Distillation
Separating liquids by harnessing the difference in their boiling points
When you heat a mixture of liquids, not all of them boil at the same time. Each liquid has its own boiling point — and distillation uses this difference to pull them apart.
Distillation is used in two main situations:
- To separate a volatile liquid from a non-volatile impurity (e.g., getting pure water from water containing dissolved salts)
- To separate two or more liquids whose boiling points are different enough to separate cleanly
Simple Distillation
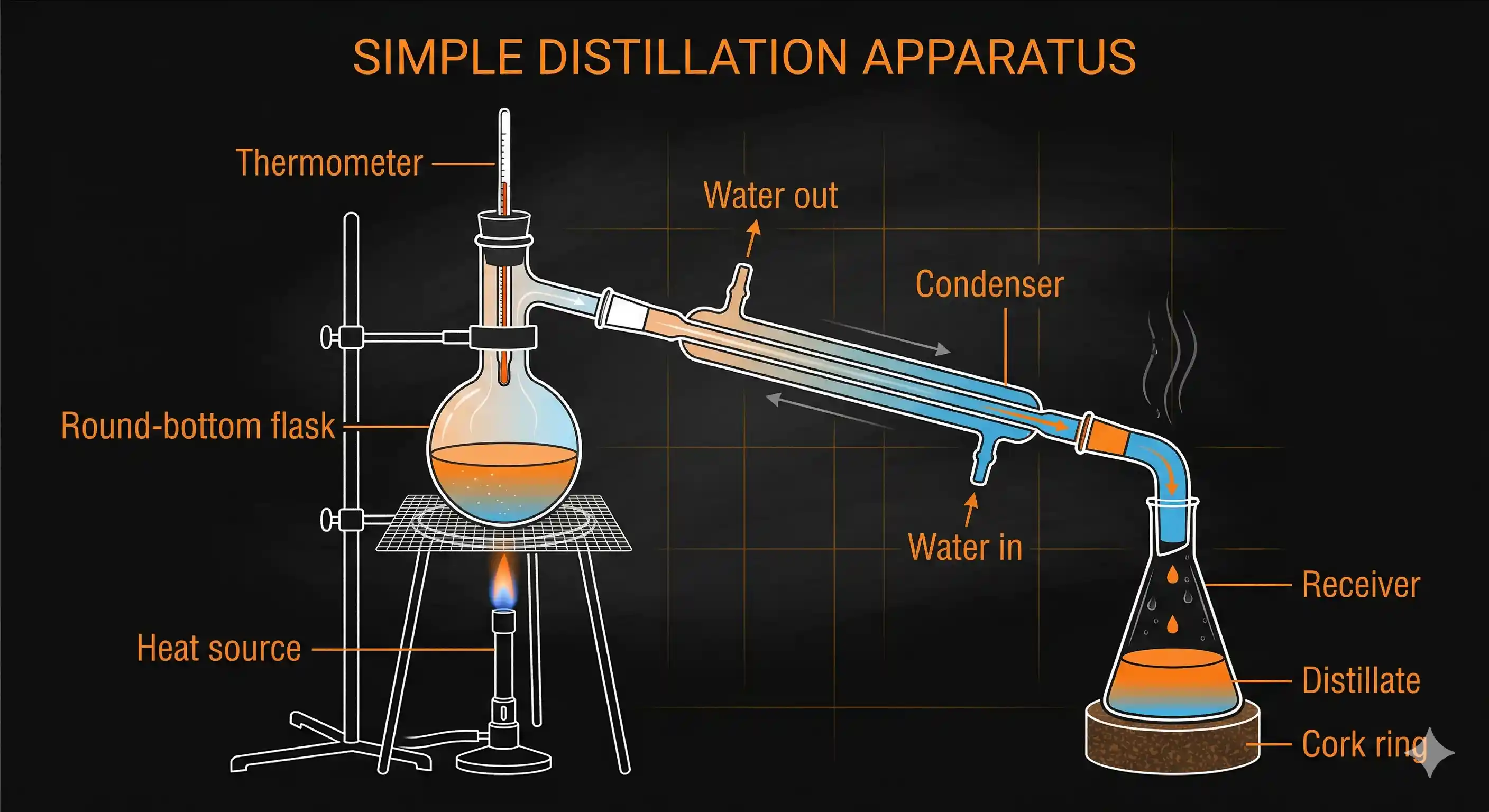
Simple distillation is the most straightforward version. Here's what happens:
- The liquid mixture is taken in a round-bottom flask and heated carefully.
- The component with the lower boiling point vaporises first.
- The vapour travels up into the condenser — a glass tube surrounded by flowing cold water — where it cools down and turns back to liquid.
- The condensed liquid (called the distillate) drips into a collecting flask (receiver).
- As heating continues, the temperature in the flask rises, and the higher boiling component eventually vaporises and is collected separately.
A thermometer is placed at the opening of the condenser to monitor the temperature. The boiling point of the distillate tells you which component is being collected at any moment.
Real-World Applications
Simple distillation has been practised for millennia — and remains indispensable in modern industry:
-
Traditional Indian Perfumery (Kannauj, UP) — The "Perfume Capital of India" has produced attar (natural perfume) since Mughal times using handcrafted copper deg-bhapka stills. Flowers (rose, jasmine, kewra) are loaded into the copper deg with water. Steam carries the volatile aroma molecules through copper pipes into a bhapka (receiver) submerged in cold water — simple distillation unchanged from its 4th-century Taxila origins.
-
Seawater Desalination — Cities like Chennai and Dubai use large-scale Multi-Stage Flash (MSF) distillation plants. Seawater is heated in a series of chambers, each at progressively lower pressure, flashing to steam. The steam condenses on cold surfaces as pure water. The Nemmeli Desalination Plant (Tamil Nadu) produces 100 MLD of drinking water from the Bay of Bengal purely by distillation.
-
Water-for-Injection (WFI) in Pharma — Medicines meant to be injected intravenously require ultra-pure water — pure enough that even trace ions would be dangerous. Pharmaceutical companies distil water multiple times using stainless steel multi-effect distillers. The final product must have conductivity below 1.3 μS/cm.
-
Automobile Batteries and Radiators — Only distilled water is used in lead-acid batteries and radiator coolant systems. Tap water minerals would corrode the electrodes or block coolant channels over time.

The Deg-Bhapka system is a traditional, centuries-old Indian hydro-distillation technique primarily used in Kannauj, Uttar Pradesh—known as the "Perfume Capital of India"—to produce natural attars (alcohol-free perfumes) and essential oils. This artisanal method, which dates back over a thousand years to the Mughal era, involves distilling botanicals (flowers, herbs, roots) in copper pots over a wood fire.

Q1.Simple distillation is most effective when separating two liquids whose boiling points differ by:
When you heat a mixture of liquids, not all of them boil at the same time. Each liquid has its own boiling point — and distillation uses this difference to pull them apart.
Distillation is used in two main situations:
- To separate a volatile liquid from a non-volatile impurity (e.g., getting pure water from water containing dissolved salts)
- To separate two or more liquids whose boiling points are different enough to separate cleanly
Simple Distillation
Simple distillation is the most straightforward version. Here's what happens:
- The liquid mixture is taken in a round-bottom flask and heated carefully.
- The component with the lower boiling point vaporises first.
- The vapour travels up into the condenser — a glass tube surrounded by flowing cold water — where it cools down and turns back to liquid.
- The condensed liquid (called the distillate) drips into a collecting flask (receiver).
- As heating continues, the temperature in the flask rises, and the higher boiling component eventually vaporises and is collected separately.
A thermometer is placed at the opening of the condenser to monitor the temperature. The boiling point of the distillate tells you which component is being collected at any moment.

Real-World Applications
Simple distillation has been practised for millennia — and remains indispensable in modern industry:
-
Traditional Indian Perfumery (Kannauj, UP) — The "Perfume Capital of India" has produced attar (natural perfume) since Mughal times using handcrafted copper deg-bhapka stills. Flowers (rose, jasmine, kewra) are loaded into the copper deg with water. Steam carries the volatile aroma molecules through copper pipes into a bhapka (receiver) submerged in cold water — simple distillation unchanged from its 4th-century Taxila origins.
-
Seawater Desalination — Cities like Chennai and Dubai use large-scale Multi-Stage Flash (MSF) distillation plants. Seawater is heated in a series of chambers, each at progressively lower pressure, flashing to steam. The steam condenses on cold surfaces as pure water. The Nemmeli Desalination Plant (Tamil Nadu) produces 100 MLD of drinking water from the Bay of Bengal purely by distillation.
-
Water-for-Injection (WFI) in Pharma — Medicines meant to be injected intravenously require ultra-pure water — pure enough that even trace ions would be dangerous. Pharmaceutical companies distil water multiple times using stainless steel multi-effect distillers. The final product must have conductivity below 1.3 μS/cm.
-
Automobile Batteries and Radiators — Only distilled water is used in lead-acid batteries and radiator coolant systems. Tap water minerals would corrode the electrodes or block coolant channels over time.

The Deg-Bhapka system is a traditional, centuries-old Indian hydro-distillation technique primarily used in Kannauj, Uttar Pradesh—known as the "Perfume Capital of India"—to produce natural attars (alcohol-free perfumes) and essential oils. This artisanal method, which dates back over a thousand years to the Mughal era, involves distilling botanicals (flowers, herbs, roots) in copper pots over a wood fire.

Q1.Simple distillation is most effective when separating two liquids whose boiling points differ by: