Qualitative Analysis of Organic Compounds
How chemists detect which elements are hiding inside an unknown organic compound
You pick up an unlabelled white powder. Is it dangerous? Is it a drug? Is it just table sugar? Before any structural analysis is possible, a chemist first asks: what elements are in this thing? That's qualitative analysis — and it's the gateway to understanding any unknown organic compound.
Every organic compound contains carbon and hydrogen as its backbone. Beyond that, it may also contain oxygen, nitrogen, sulphur, halogens (Cl, Br, I), and phosphorus. Qualitative analysis means detecting which of these elements are present.
The methods differ depending on the element:
- Carbon and hydrogen — detected by heating with copper(II) oxide
- Nitrogen, sulphur, halogens, phosphorus — detected by Lassaigne's test (sodium fusion)
Detection of Carbon and Hydrogen
The compound is heated strongly with copper(II) oxide ( ). The CuO acts as an oxidising agent, converting:
- Carbon → carbon dioxide ( )
- Hydrogen → water ( )
The reactions are:
Testing the products:
- is confirmed by lime water — it turns milky/turbid:
- is confirmed by anhydrous copper sulphate — it turns from white to blue:
If both tests are positive → compound contains both C and H.
If only CO₂ test is positive → compound contains C but no H (rare).
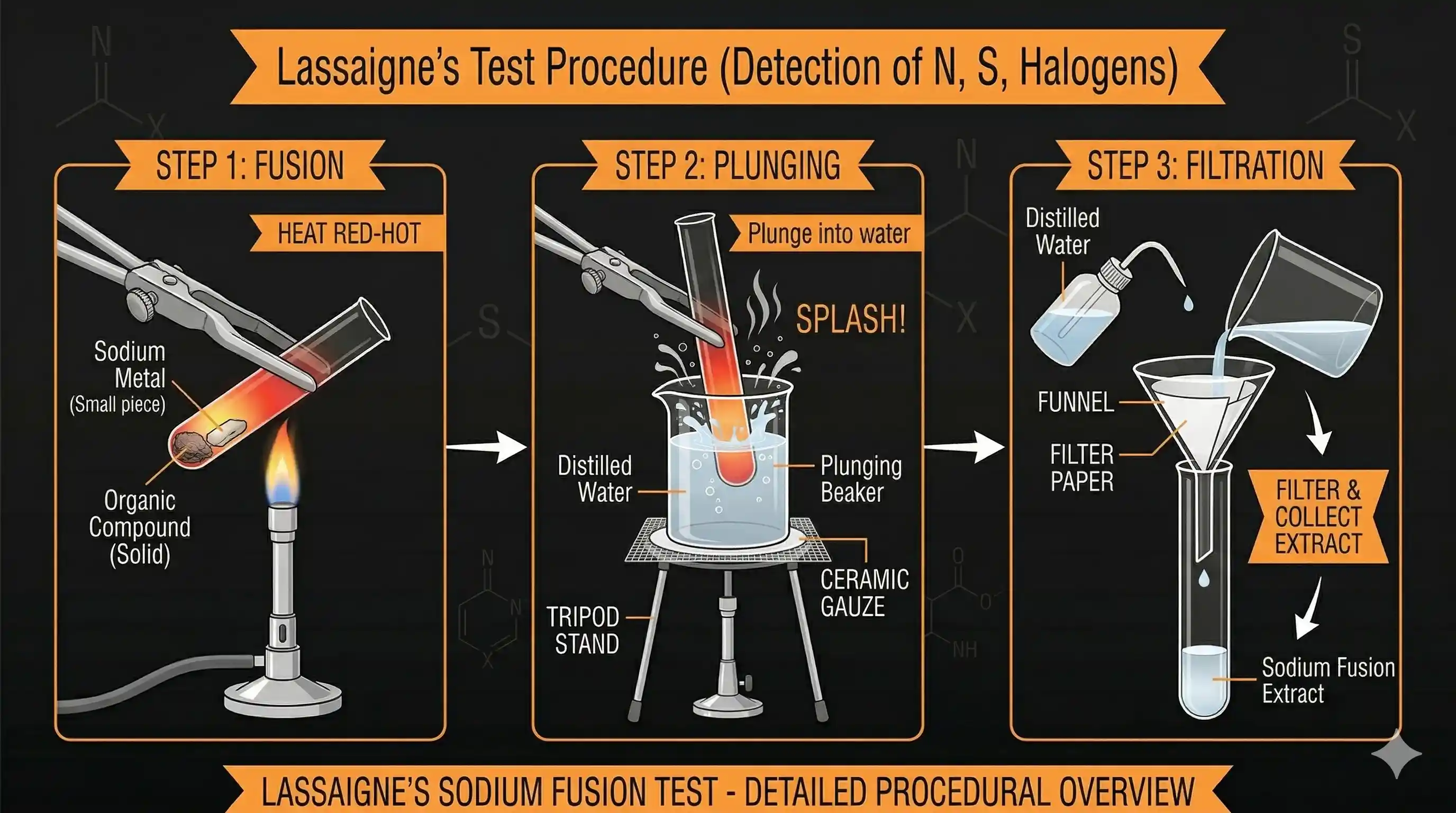
Lassaigne's Test (Sodium Fusion)
Nitrogen, sulphur, halogens, and phosphorus are bonded covalently inside organic compounds — which means they won't respond to the usual ionic tests. The trick is to break those covalent bonds and convert these elements into ionic form.
This is done by fusing the compound with sodium metal at high temperature. The sodium rips the elements out of the organic compound and forms simple ionic compounds:
The fused mass is then boiled with distilled water to extract the ionic products. This aqueous extract is the sodium fusion extract (SFE), and separate portions are used to test for each element.
Test for Nitrogen — Prussian Blue
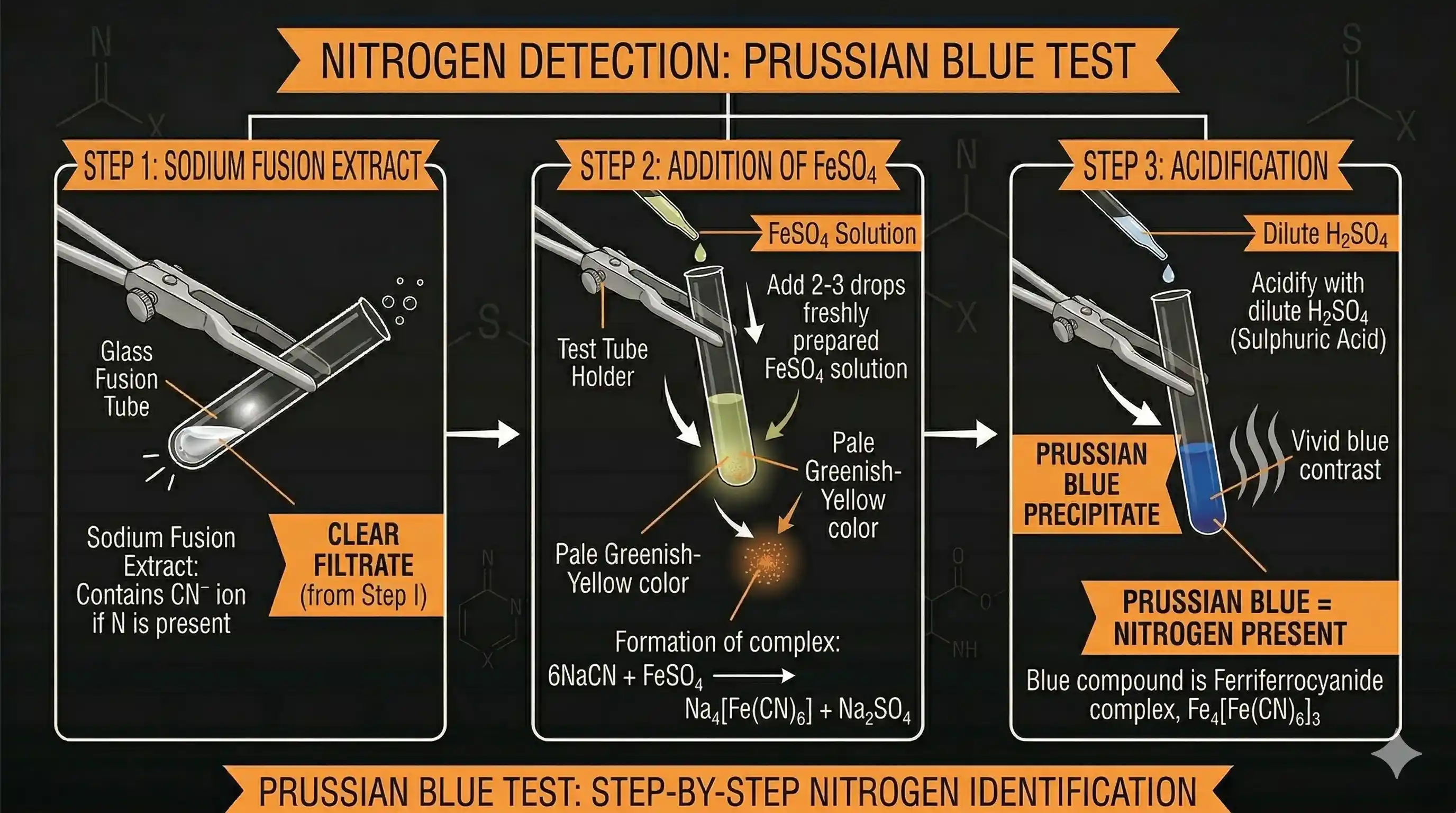
A portion of the SFE is boiled with iron(II) sulphate ( ) and then acidified with concentrated sulphuric acid.
Positive result: A Prussian blue precipitate confirms nitrogen.
The chemistry:
The second compound — iron(III) hexacyanoferrate(II) — is Prussian blue ( ).
Note: the acidification with oxidises some to , which is needed for the blue precipitate to form.
Test for Sulphur — Two Methods
(a) Lead acetate test: Acidify the SFE with acetic acid, then add lead acetate solution. A black precipitate of lead sulphide ( ) confirms sulphur:
(b) Sodium nitroprusside test: Add sodium nitroprusside ( ) to the SFE. A violet or purple colour confirms sulphur. This is a more sensitive test.
Test for Halogens — Silver Nitrate Test
Acidify the SFE with dilute nitric acid (to decompose any or that would interfere), then add silver nitrate ( ) solution.
| Precipitate | Solubility in ammonia | Element present |
|---|---|---|
| White | Soluble in dil. | Chlorine |
| Pale yellow | Partially soluble in conc. | Bromine |
| Yellow | Insoluble in conc. | Iodine |
Important: If N or S is also present in the compound, the SFE must be boiled with concentrated nitric acid first to destroy and — otherwise they would form (white) or (black) and give a false positive.
Test for Phosphorus
The SFE is acidified with concentrated nitric acid and treated with ammonium molybdate ( ) and ammonia.
Positive result: A canary yellow precipitate of ammonium phosphomolybdate confirms phosphorus:

Q1.In Lassaigne's test, the organic compound is fused with sodium metal. The purpose of this step is to:
You pick up an unlabelled white powder. Is it dangerous? Is it a drug? Is it just table sugar? Before any structural analysis is possible, a chemist first asks: what elements are in this thing? That's qualitative analysis — and it's the gateway to understanding any unknown organic compound.
Every organic compound contains carbon and hydrogen as its backbone. Beyond that, it may also contain oxygen, nitrogen, sulphur, halogens (Cl, Br, I), and phosphorus. Qualitative analysis means detecting which of these elements are present.
The methods differ depending on the element:
- Carbon and hydrogen — detected by heating with copper(II) oxide
- Nitrogen, sulphur, halogens, phosphorus — detected by Lassaigne's test (sodium fusion)
Detection of Carbon and Hydrogen
The compound is heated strongly with copper(II) oxide ( ). The CuO acts as an oxidising agent, converting:
- Carbon → carbon dioxide ( )
- Hydrogen → water ( )
The reactions are:
Testing the products:
- is confirmed by lime water — it turns milky/turbid:
- is confirmed by anhydrous copper sulphate — it turns from white to blue:
If both tests are positive → compound contains both C and H.
If only CO₂ test is positive → compound contains C but no H (rare).
Lassaigne's Test (Sodium Fusion)
Nitrogen, sulphur, halogens, and phosphorus are bonded covalently inside organic compounds — which means they won't respond to the usual ionic tests. The trick is to break those covalent bonds and convert these elements into ionic form.
This is done by fusing the compound with sodium metal at high temperature. The sodium rips the elements out of the organic compound and forms simple ionic compounds:
The fused mass is then boiled with distilled water to extract the ionic products. This aqueous extract is the sodium fusion extract (SFE), and separate portions are used to test for each element.

Test for Nitrogen — Prussian Blue

A portion of the SFE is boiled with iron(II) sulphate ( ) and then acidified with concentrated sulphuric acid.
Positive result: A Prussian blue precipitate confirms nitrogen.
The chemistry:
The second compound — iron(III) hexacyanoferrate(II) — is Prussian blue ( ).
Note: the acidification with oxidises some to , which is needed for the blue precipitate to form.
Test for Sulphur — Two Methods
(a) Lead acetate test: Acidify the SFE with acetic acid, then add lead acetate solution. A black precipitate of lead sulphide ( ) confirms sulphur:
(b) Sodium nitroprusside test: Add sodium nitroprusside ( ) to the SFE. A violet or purple colour confirms sulphur. This is a more sensitive test.
Test for Halogens — Silver Nitrate Test
Acidify the SFE with dilute nitric acid (to decompose any or that would interfere), then add silver nitrate ( ) solution.
| Precipitate | Solubility in ammonia | Element present |
|---|---|---|
| White | Soluble in dil. | Chlorine |
| Pale yellow | Partially soluble in conc. | Bromine |
| Yellow | Insoluble in conc. | Iodine |
Important: If N or S is also present in the compound, the SFE must be boiled with concentrated nitric acid first to destroy and — otherwise they would form (white) or (black) and give a false positive.
Test for Phosphorus
The SFE is acidified with concentrated nitric acid and treated with ammonium molybdate ( ) and ammonia.
Positive result: A canary yellow precipitate of ammonium phosphomolybdate confirms phosphorus:

Q1.In Lassaigne's test, the organic compound is fused with sodium metal. The purpose of this step is to: