Special Distillation Techniques
When the compound would decompose before it boils — or is stuck in water
Normal distillation has one big weakness: you have to heat the liquid to its boiling point. For many organic compounds — especially large, complex molecules used in pharmaceuticals and perfumes — the boiling point is too high. By the time the liquid reaches its boiling point, it has already decomposed or changed colour.
And then there are liquids that are immiscible with water (they don't mix). For these, a different strategy is needed.
Two special techniques handle these problems: distillation under reduced pressure and steam distillation.
Distillation Under Reduced Pressure (Vacuum Distillation)
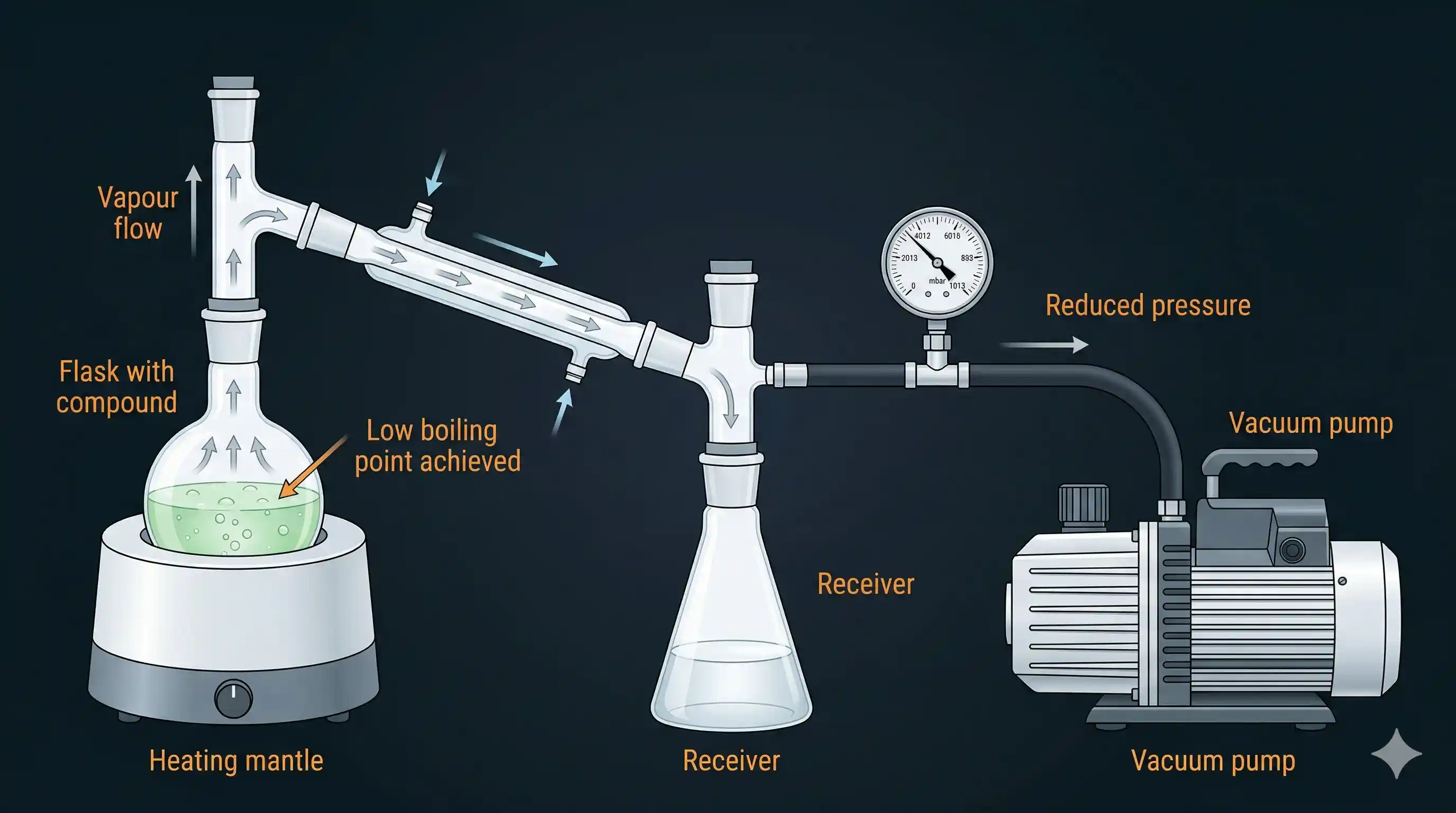
A liquid boils when its vapour pressure equals the external (atmospheric) pressure. At normal atmospheric pressure ( atm), water boils at .
But here's the clever part: if you reduce the external pressure — using a vacuum pump or water pump — the liquid can boil at a much lower temperature. This is exactly how vacuum distillation works.
The setup: the distillation flask is connected to a vacuum pump that pulls out most of the air, reducing the pressure above the liquid. At the reduced pressure, the liquid boils at a safe, lower temperature — well below the temperature at which it would decompose.
Classic example: Glycerol (used in medicines, cosmetics, food) has a very high boiling point (290°C) and decomposes near its boiling point. In soap manufacturing, glycerol is recovered from the spent lye using vacuum distillation — it can be distilled safely at around 180°C under reduced pressure.

Steam Distillation
Steam distillation is used for liquids that are immiscible with water (they don't mix with water to form a solution, but float as a separate layer) and that are volatile with steam (they can vaporise along with water vapour).
The key principle comes from Dalton's Law of Partial Pressures:
For a mixture of two immiscible liquids, the total vapour pressure is the sum of the individual vapour pressures:
where is the vapour pressure of the organic compound and is the vapour pressure of water.
Since is added to , the mixture boils when their combined vapour pressures reach atmospheric pressure — which happens at a temperature lower than the boiling point of either component alone. The organic compound gets to vaporise at a temperature well below 100°C, which is perfect for heat-sensitive materials.
The process:
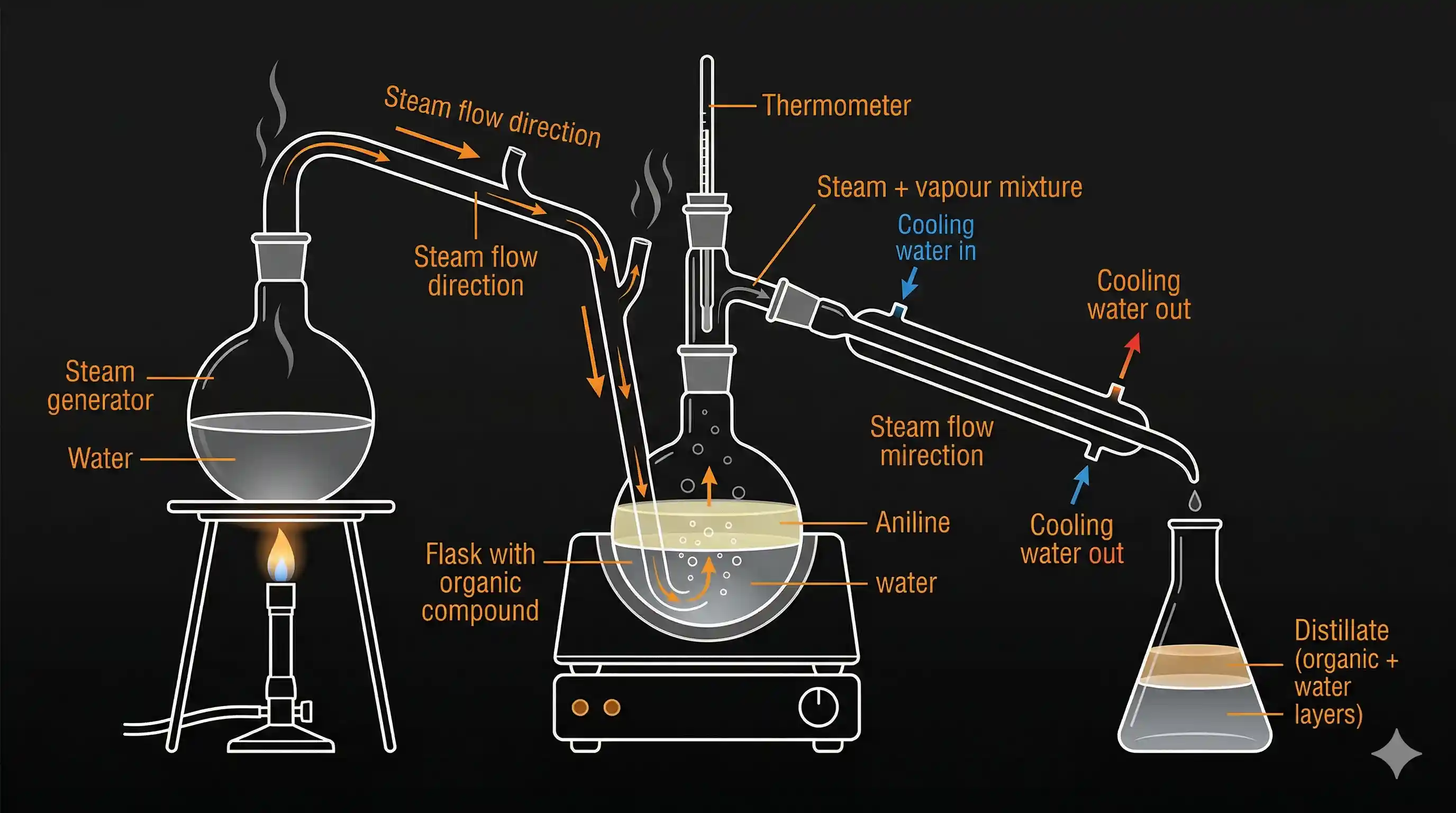
- Steam from a steam generator is passed through the heated flask containing the organic liquid.
- Steam and organic vapour mix and travel together into the condenser.
- The condensed mixture (water + organic liquid) is collected and then separated using a separating funnel (since they are immiscible, they form two clean layers).
Real example — Aniline from aniline–water mixture:
Aniline ( ) boils at 184°C normally, but it is immiscible with water. Using steam distillation, the aniline–water mixture boils at about 98°C — well below the boiling point of aniline alone. This prevents aniline (which can degrade at high temperatures) from decomposing, and separates it cleanly from the water.
Steam distillation is the industry-standard method for extracting essential oils from plant material because it preserves delicate aromatic compounds that would break down under direct heat. Rose, lavender, eucalyptus, peppermint, sandalwood, and tea tree oils are all commercially produced this way.
Perfumery and cosmetics is the largest application — essential oils extracted via steam distillation form the backbone of fine fragrances, skincare serums, soaps, and lotions. India's historic attar industry in Kannauj has used this method for centuries, producing sandalwood and vetiver oils that are traded globally.
Pharmaceuticals and healthcare rely on it for eucalyptus oil in cough syrups and chest rubs, peppermint oil in digestive medicines, and tea tree oil in antiseptic formulations.
Food and flavoring is another major sector — clove, cinnamon, and peppermint oils produced this way flavor everything from toothpaste and confectionery to packaged beverages.
Aromatherapy has grown into a significant industry of its own, with lavender, chamomile, and frankincense oils being among the most commercially extracted.
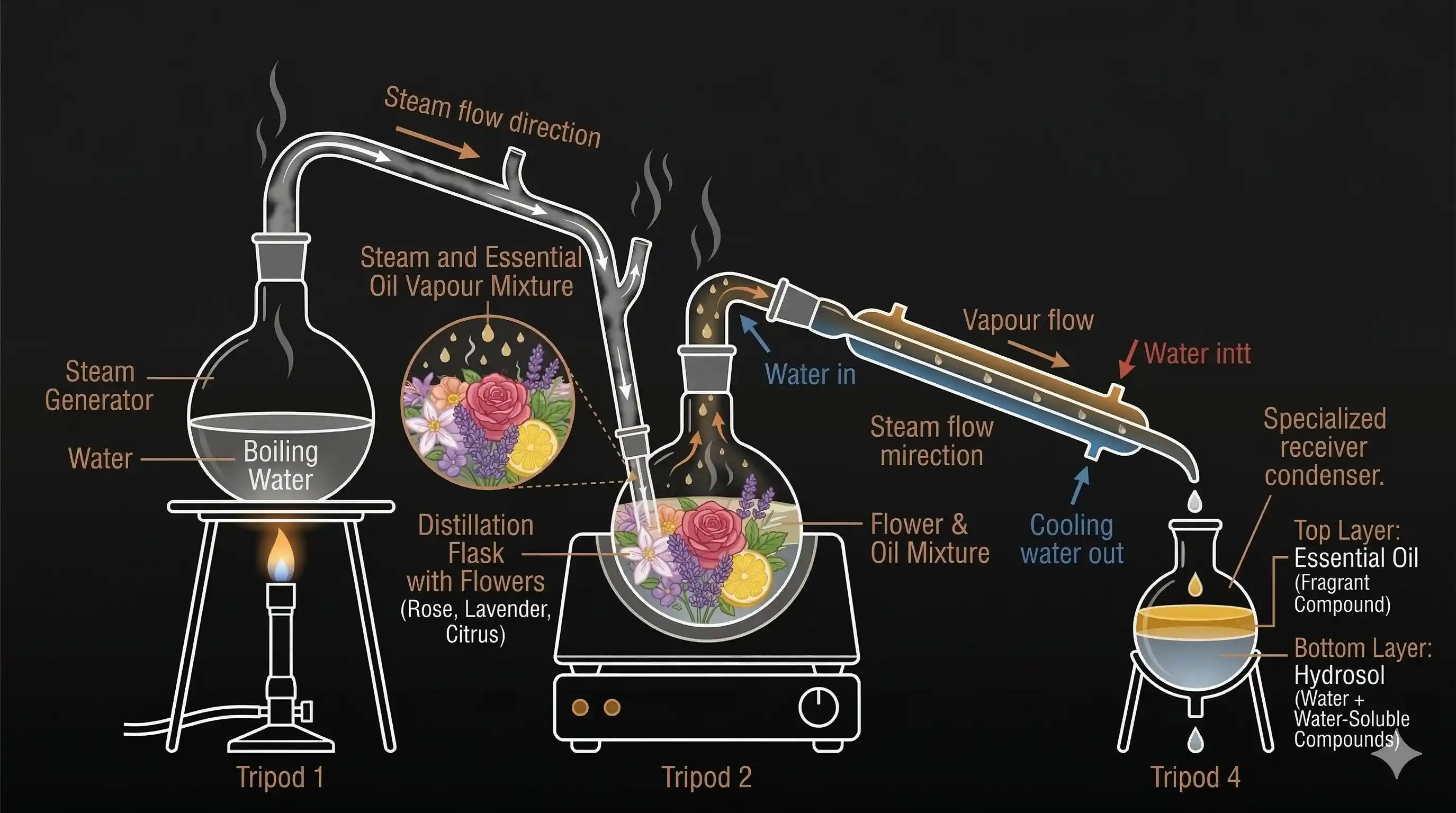

One striking example of the process's value: several tonnes of rose petals yield just one kilogram of rose essential oil, which explains why pure rose attar is one of the most expensive natural substances in the world.
Real-World Applications at a Glance
Vacuum Distillation is the standard technique whenever a compound:
- Has a boiling point above 200°C at atmospheric pressure
- Would decompose, polymerise, or oxidise before reaching its boiling point
Industrial examples:
- Vitamin production (Vitamin E, Vitamin K₂) — purified by molecular distillation at <0.001 mmHg
- Petroleum lubricant refining — high-boiling lubricant fractions that would crack at atmospheric distillation temperatures
- Glycerol purification (BP 290°C → distils at ~210°C under vacuum) — used in pharmaceuticals, cosmetics, and food
- Perfume distillation — high-value aroma molecules like vetiver oil distil under vacuum to prevent decomposition
Steam Distillation is the standard technique whenever a compound:
- Is immiscible with water
- Is steam-volatile (has non-negligible vapour pressure at 100°C)
- Would decompose if distilled alone (boiling point too high)
Industrial examples:
- Essential oil production — rose, jasmine, sandalwood, eucalyptus, peppermint, clove
- Aniline recovery from industrial waste streams
- Turpentine production from pine resin (BP 160°C, steam distilled at ~95°C)
- Food flavour extraction — vanilla, cinnamon, cardamom oleoresins for the food industry
India is the world's largest producer of essential oils by steam distillation — exporting over ₹3,000 crore worth annually.

Q1.Distillation under reduced pressure is used when the compound:
Normal distillation has one big weakness: you have to heat the liquid to its boiling point. For many organic compounds — especially large, complex molecules used in pharmaceuticals and perfumes — the boiling point is too high. By the time the liquid reaches its boiling point, it has already decomposed or changed colour.
And then there are liquids that are immiscible with water (they don't mix). For these, a different strategy is needed.
Two special techniques handle these problems: distillation under reduced pressure and steam distillation.
Distillation Under Reduced Pressure (Vacuum Distillation)
A liquid boils when its vapour pressure equals the external (atmospheric) pressure. At normal atmospheric pressure ( atm), water boils at .
But here's the clever part: if you reduce the external pressure — using a vacuum pump or water pump — the liquid can boil at a much lower temperature. This is exactly how vacuum distillation works.
The setup: the distillation flask is connected to a vacuum pump that pulls out most of the air, reducing the pressure above the liquid. At the reduced pressure, the liquid boils at a safe, lower temperature — well below the temperature at which it would decompose.
Classic example: Glycerol (used in medicines, cosmetics, food) has a very high boiling point (290°C) and decomposes near its boiling point. In soap manufacturing, glycerol is recovered from the spent lye using vacuum distillation — it can be distilled safely at around 180°C under reduced pressure.


Steam Distillation
Steam distillation is used for liquids that are immiscible with water (they don't mix with water to form a solution, but float as a separate layer) and that are volatile with steam (they can vaporise along with water vapour).
The key principle comes from Dalton's Law of Partial Pressures:
For a mixture of two immiscible liquids, the total vapour pressure is the sum of the individual vapour pressures:
where is the vapour pressure of the organic compound and is the vapour pressure of water.
Since is added to , the mixture boils when their combined vapour pressures reach atmospheric pressure — which happens at a temperature lower than the boiling point of either component alone. The organic compound gets to vaporise at a temperature well below 100°C, which is perfect for heat-sensitive materials.
The process:
- Steam from a steam generator is passed through the heated flask containing the organic liquid.
- Steam and organic vapour mix and travel together into the condenser.
- The condensed mixture (water + organic liquid) is collected and then separated using a separating funnel (since they are immiscible, they form two clean layers).
Real example — Aniline from aniline–water mixture:
Aniline ( ) boils at 184°C normally, but it is immiscible with water. Using steam distillation, the aniline–water mixture boils at about 98°C — well below the boiling point of aniline alone. This prevents aniline (which can degrade at high temperatures) from decomposing, and separates it cleanly from the water.

Steam distillation is the industry-standard method for extracting essential oils from plant material because it preserves delicate aromatic compounds that would break down under direct heat. Rose, lavender, eucalyptus, peppermint, sandalwood, and tea tree oils are all commercially produced this way.
Perfumery and cosmetics is the largest application — essential oils extracted via steam distillation form the backbone of fine fragrances, skincare serums, soaps, and lotions. India's historic attar industry in Kannauj has used this method for centuries, producing sandalwood and vetiver oils that are traded globally.
Pharmaceuticals and healthcare rely on it for eucalyptus oil in cough syrups and chest rubs, peppermint oil in digestive medicines, and tea tree oil in antiseptic formulations.
Food and flavoring is another major sector — clove, cinnamon, and peppermint oils produced this way flavor everything from toothpaste and confectionery to packaged beverages.
Aromatherapy has grown into a significant industry of its own, with lavender, chamomile, and frankincense oils being among the most commercially extracted.


One striking example of the process's value: several tonnes of rose petals yield just one kilogram of rose essential oil, which explains why pure rose attar is one of the most expensive natural substances in the world.
Real-World Applications at a Glance
Vacuum Distillation is the standard technique whenever a compound:
- Has a boiling point above 200°C at atmospheric pressure
- Would decompose, polymerise, or oxidise before reaching its boiling point
Industrial examples:
- Vitamin production (Vitamin E, Vitamin K₂) — purified by molecular distillation at <0.001 mmHg
- Petroleum lubricant refining — high-boiling lubricant fractions that would crack at atmospheric distillation temperatures
- Glycerol purification (BP 290°C → distils at ~210°C under vacuum) — used in pharmaceuticals, cosmetics, and food
- Perfume distillation — high-value aroma molecules like vetiver oil distil under vacuum to prevent decomposition
Steam Distillation is the standard technique whenever a compound:
- Is immiscible with water
- Is steam-volatile (has non-negligible vapour pressure at 100°C)
- Would decompose if distilled alone (boiling point too high)
Industrial examples:
- Essential oil production — rose, jasmine, sandalwood, eucalyptus, peppermint, clove
- Aniline recovery from industrial waste streams
- Turpentine production from pine resin (BP 160°C, steam distilled at ~95°C)
- Food flavour extraction — vanilla, cinnamon, cardamom oleoresins for the food industry
India is the world's largest producer of essential oils by steam distillation — exporting over ₹3,000 crore worth annually.

Q1.Distillation under reduced pressure is used when the compound: