Fractional Distillation
What to do when two liquids refuse to boil at very different temperatures
Simple distillation hits a wall when the two liquids you want to separate have similar boiling points. Their vapours form together, condense together, and end up mixed in the receiver — no separation achieved.
For example, ethanol boils at 78°C and water at 100°C. That's a 22°C difference — too close for simple distillation to work cleanly. This is where fractional distillation comes in.
The Fractionating Column: A Series of Mini-Distillations
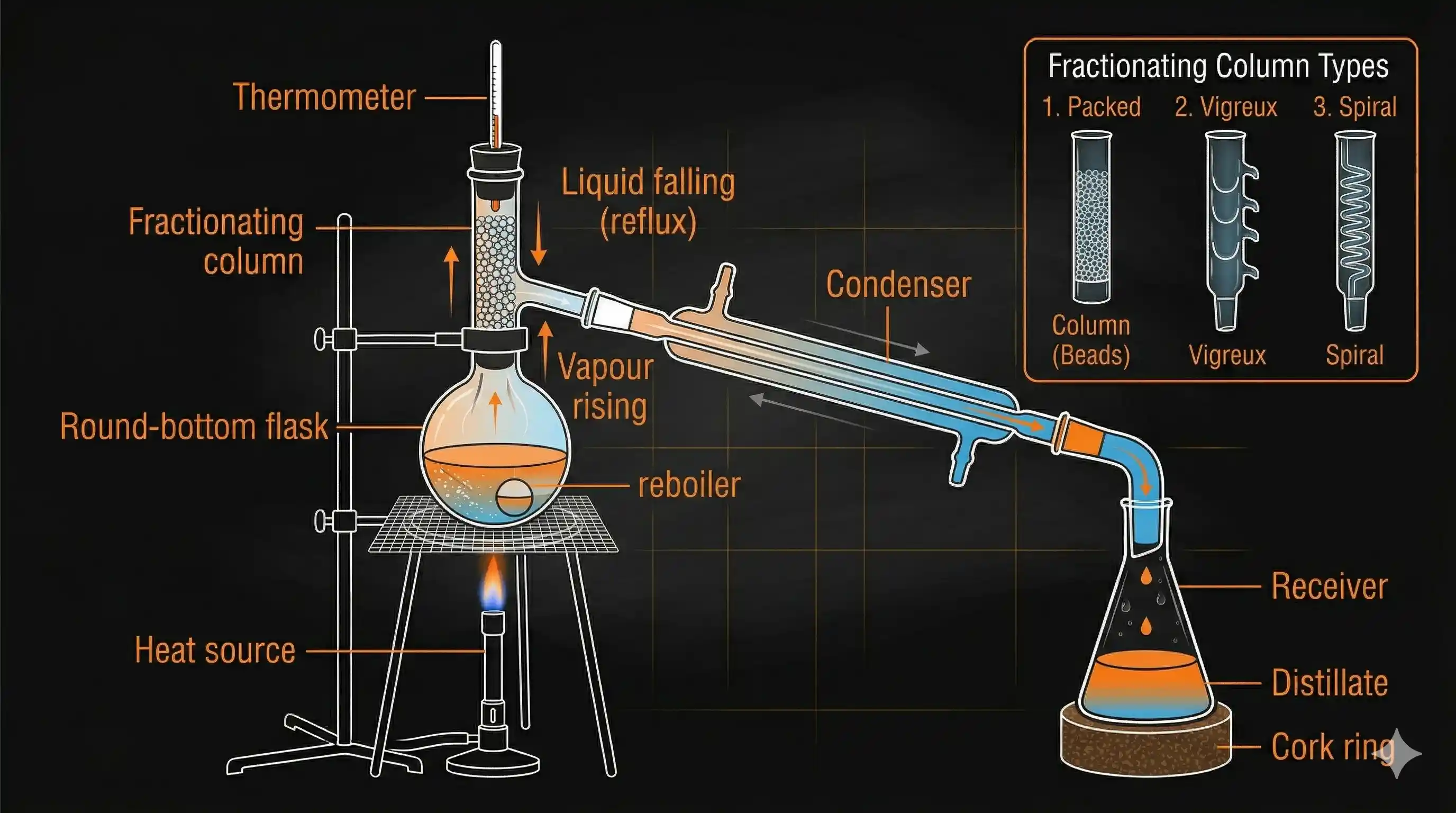
A fractionating column is fitted onto the mouth of the round-bottom flask between the flask and the condenser. It's packed with glass beads, rings, or has a special internal structure — the goal is to provide many surfaces inside the column.
Here's what happens inside:
- Vapour rises from the boiling mixture and enters the fractionating column.
- As vapour rises, it meets cooler surfaces and partially condenses.
- The condensed liquid trickles downward and gets reheated by the rising vapour.
- The more volatile component (lower boiling point) re-vaporises more easily and keeps climbing up.
- The less volatile component (higher boiling point) tends to condense and fall back down.
This cycle of condensation → reheating → re-vaporisation repeats many times as vapour travels up the column. By the time vapour reaches the top, it is almost entirely the lower-boiling component — pure enough to be collected separately.
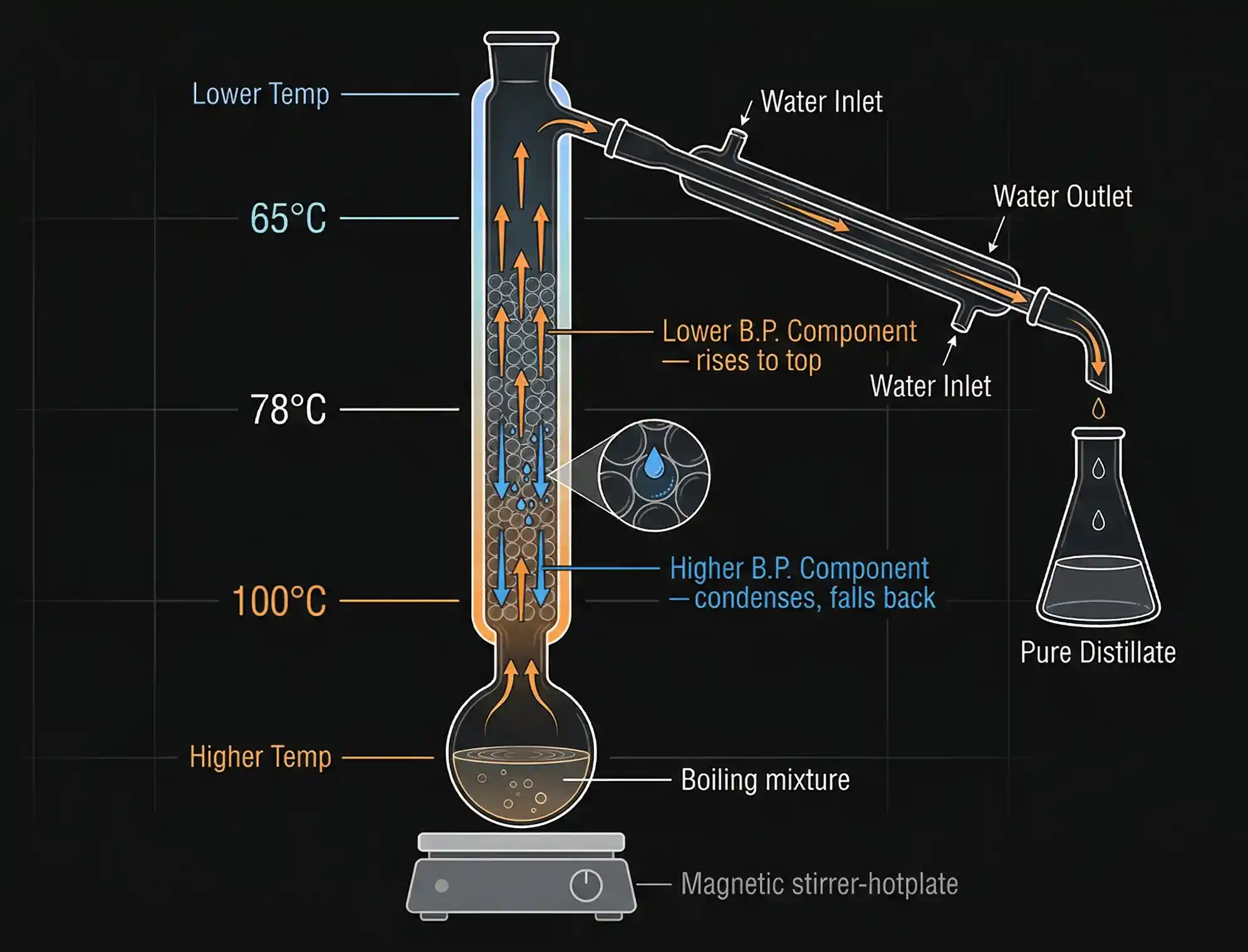
Theoretical Plates: Measuring Column Efficiency
Each complete cycle of condensation + re-vaporisation inside the column is called a theoretical plate. The more theoretical plates a column has, the better the separation.
Think of each plate as one mini-distillation. A column with 10 theoretical plates performs 10 distillations in one shot. Industrial fractionating columns used in petroleum refineries can have hundreds of theoretical plates, allowing them to separate dozens of different fractions from crude oil in a single continuous operation.
Real-World Application: Crude Oil Refining

India's refinery landscape: The Jamnagar Refinery Complex (Reliance Industries) in Gujarat is the world's largest integrated refinery — processing 1.24 million barrels of crude oil per day across two refinery units. Its fractional distillation columns produce fuel for over 300 million vehicles and feedstock for plastics, textiles, and pharmaceuticals. Every fraction — from LPG cooked in Indian kitchens to the bitumen on every national highway — is separated purely on the basis of boiling point differences, through exactly the column you just studied.
The theoretical plates concept you learned translates directly to refinery design: each metre of packing in an industrial column provides a set number of theoretical plates. Refinery engineers use this to calculate minimum column height for a desired separation — sometimes requiring columns taller than a 20-storey building.
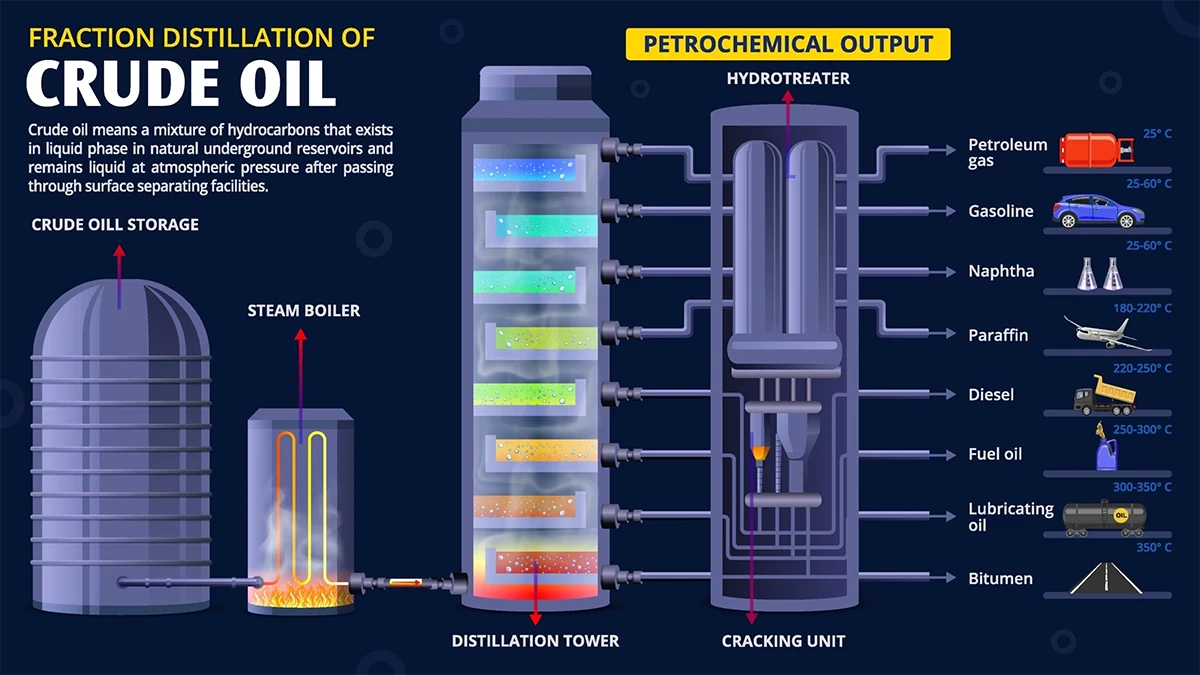
One of the most important industrial applications of fractional distillation is the refining of crude oil (petroleum).
Crude oil is a complex mixture of hundreds of hydrocarbon compounds, each with a different boiling point. When crude oil is fed into a massive industrial fractionating column, lighter fractions (lower boiling points) rise higher in the column and are collected at the top; heavier fractions are collected lower down or remain as residue.
Loading simulator…
Q1.A fractionating column improves separation by:
Simple distillation hits a wall when the two liquids you want to separate have similar boiling points. Their vapours form together, condense together, and end up mixed in the receiver — no separation achieved.
For example, ethanol boils at 78°C and water at 100°C. That's a 22°C difference — too close for simple distillation to work cleanly. This is where fractional distillation comes in.
The Fractionating Column: A Series of Mini-Distillations
A fractionating column is fitted onto the mouth of the round-bottom flask between the flask and the condenser. It's packed with glass beads, rings, or has a special internal structure — the goal is to provide many surfaces inside the column.
Here's what happens inside:
- Vapour rises from the boiling mixture and enters the fractionating column.
- As vapour rises, it meets cooler surfaces and partially condenses.
- The condensed liquid trickles downward and gets reheated by the rising vapour.
- The more volatile component (lower boiling point) re-vaporises more easily and keeps climbing up.
- The less volatile component (higher boiling point) tends to condense and fall back down.
This cycle of condensation → reheating → re-vaporisation repeats many times as vapour travels up the column. By the time vapour reaches the top, it is almost entirely the lower-boiling component — pure enough to be collected separately.

Theoretical Plates: Measuring Column Efficiency
Each complete cycle of condensation + re-vaporisation inside the column is called a theoretical plate. The more theoretical plates a column has, the better the separation.
Think of each plate as one mini-distillation. A column with 10 theoretical plates performs 10 distillations in one shot. Industrial fractionating columns used in petroleum refineries can have hundreds of theoretical plates, allowing them to separate dozens of different fractions from crude oil in a single continuous operation.

Real-World Application: Crude Oil Refining


India's refinery landscape: The Jamnagar Refinery Complex (Reliance Industries) in Gujarat is the world's largest integrated refinery — processing 1.24 million barrels of crude oil per day across two refinery units. Its fractional distillation columns produce fuel for over 300 million vehicles and feedstock for plastics, textiles, and pharmaceuticals. Every fraction — from LPG cooked in Indian kitchens to the bitumen on every national highway — is separated purely on the basis of boiling point differences, through exactly the column you just studied.
The theoretical plates concept you learned translates directly to refinery design: each metre of packing in an industrial column provides a set number of theoretical plates. Refinery engineers use this to calculate minimum column height for a desired separation — sometimes requiring columns taller than a 20-storey building.
One of the most important industrial applications of fractional distillation is the refining of crude oil (petroleum).
Crude oil is a complex mixture of hundreds of hydrocarbon compounds, each with a different boiling point. When crude oil is fed into a massive industrial fractionating column, lighter fractions (lower boiling points) rise higher in the column and are collected at the top; heavier fractions are collected lower down or remain as residue.
Loading simulator…
Q1.A fractionating column improves separation by: