Sublimation
Have you ever noticed that naphthalene balls (mothballs) placed in your cupboard slowly shrink and disappear — but your cupboard never gets wet? Or how dry ice (solid CO₂) used at events just vanishes into a cloud of mist without any puddle? That's sublimation at work.
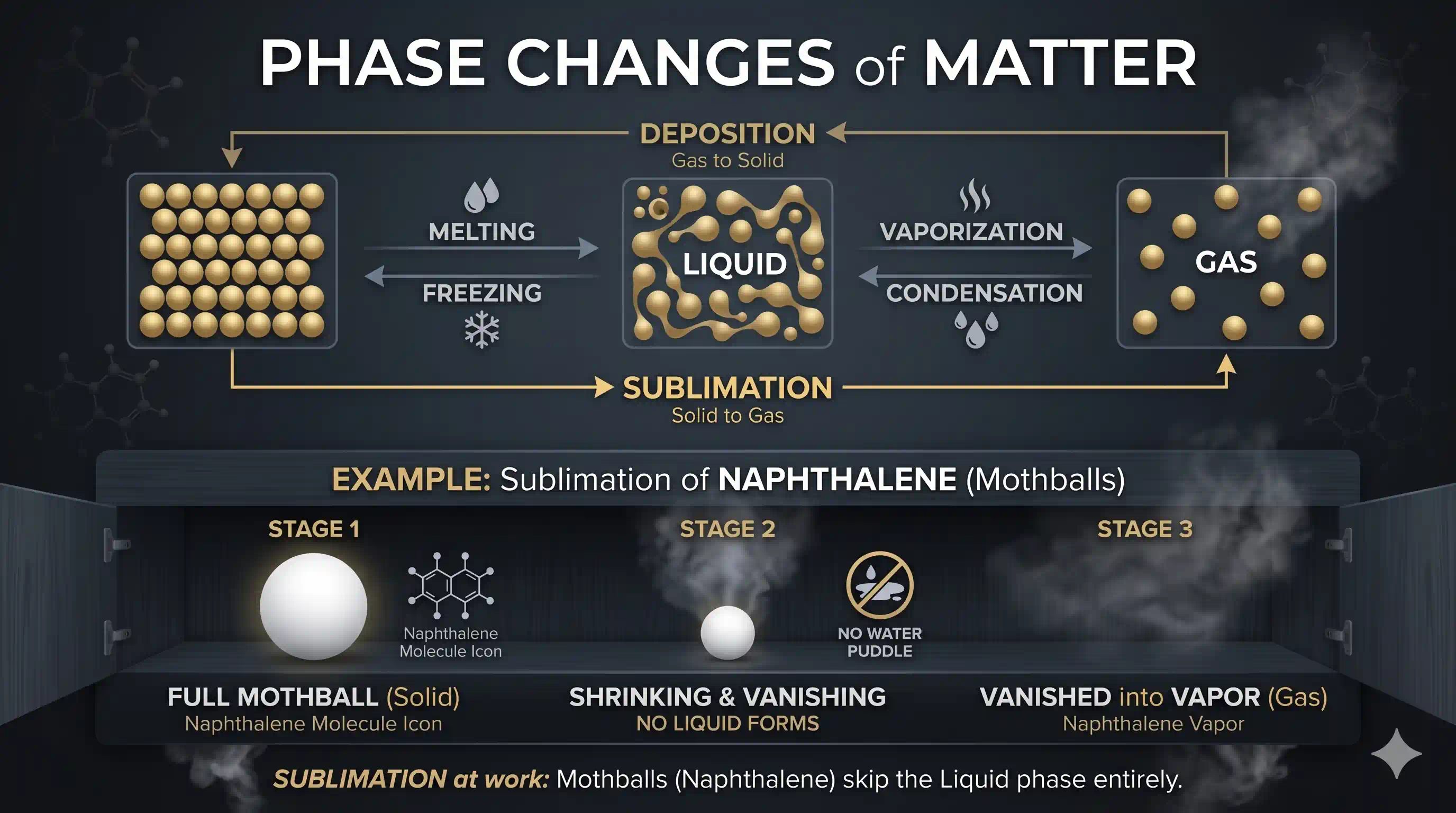
What is Sublimation?
Most substances follow a predictable path when heated: solid → liquid → gas. But a few "rule-breakers" skip the liquid stage entirely and jump straight from solid to gas. This direct solid-to-gas transformation is called sublimation.
How do we use this for purification?
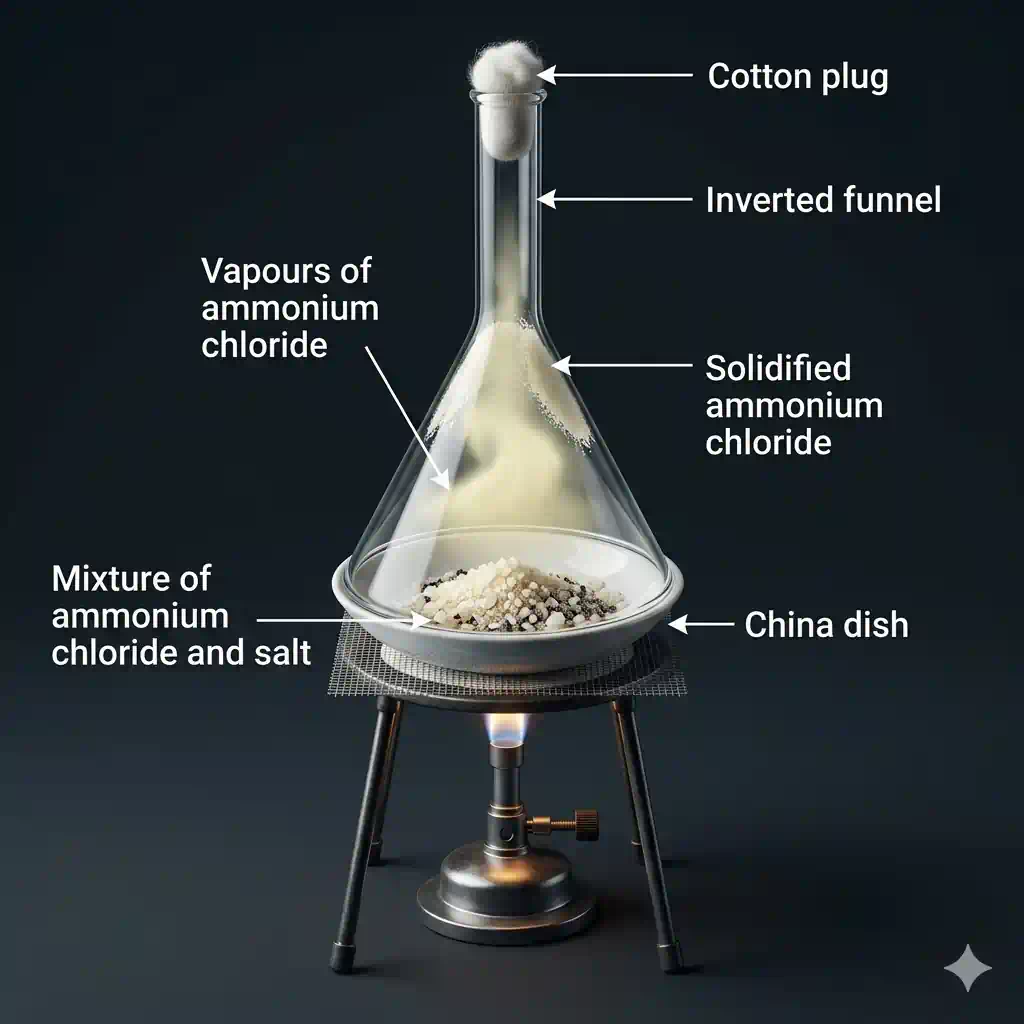
If a mixture contains a solid that sublimes (like iodine or naphthalene) mixed with a solid that does NOT sublime (like sand or salt), we can separate them cleanly:
- Heat the mixture gently.
- The sublimable solid turns directly into vapour and rises.
- The vapour is cooled on a surface above (an inverted funnel), where it deposits back as pure solid crystals.
- The non-sublimable impurities are left behind at the bottom.
The result is a pure sample of the sublimable substance — no filtering, no dissolving needed.
JEE / NEET Exam Insight
Substances that sublime: Iodine (), Naphthalene (), Camphor, Ammonium chloride (), Dry ice ().
Key condition: The substance must be able to sublime AND the impurity must NOT sublime. If both sublime, this method fails.
Most common exam question: "Which method would you use to purify iodine contaminated with sand?" → Sublimation.
Q1.Sublimation is a phase change in which a substance converts directly from:
Have you ever noticed that naphthalene balls (mothballs) placed in your cupboard slowly shrink and disappear — but your cupboard never gets wet? Or how dry ice (solid CO₂) used at events just vanishes into a cloud of mist without any puddle? That's sublimation at work.

What is Sublimation?
Most substances follow a predictable path when heated: solid → liquid → gas. But a few "rule-breakers" skip the liquid stage entirely and jump straight from solid to gas. This direct solid-to-gas transformation is called sublimation.
How do we use this for purification?
If a mixture contains a solid that sublimes (like iodine or naphthalene) mixed with a solid that does NOT sublime (like sand or salt), we can separate them cleanly:
- Heat the mixture gently.
- The sublimable solid turns directly into vapour and rises.
- The vapour is cooled on a surface above (an inverted funnel), where it deposits back as pure solid crystals.
- The non-sublimable impurities are left behind at the bottom.
The result is a pure sample of the sublimable substance — no filtering, no dissolving needed.

Q1.Sublimation is a phase change in which a substance converts directly from: