Chromatography
How a single technique can separate, identify, and purify compounds — all at once
A forensic analyst runs a TLC plate on ink recovered from a suspected forged document. To the naked eye, the ink matches the original perfectly. Under UV light on the developed plate, the original shows one spot — but the suspected forgery shows three. One extra dye component matches a synthetic compound not manufactured until 1998. The document is allegedly dated 1979. The forgery is proven without a confession.
In another lab, gas chromatography detects a single pesticide molecule among one billion water molecules in a fruit sample — below any health threshold, but still measurable. At a crime scene, unknown white powder is identified not by colour or smell but by its chromatographic fingerprint: the pattern of retention times is as unique as a barcode.
Chromatography is now used in anti-doping tests (WADA screens for hundreds of prohibited substances in a single urine sample), airport security (trace explosive detectors analyse swabs chromatographically in seconds), and food authentication (the olive oil in your bottle can be fingerprinted to confirm its geographic origin).
Mikhail Tswett invented the technique in 1903 to separate plant pigments. The same principle — differential migration based on affinity for stationary vs. mobile phase — now catches forgers, dopers, and poisoners. Chromatography is arguably the single most powerful analytical tool ever developed.
What is Chromatography?
‘Chromatography’ is an analytical technique commonly used for separating a mixture of chemical substances into its individual components, so that the individual components can be thoroughly analyzed. The name comes from the Greek chroma (colour) — because when it was first used, scientists were separating coloured plant pigments. Today, chromatography separates everything from amino acids to drugs to petroleum fractions.
The core idea is elegant. A mixture is applied onto a stationary phase (a solid or liquid that stays fixed). A mobile phase (a solvent or gas) is then allowed to flow over it. As it moves, it carries the components of the mixture along — but each component travels at a different speed, depending on how strongly it sticks to the stationary phase.
- Components that stick strongly to the stationary phase → move slowly, stay near the start
- Components that prefer the mobile phase → move quickly, travel far
Result: the mixture gets spread out in space, each component at a different position — separation is achieved.
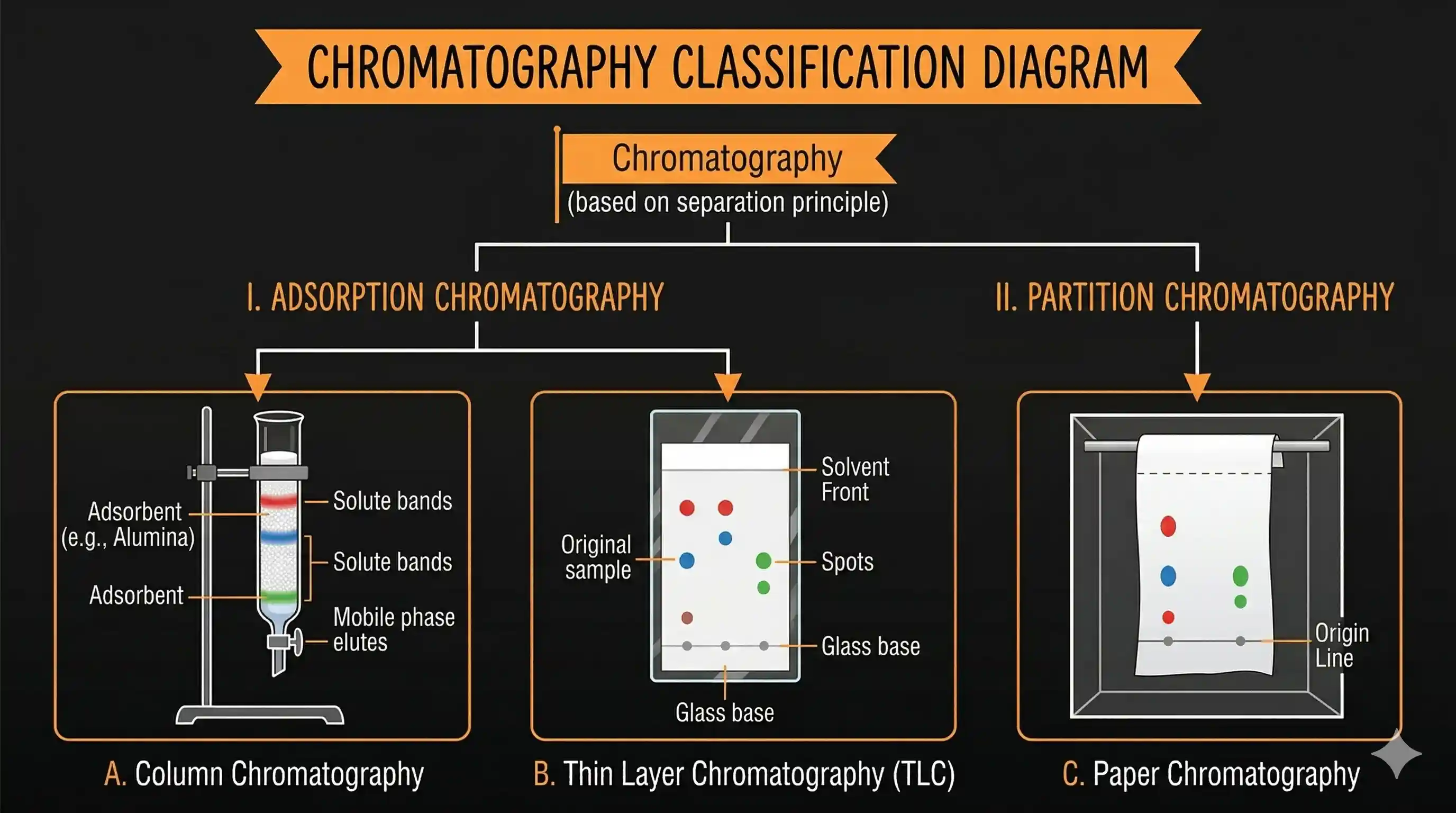

Two Principles: Adsorption vs Partition
All chromatographic techniques work by one of two principles:
(a) Adsorption Chromatography — stationary phase is a solid adsorbent (typically silica gel or alumina). Components stick to the surface to varying degrees. Techniques: Column Chromatography, Thin Layer Chromatography (TLC).
(b) Partition Chromatography — stationary phase is a liquid (e.g., water trapped inside paper fibres). Components distribute between the stationary liquid and the flowing solvent based on their relative solubility in each. Technique: Paper Chromatography.
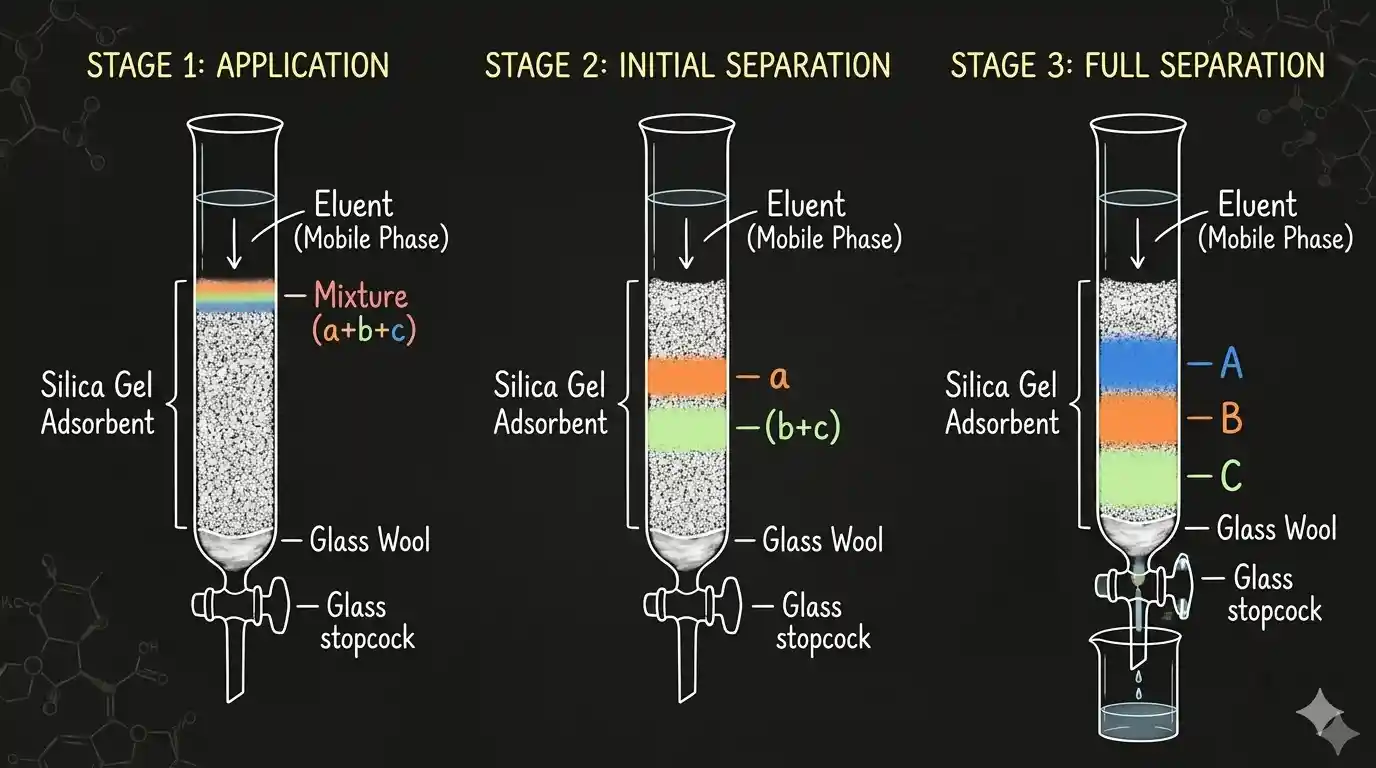
Column Chromatography
Column chromatography is the workhorse of the organic chemistry lab — used to purify compounds after a synthesis reaction.
Setup: A glass tube is packed tightly with powdered adsorbent (silica gel or alumina), fitted with a stopcock at the bottom. The impure mixture is dissolved in a small solvent and loaded onto the top of the column.
How it works:
- An eluant (solvent) is poured onto the top and flows slowly downward.
- It carries the components with it — weakly adsorbed ones move fast, strongly adsorbed ones move slow.
- Different components separate into bands at different positions down the column.
- Each fraction is collected as it exits from the bottom.
The most readily adsorbed substance stays near the top; others come down to varying distances in the column.
Thin Layer Chromatography (TLC)
TLC is fast and cheap — perfect for checking purity or monitoring a reaction.
Setup: A thin layer (about 0.2 mm thick) of silica gel or alumina is spread on a glass plate (the chromaplate). The sample is applied as a small spot about 2 cm from the bottom.
Running it:
- Place the plate upright in a closed jar with shallow eluant at the bottom.
- Solvent rises by capillary action, carrying the components upward at different speeds.
- Remove the plate before the solvent front reaches the top. Dry.
Detecting spots:
- Coloured compounds: spots are visible directly
- Colourless compounds: UV light (fluorescent compounds glow), iodine vapour (brown spots), or ninhydrin spray (for amino acids)
The Rf value characterises each compound:
is always between 0 and 1. High → weakly adsorbed (prefers mobile phase). Low → strongly adsorbed (sticks to adsorbent).
Paper Chromatography
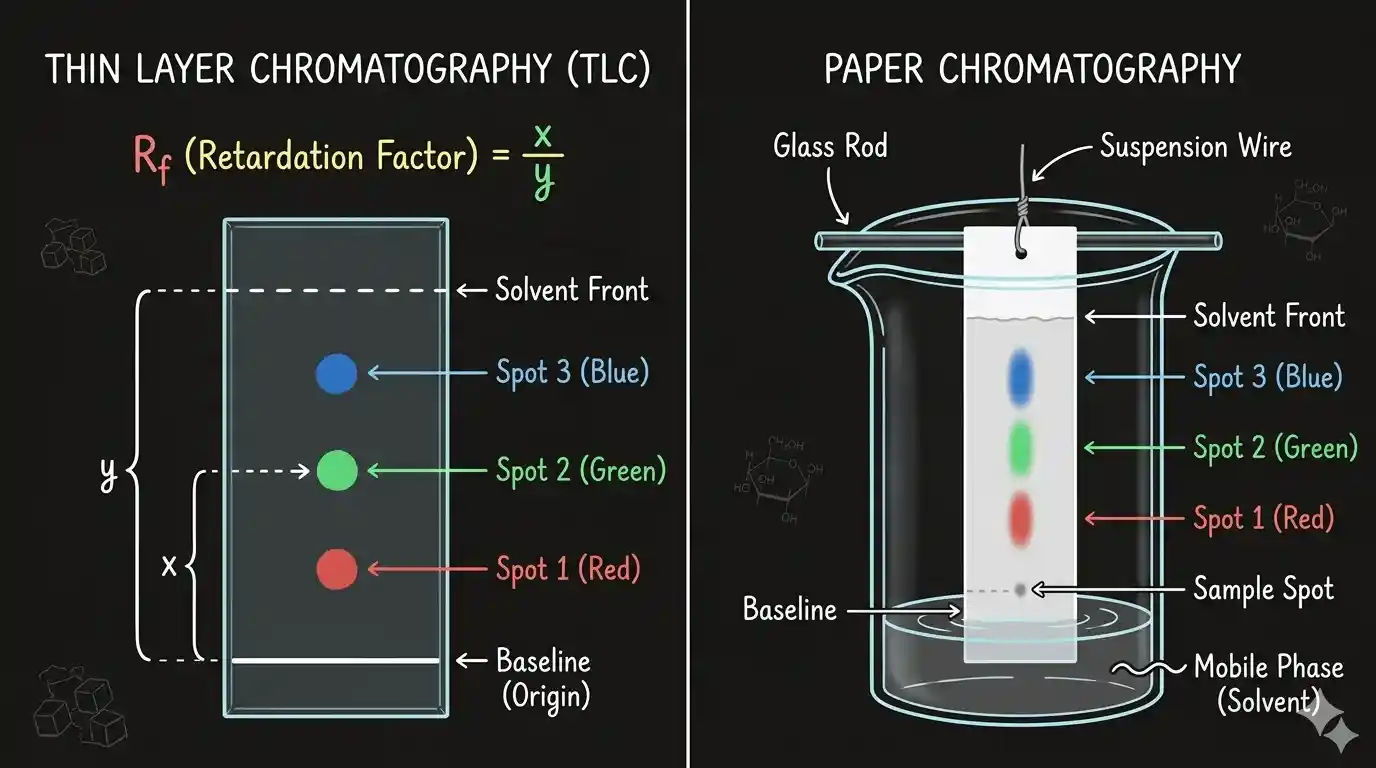
Paper chromatography is a type of partition chromatography. Special chromatography paper contains water trapped in its cellulose fibres — this water is the stationary phase.
How it works:
- Spot the mixture near the bottom of the paper strip.
- Suspend the strip in a closed container with the mobile phase solvent at the bottom (the spot must not touch the solvent directly).
- Solvent rises by capillary action, flows over the spot, and carries components upward.
- Each component partitions between the water (stationary) and the solvent (mobile) based on its relative solubility in each.
- The developed strip is the chromatogram — spots appear at different heights.
Colourless spots are detected using UV light or appropriate spray reagents, same as TLC. The value is used for identification here too.
Loading simulator…
Q1.A compound has an Rf value of 0.80 in TLC. This means:
A forensic analyst runs a TLC plate on ink recovered from a suspected forged document. To the naked eye, the ink matches the original perfectly. Under UV light on the developed plate, the original shows one spot — but the suspected forgery shows three. One extra dye component matches a synthetic compound not manufactured until 1998. The document is allegedly dated 1979. The forgery is proven without a confession.
In another lab, gas chromatography detects a single pesticide molecule among one billion water molecules in a fruit sample — below any health threshold, but still measurable. At a crime scene, unknown white powder is identified not by colour or smell but by its chromatographic fingerprint: the pattern of retention times is as unique as a barcode.
Chromatography is now used in anti-doping tests (WADA screens for hundreds of prohibited substances in a single urine sample), airport security (trace explosive detectors analyse swabs chromatographically in seconds), and food authentication (the olive oil in your bottle can be fingerprinted to confirm its geographic origin).
Mikhail Tswett invented the technique in 1903 to separate plant pigments. The same principle — differential migration based on affinity for stationary vs. mobile phase — now catches forgers, dopers, and poisoners. Chromatography is arguably the single most powerful analytical tool ever developed.
What is Chromatography?
‘Chromatography’ is an analytical technique commonly used for separating a mixture of chemical substances into its individual components, so that the individual components can be thoroughly analyzed. The name comes from the Greek chroma (colour) — because when it was first used, scientists were separating coloured plant pigments. Today, chromatography separates everything from amino acids to drugs to petroleum fractions.
The core idea is elegant. A mixture is applied onto a stationary phase (a solid or liquid that stays fixed). A mobile phase (a solvent or gas) is then allowed to flow over it. As it moves, it carries the components of the mixture along — but each component travels at a different speed, depending on how strongly it sticks to the stationary phase.
- Components that stick strongly to the stationary phase → move slowly, stay near the start
- Components that prefer the mobile phase → move quickly, travel far
Result: the mixture gets spread out in space, each component at a different position — separation is achieved.


Two Principles: Adsorption vs Partition
All chromatographic techniques work by one of two principles:
(a) Adsorption Chromatography — stationary phase is a solid adsorbent (typically silica gel or alumina). Components stick to the surface to varying degrees. Techniques: Column Chromatography, Thin Layer Chromatography (TLC).
(b) Partition Chromatography — stationary phase is a liquid (e.g., water trapped inside paper fibres). Components distribute between the stationary liquid and the flowing solvent based on their relative solubility in each. Technique: Paper Chromatography.
Column Chromatography
Column chromatography is the workhorse of the organic chemistry lab — used to purify compounds after a synthesis reaction.
Setup: A glass tube is packed tightly with powdered adsorbent (silica gel or alumina), fitted with a stopcock at the bottom. The impure mixture is dissolved in a small solvent and loaded onto the top of the column.
How it works:
- An eluant (solvent) is poured onto the top and flows slowly downward.
- It carries the components with it — weakly adsorbed ones move fast, strongly adsorbed ones move slow.
- Different components separate into bands at different positions down the column.
- Each fraction is collected as it exits from the bottom.
The most readily adsorbed substance stays near the top; others come down to varying distances in the column.

Thin Layer Chromatography (TLC)
TLC is fast and cheap — perfect for checking purity or monitoring a reaction.
Setup: A thin layer (about 0.2 mm thick) of silica gel or alumina is spread on a glass plate (the chromaplate). The sample is applied as a small spot about 2 cm from the bottom.
Running it:
- Place the plate upright in a closed jar with shallow eluant at the bottom.
- Solvent rises by capillary action, carrying the components upward at different speeds.
- Remove the plate before the solvent front reaches the top. Dry.
Detecting spots:
- Coloured compounds: spots are visible directly
- Colourless compounds: UV light (fluorescent compounds glow), iodine vapour (brown spots), or ninhydrin spray (for amino acids)
The Rf value characterises each compound:
is always between 0 and 1. High → weakly adsorbed (prefers mobile phase). Low → strongly adsorbed (sticks to adsorbent).
Paper Chromatography

Paper chromatography is a type of partition chromatography. Special chromatography paper contains water trapped in its cellulose fibres — this water is the stationary phase.
How it works:
- Spot the mixture near the bottom of the paper strip.
- Suspend the strip in a closed container with the mobile phase solvent at the bottom (the spot must not touch the solvent directly).
- Solvent rises by capillary action, flows over the spot, and carries components upward.
- Each component partitions between the water (stationary) and the solvent (mobile) based on its relative solubility in each.
- The developed strip is the chromatogram — spots appear at different heights.
Colourless spots are detected using UV light or appropriate spray reagents, same as TLC. The value is used for identification here too.
Loading simulator…
Q1.A compound has an Rf value of 0.80 in TLC. This means: