Estimation of Nitrogen
Two methods — Dumas and Kjeldahl — each used in different situations
Proteins are the machinery of life — enzymes, antibodies, haemoglobin. All proteins contain nitrogen. The Kjeldahl method, developed in 1883, is still used today in food labs worldwide to measure the protein content of milk, flour, and meat — because protein % ≈ nitrogen % × 6.25. That's how your nutrition label gets its protein number.
There are two methods for estimating nitrogen in an organic compound:
- Dumas method — converts all nitrogen to gas and measures its volume
- Kjeldahl's method — converts nitrogen to and measures it by back-titration
Each has its own conditions and limitations.
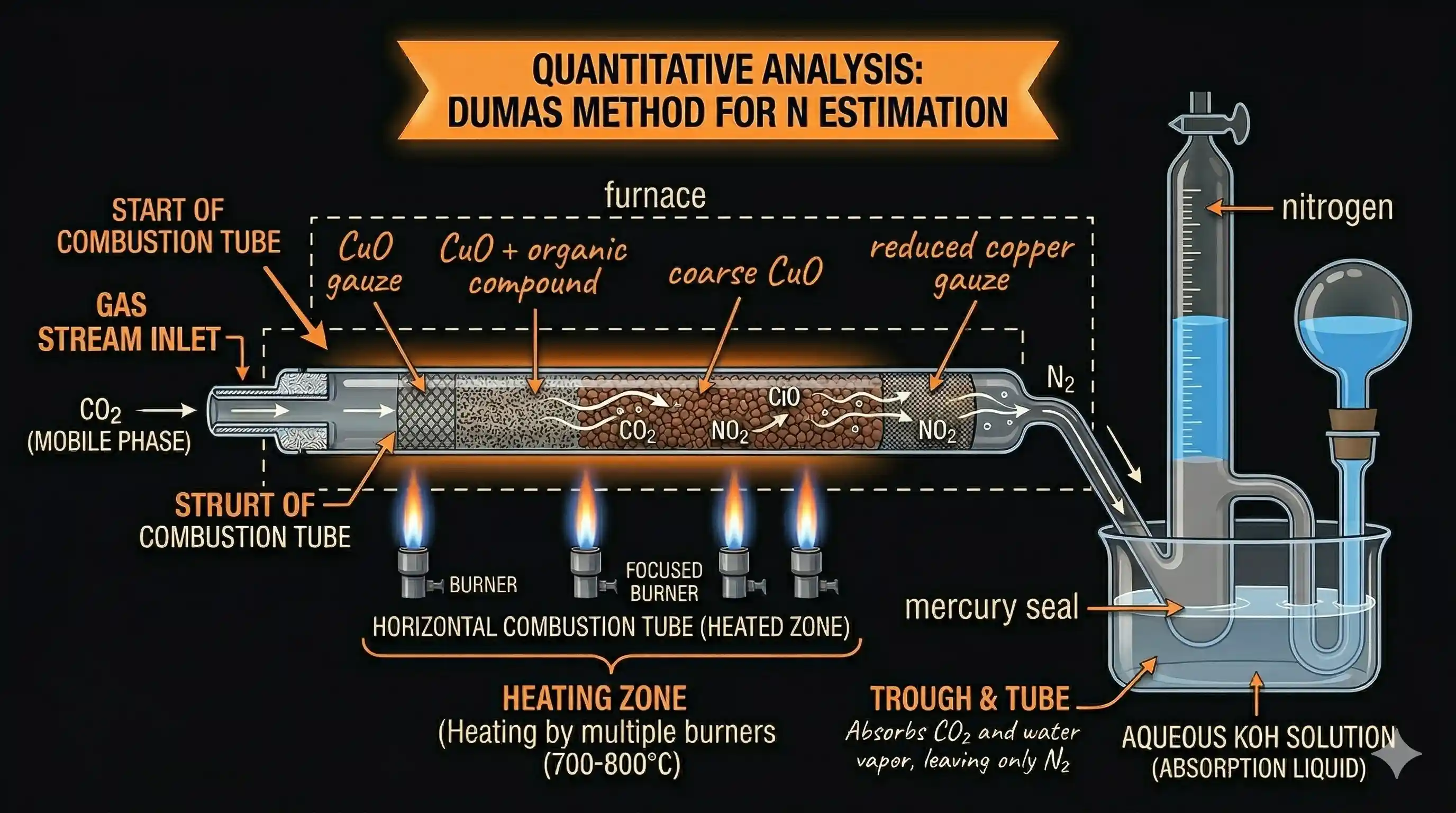
Dumas Method
Principle: The nitrogen-containing compound is heated strongly with copper oxide ( ) in an atmosphere of . This converts all nitrogen in the compound to free nitrogen gas:
Any nitrogen oxides ( ) that form are reduced back to by passing over heated copper gauze.
The gaseous mixture ( + ) is collected over aqueous KOH solution. KOH absorbs the , and the remaining gas is pure collected in a graduated tube.
Calculating percentage of nitrogen:
Let mass of compound = g, volume of collected = mL at temperature K and pressure mm Hg.
First convert to STP:
Note: (since gas is collected over water)
Since 22400 mL of at STP weighs 28 g:
Problem
In Dumas' method for estimation of nitrogen, 0.3 g of an organic compound gave 50 mL of nitrogen collected at 300 K temperature and 715 mm pressure. Calculate the percentage of nitrogen in the compound. (Aqueous tension at 300 K = 15 mm)
Kjeldahl's Method
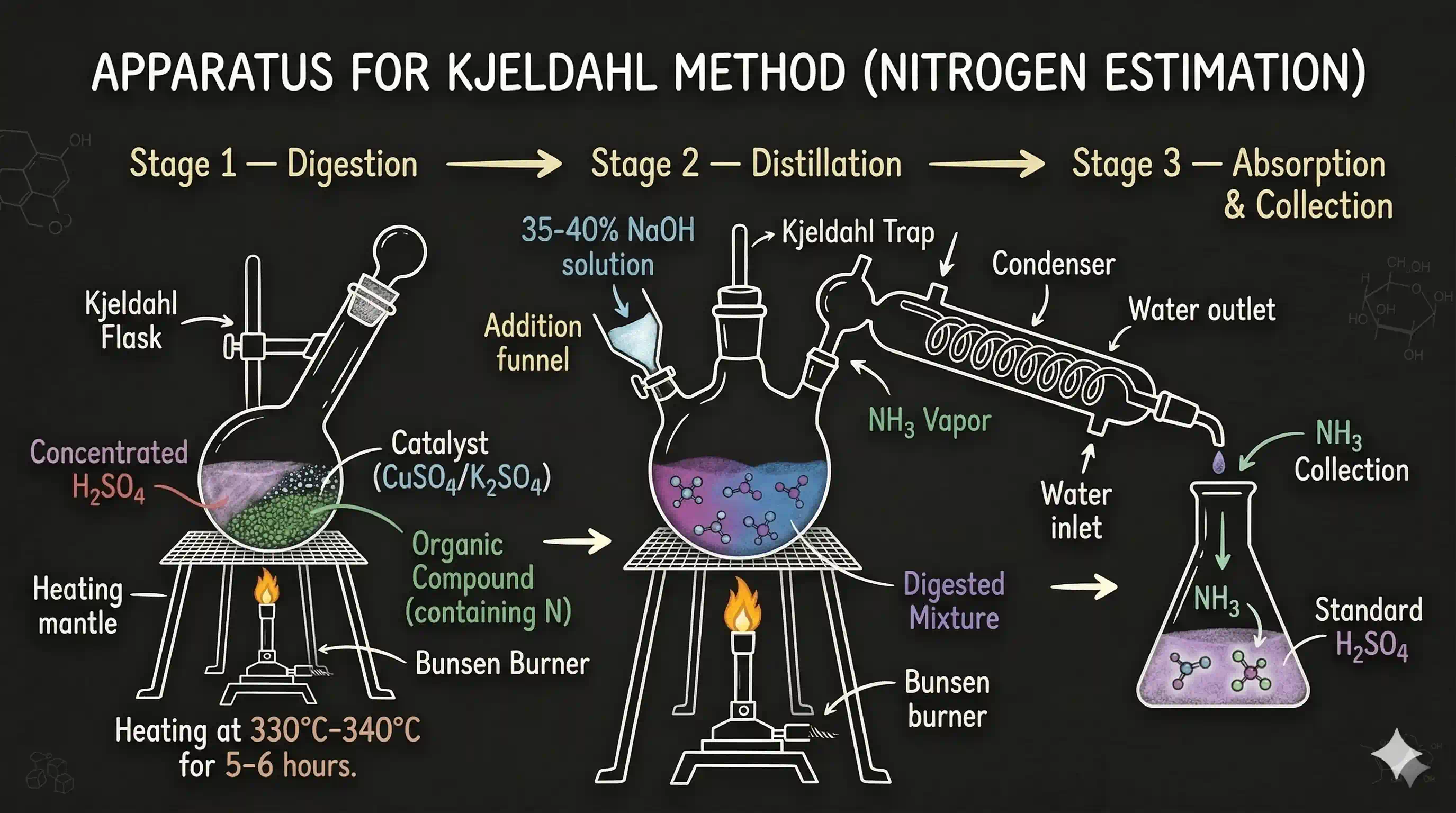
Principle: The compound is heated with concentrated sulphuric acid ( ). All nitrogen in the compound converts to ammonium sulphate:
The mixture is then made alkaline by adding excess NaOH, which liberates ammonia:
The gas is distilled over and absorbed in a known excess volume of standard sulphuric acid ( mL of molarity ):
The unreacted (excess) acid is back-titrated with standard NaOH ( mL of molarity ).
Formula:
where = mass of compound in grams, = mL of taken, = mL of NaOH used in back-titration.
Limitation: Kjeldahl's method cannot be used for compounds where nitrogen is in a nitro group ( ), azo group ( ), or in a ring (like pyridine). In these cases, the nitrogen does not convert to ammonium sulphate under the reaction conditions.
Problem
During estimation of nitrogen present in an organic compound by Kjeldahl's method, the ammonia evolved from 0.5 g of the compound in Kjeldahl's estimation neutralised 10 mL of 1 M H₂SO₄. Find out the percentage of nitrogen in the compound.
Dumas vs Kjeldahl
Dumas Method
- Compound heated with CuO in CO₂ atmosphere
- N₂ gas collected over KOH solution
- Volume of N₂ measured and converted to STP
- Works for all nitrogen-containing compounds
- More complex apparatus; used in research
Kjeldahl's Method
- Compound digested with conc. H₂SO₄
- N → (NH₄)₂SO₄ → NH₃ → absorbed in acid
- Excess acid back-titrated with NaOH
- Does NOT work for —NO₂, —N=N—, or ring N
- Simpler; widely used in food/agriculture labs
Dumas Method
- Compound heated with CuO in CO₂ atmosphere
- N₂ gas collected over KOH solution
- Volume of N₂ measured and converted to STP
- Works for all nitrogen-containing compounds
- More complex apparatus; used in research
Kjeldahl's Method
- Compound digested with conc. H₂SO₄
- N → (NH₄)₂SO₄ → NH₃ → absorbed in acid
- Excess acid back-titrated with NaOH
- Does NOT work for —NO₂, —N=N—, or ring N
- Simpler; widely used in food/agriculture labs
Q1.Kjeldahl's method for nitrogen estimation cannot be applied to which type of compound?
Proteins are the machinery of life — enzymes, antibodies, haemoglobin. All proteins contain nitrogen. The Kjeldahl method, developed in 1883, is still used today in food labs worldwide to measure the protein content of milk, flour, and meat — because protein % ≈ nitrogen % × 6.25. That's how your nutrition label gets its protein number.
There are two methods for estimating nitrogen in an organic compound:
- Dumas method — converts all nitrogen to gas and measures its volume
- Kjeldahl's method — converts nitrogen to and measures it by back-titration
Each has its own conditions and limitations.
Dumas Method
Principle: The nitrogen-containing compound is heated strongly with copper oxide ( ) in an atmosphere of . This converts all nitrogen in the compound to free nitrogen gas:
Any nitrogen oxides ( ) that form are reduced back to by passing over heated copper gauze.
The gaseous mixture ( + ) is collected over aqueous KOH solution. KOH absorbs the , and the remaining gas is pure collected in a graduated tube.
Calculating percentage of nitrogen:
Let mass of compound = g, volume of collected = mL at temperature K and pressure mm Hg.
First convert to STP:
Note: (since gas is collected over water)
Since 22400 mL of at STP weighs 28 g:

Problem
In Dumas' method for estimation of nitrogen, 0.3 g of an organic compound gave 50 mL of nitrogen collected at 300 K temperature and 715 mm pressure. Calculate the percentage of nitrogen in the compound. (Aqueous tension at 300 K = 15 mm)
Kjeldahl's Method

Principle: The compound is heated with concentrated sulphuric acid ( ). All nitrogen in the compound converts to ammonium sulphate:
The mixture is then made alkaline by adding excess NaOH, which liberates ammonia:
The gas is distilled over and absorbed in a known excess volume of standard sulphuric acid ( mL of molarity ):
The unreacted (excess) acid is back-titrated with standard NaOH ( mL of molarity ).
Formula:
where = mass of compound in grams, = mL of taken, = mL of NaOH used in back-titration.
Limitation: Kjeldahl's method cannot be used for compounds where nitrogen is in a nitro group ( ), azo group ( ), or in a ring (like pyridine). In these cases, the nitrogen does not convert to ammonium sulphate under the reaction conditions.
Problem
During estimation of nitrogen present in an organic compound by Kjeldahl's method, the ammonia evolved from 0.5 g of the compound in Kjeldahl's estimation neutralised 10 mL of 1 M H₂SO₄. Find out the percentage of nitrogen in the compound.
Dumas vs Kjeldahl
Dumas Method
- Compound heated with CuO in CO₂ atmosphere
- N₂ gas collected over KOH solution
- Volume of N₂ measured and converted to STP
- Works for all nitrogen-containing compounds
- More complex apparatus; used in research
Kjeldahl's Method
- Compound digested with conc. H₂SO₄
- N → (NH₄)₂SO₄ → NH₃ → absorbed in acid
- Excess acid back-titrated with NaOH
- Does NOT work for —NO₂, —N=N—, or ring N
- Simpler; widely used in food/agriculture labs
Dumas Method
- Compound heated with CuO in CO₂ atmosphere
- N₂ gas collected over KOH solution
- Volume of N₂ measured and converted to STP
- Works for all nitrogen-containing compounds
- More complex apparatus; used in research
Kjeldahl's Method
- Compound digested with conc. H₂SO₄
- N → (NH₄)₂SO₄ → NH₃ → absorbed in acid
- Excess acid back-titrated with NaOH
- Does NOT work for —NO₂, —N=N—, or ring N
- Simpler; widely used in food/agriculture labs
Q1.Kjeldahl's method for nitrogen estimation cannot be applied to which type of compound?