answer is differential extraction (also called solvent extraction): you shake the aqueous solution with an immiscible organic solvent in which your compound is more soluble. The compound migrates from the water phase into the organic layer. Then you separate the two layers and evaporate the organic solvent — leaving behind the pure compound.
The Core Rule: Like Dissolves Like
The driving force is simple: the compound prefers the solvent it is more soluble in. For most organic compounds, a non-polar organic solvent like ether, chloroform, or ethyl acetate provides a far more hospitable environment than polar water.
For this to work, two conditions must be met:
- The organic solvent must be immiscible with water — so they form two distinct, separable layers
- The compound must be significantly more soluble in the organic solvent than in water
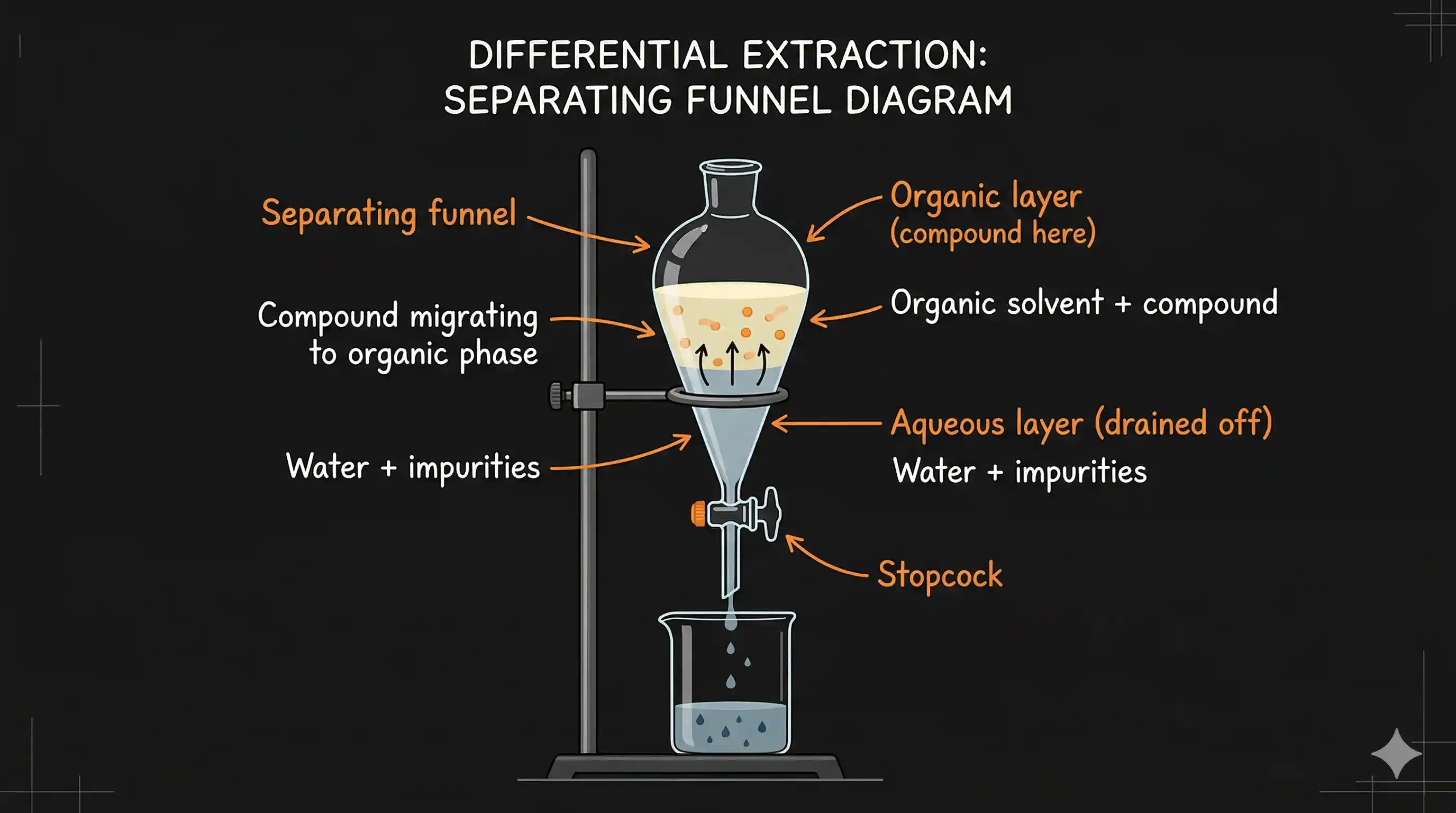
The Process: Using a Separating Funnel
- Pour the aqueous solution of the compound into a separating funnel.
- Add a measured volume of the organic solvent (e.g., diethyl ether).
- Stopper the funnel and shake it vigorously (with occasional venting to release pressure build-up). The compound redistributes between the two phases based on its relative solubility.
- Allow the funnel to stand still — the two layers separate clearly (organic layer usually floats on top since most organic solvents are less dense than water).
- Open the stopcock and drain the lower (aqueous) layer out through the bottom into a beaker.
- Pour the upper (organic) layer out through the top of the funnel.
- Evaporate the organic solvent by gentle heating or rotary evaporation — the pure compound remains behind.
For better recovery, the extraction is often done 2–3 times with fresh organic solvent. Multiple small-volume extractions are more efficient than one large-volume extraction.

Continuous Extraction: When the Compound Barely Moves
Differential extraction has a limitation: if the compound has low solubility in the organic solvent, you'd need enormous amounts of solvent to extract even a small quantity of the compound. Pouring in litres of ether just to collect a few milligrams is impractical.
The solution is continuous extraction. A clever apparatus continuously cycles the same organic solvent through the aqueous solution, evaporates it, and sends it back for another pass. Over time, even a poorly-extractable compound is gradually and completely pulled out — using the same modest volume of solvent repeatedly.
This technique is used in industrial processes where the cost of solvent matters and the compound is only sparingly soluble in the organic phase.
Real-World Applications
Liquid-liquid extraction (differential extraction) is an essential industrial process wherever two immiscible phases carry different compounds:
-
Pharmaceutical Manufacturing — Most drug synthesis reactions occur in water but the product partitions into an organic phase (ethyl acetate, dichloromethane, or toluene). Industrial extraction is done in continuous mixer-settler units or pulsed columns that handle thousands of litres per hour. The organic extract is then distilled to recover the solvent and crystallise the product.
-
Metal Hydrometallurgy — Copper, cobalt, uranium, and rare earth elements are extracted from acidic leach solutions using organic extractants. The Hindustan Copper Limited plant at Khetri, Rajasthan uses solvent extraction of copper ions from sulfate leach liquor. India's rare earth mining in Kerala uses similar extraction to separate cerium, lanthanum, and neodymium — critical for electric vehicle motors.
-
Decaffeination of Coffee — Green coffee beans are soaked in hot water (extracting caffeine and flavours). The water is then extracted with supercritical CO₂ or ethyl acetate (immiscible with water) — caffeine partitions into the organic phase while most flavour molecules remain in the water. The water is returned to the beans, removing caffeine but preserving flavour.
-
Vegetable Oil Extraction — Hexane (organic solvent, immiscible with water) is used to extract edible oil from soya beans, sunflower seeds, and groundnuts. The hexane-oil solution is then distilled to recover hexane (for reuse) and obtain crude vegetable oil.
-
Nuclear Reprocessing (PUREX Process) — Spent nuclear fuel is dissolved in nitric acid and extracted with tributyl phosphate in kerosene. Uranium and plutonium partition into the organic phase; fission products remain in the aqueous phase. India's Trombay BARC facility uses this for its nuclear programme.
Q1.For differential extraction to work, which condition is essential?
Imagine a compound dissolved in water — maybe it was synthesised in an aqueous reaction medium — and you need to isolate it. Evaporating all the water would destroy many compounds. Distillation won't work if the compound has a very high boiling point.
The answer is differential extraction (also called solvent extraction): you shake the aqueous solution with an immiscible organic solvent in which your compound is more soluble. The compound migrates from the water phase into the organic layer. Then you separate the two layers and evaporate the organic solvent — leaving behind the pure compound.
The Core Rule: Like Dissolves Like
The driving force is simple: the compound prefers the solvent it is more soluble in. For most organic compounds, a non-polar organic solvent like ether, chloroform, or ethyl acetate provides a far more hospitable environment than polar water.
For this to work, two conditions must be met:
- The organic solvent must be immiscible with water — so they form two distinct, separable layers
- The compound must be significantly more soluble in the organic solvent than in water
The Process: Using a Separating Funnel
- Pour the aqueous solution of the compound into a separating funnel.
- Add a measured volume of the organic solvent (e.g., diethyl ether).
- Stopper the funnel and shake it vigorously (with occasional venting to release pressure build-up). The compound redistributes between the two phases based on its relative solubility.
- Allow the funnel to stand still — the two layers separate clearly (organic layer usually floats on top since most organic solvents are less dense than water).
- Open the stopcock and drain the lower (aqueous) layer out through the bottom into a beaker.
- Pour the upper (organic) layer out through the top of the funnel.
- Evaporate the organic solvent by gentle heating or rotary evaporation — the pure compound remains behind.
For better recovery, the extraction is often done 2–3 times with fresh organic solvent. Multiple small-volume extractions are more efficient than one large-volume extraction.



Continuous Extraction: When the Compound Barely Moves
Differential extraction has a limitation: if the compound has low solubility in the organic solvent, you'd need enormous amounts of solvent to extract even a small quantity of the compound. Pouring in litres of ether just to collect a few milligrams is impractical.
The solution is continuous extraction. A clever apparatus continuously cycles the same organic solvent through the aqueous solution, evaporates it, and sends it back for another pass. Over time, even a poorly-extractable compound is gradually and completely pulled out — using the same modest volume of solvent repeatedly.
This technique is used in industrial processes where the cost of solvent matters and the compound is only sparingly soluble in the organic phase.
Real-World Applications
Liquid-liquid extraction (differential extraction) is an essential industrial process wherever two immiscible phases carry different compounds:
-
Pharmaceutical Manufacturing — Most drug synthesis reactions occur in water but the product partitions into an organic phase (ethyl acetate, dichloromethane, or toluene). Industrial extraction is done in continuous mixer-settler units or pulsed columns that handle thousands of litres per hour. The organic extract is then distilled to recover the solvent and crystallise the product.
-
Metal Hydrometallurgy — Copper, cobalt, uranium, and rare earth elements are extracted from acidic leach solutions using organic extractants. The Hindustan Copper Limited plant at Khetri, Rajasthan uses solvent extraction of copper ions from sulfate leach liquor. India's rare earth mining in Kerala uses similar extraction to separate cerium, lanthanum, and neodymium — critical for electric vehicle motors.
-
Decaffeination of Coffee — Green coffee beans are soaked in hot water (extracting caffeine and flavours). The water is then extracted with supercritical CO₂ or ethyl acetate (immiscible with water) — caffeine partitions into the organic phase while most flavour molecules remain in the water. The water is returned to the beans, removing caffeine but preserving flavour.
-
Vegetable Oil Extraction — Hexane (organic solvent, immiscible with water) is used to extract edible oil from soya beans, sunflower seeds, and groundnuts. The hexane-oil solution is then distilled to recover hexane (for reuse) and obtain crude vegetable oil.
-
Nuclear Reprocessing (PUREX Process) — Spent nuclear fuel is dissolved in nitric acid and extracted with tributyl phosphate in kerosene. Uranium and plutonium partition into the organic phase; fission products remain in the aqueous phase. India's Trombay BARC facility uses this for its nuclear programme.
Q1.For differential extraction to work, which condition is essential?