Estimation of Carbon and Hydrogen
Burning a compound completely and weighing the products to find the % of C and H
Qualitative analysis told you which elements are present. Now quantitative analysis answers the harder question: how much of each element? The percentage composition of carbon and hydrogen is found by a beautifully simple principle — burn the compound completely and weigh what comes out.
The Principle
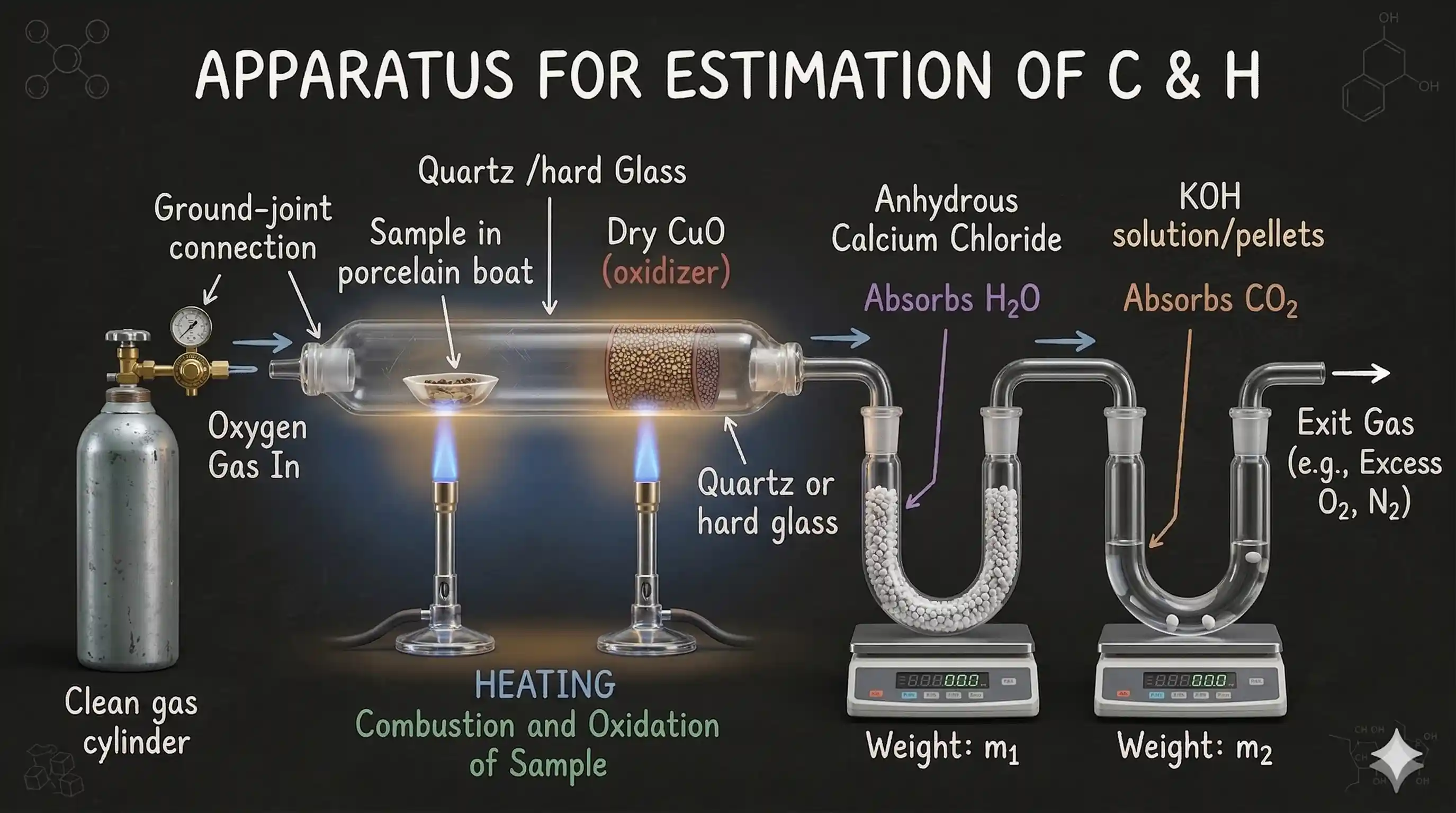
A known mass of the organic compound is burnt in excess oxygen in the presence of copper(II) oxide (to ensure complete combustion). All the carbon in the compound is converted to and all the hydrogen to :
The products are passed through two absorption chambers:
- Anhydrous calcium chloride ( ) — absorbs
- Potassium hydroxide ( ) — absorbs
The increase in mass of each absorber gives the mass of ( g) and ( g) produced from a known mass of compound ( g).
The Formulas
Since all the carbon in the compound ends up in :
- Molar mass of = 44 g/mol; contains 12 g of C per mole
- So 44 g of contains 12 g of C
Since all hydrogen ends up in :
- Molar mass of = 18 g/mol; contains 2 g of H per mole
- So 18 g of contains 2 g of H
Where = mass of organic compound, = mass of formed, = mass of formed.
Problem
On complete combustion, 0.246 g of an organic compound gave 0.198 g of carbon dioxide and 0.1014 g of water. Determine the percentage composition of carbon and hydrogen in the compound.
Why CaCl₂ comes before KOH?
absorbs water, absorbs . The order matters: CaCl₂ must come first. If KOH came first, it would absorb both and water — making it impossible to calculate each separately. The calcium chloride tube is weighed before and after to find ; the KOH tube is weighed before and after to find .
Q1.In the estimation of carbon and hydrogen, calcium chloride is placed before the KOH absorber. This is because:
Qualitative analysis told you which elements are present. Now quantitative analysis answers the harder question: how much of each element? The percentage composition of carbon and hydrogen is found by a beautifully simple principle — burn the compound completely and weigh what comes out.
The Principle
A known mass of the organic compound is burnt in excess oxygen in the presence of copper(II) oxide (to ensure complete combustion). All the carbon in the compound is converted to and all the hydrogen to :
The products are passed through two absorption chambers:
- Anhydrous calcium chloride ( ) — absorbs
- Potassium hydroxide ( ) — absorbs
The increase in mass of each absorber gives the mass of ( g) and ( g) produced from a known mass of compound ( g).

The Formulas
Since all the carbon in the compound ends up in :
- Molar mass of = 44 g/mol; contains 12 g of C per mole
- So 44 g of contains 12 g of C
Since all hydrogen ends up in :
- Molar mass of = 18 g/mol; contains 2 g of H per mole
- So 18 g of contains 2 g of H
Where = mass of organic compound, = mass of formed, = mass of formed.
Problem
On complete combustion, 0.246 g of an organic compound gave 0.198 g of carbon dioxide and 0.1014 g of water. Determine the percentage composition of carbon and hydrogen in the compound.
Q1.In the estimation of carbon and hydrogen, calcium chloride is placed before the KOH absorber. This is because: