Rutherford's Nuclear Model of the Atom
One experiment that fired artillery shells at tissue paper — and changed physics forever
Ernest Rutherford designed the gold foil experiment in 1909 not to discover a new structure of the atom — but to confirm his supervisor J.J. Thomson's plum pudding model. He expected to prove that atoms were soft, featureless spheres of spread-out charge that α-particles would glide through. What he found instead was so shocking that it rewrote the entire picture of matter — and gave us the nuclear atom we still use today.
Radioactivity: The Key That Unlocked the Atom
To understand why Rutherford's experiment worked, you first need to understand what he had in his hands: a gun that fired particles from inside atoms.
The story begins in 1895 when Wilhelm Röntgen discovered X-rays — invisible radiation produced when fast electrons smash into dense metal targets. These rays passed through skin but not bone, could not be deflected by electric or magnetic fields, and had wavelengths around 0.1 nm. Nobody knew exactly what they were, which is why Röntgen named them X-rays.
One year later, Henri Becquerel (1852–1908) stumbled upon something even stranger. Certain heavy elements emitted radiation spontaneously, without any external energy input. He called this phenomenon radioactivity and the elements responsible radioactive elements.
Becquerel's discovery was taken up by Marie Curie, Pierre Curie, Rutherford, and Frederick Soddy. It was Rutherford who sorted the radiation into three distinct types and named them:
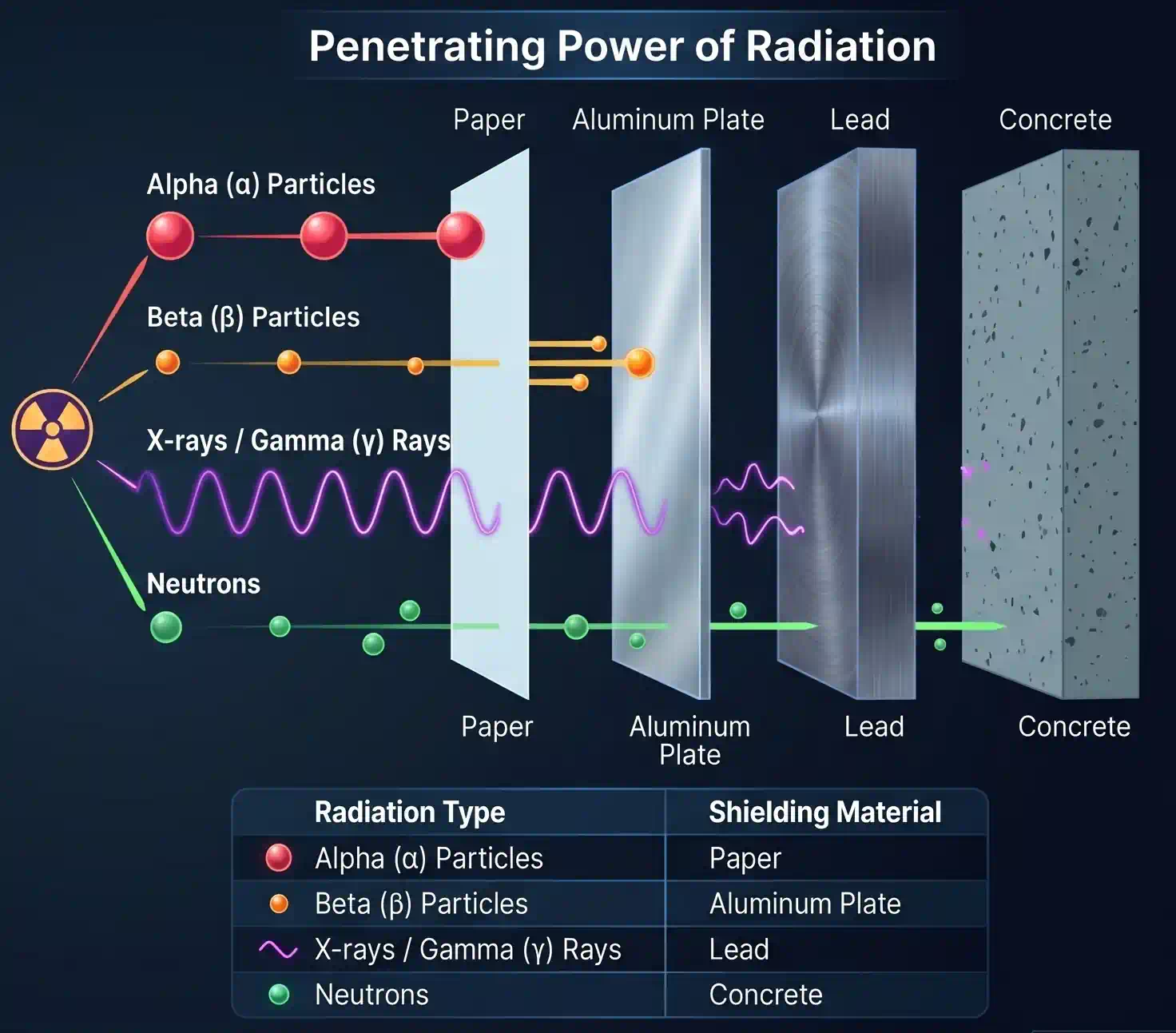
Properties of the three types of radiation from radioactive elements
| Radiation | Nature | Charge | Mass | Penetrating Power | Deflection by Fields |
|---|---|---|---|---|---|
| α (alpha) | Helium nuclei | +2 | 4 u | Lowest — stopped by a sheet of paper | Yes — towards negative plate |
| β (beta) | Fast electrons | −1 | ~0 | ~100× more than α — stopped by Al foil | Yes — towards positive plate |
| γ (gamma) | High-energy EM radiation | 0 | 0 | ~1000× more than α — needs thick lead/concrete | No — not deflected |
The gold foil scattering experiment
Thomson's model of the atom was also known as the plum pudding model or a fruitcake model. Since there was no experimental evidence to support Thomson's model of the atom, Ernst Rutherford designed the gold foil experiment in 1909. If the atom was really like a fruit cake, then the most direct way to find out what is inside a fruitcake is to poke a finger into it, which is essentially what Hans Geiger and Ernest Marsden did in 1911. At the suggestion of Ernest Rutherford, they used as probes the fast alpha particles emitted by certain radioactive elements. Alpha particles are helium atoms that have lost two electrons each, leaving them with a charge of 2e.
Geiger and Marsden placed a sample of an alpha-emitting substance behind a lead screen with a small hole in it, so that a narrow beam of alpha particles was produced. This beam was directed at a thin gold foil. A zinc sulfide screen, which gives off a visible flash of light when struck by an alpha particle, was set on the other side of the foil with a microscope to see the flashes.
What Thomson's Model Predicted
If atoms were soft spheres of uniformly distributed positive charge (as Thomson proposed), the α-particles would encounter only a weak, spread-out electrostatic force at any point inside the atom. A fast, massive α-particle has enormous kinetic energy — far more than a diffuse cloud of charge could redirect.
The Geiger–Marsden Experiment (1909)
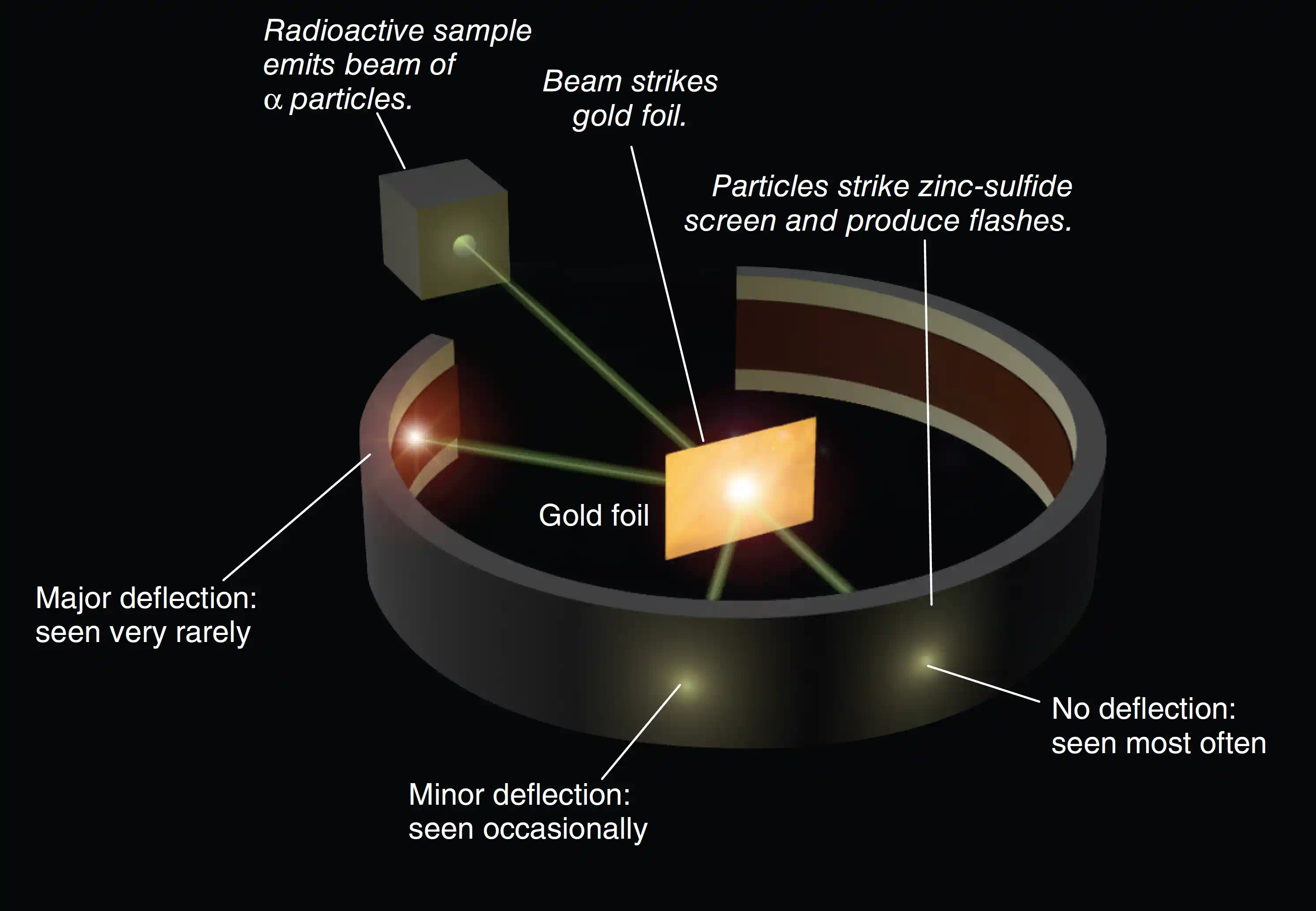
The apparatus was elegantly simple:
- A radioactive source (radium or polonium) inside a lead block emitted α-particles. A narrow slit in the lead acted as a collimator, producing a focused beam.
- The beam was aimed at an ultra-thin gold foil (~100 nm, equivalent to about 400 atoms thick).
- A circular ZnS fluorescent screen surrounded the foil. Every α-particle that struck the screen produced a faint but visible flash, which Geiger and Marsden counted by eye through a low-power microscope — in the dark, for hours at a time.
By rotating the screen to different angles, the team could measure how many particles were scattered in each direction, building up a complete picture of the scattering pattern.
Loading simulator…
Why α-Particles Were the Perfect Atomic Probe
Rutherford understood α-particles better than anyone alive. They were:
- Massive: 4 atomic mass units — about 8000 times heavier than an electron
- Fast: emitted at ~ m/s (about 5% of the speed of light) — enough kinetic energy to probe atomic interiors
- Positively charged: with a charge of +2, they would experience strong electrostatic forces near any concentrated positive charge
- Individually detectable: each α-particle that struck a zinc sulphide (ZnS) screen produced a tiny flash of light, allowing Geiger and Marsden to count each one
This combination made α-particles ideal atomic bullets. If you wanted to map the interior of an atom, there was no better tool available in 1909.
Gold was chosen for the foil because it is extraordinarily malleable — it can be beaten into a sheet just ~100 nm thick (a few hundred atoms), thin enough for most particles to pass through while still presenting a real target.
What Rutherford's Team Actually Observed
The results were so unexpected that Rutherford initially didn't believe them. Three distinct patterns emerged:
(i) Most α-particles passed straight through — undeflected, as if the foil weren't there at all. This confirmed that atoms are mostly empty space.
(ii) A small fraction were deflected by small angles — a few degrees. This was consistent with weak electrostatic interactions near the edges of some concentrated charge.
(iii) About 1 in every 20,000 particles bounced nearly straight back — deflected by more than 90°, some by almost 180°. This was the observation that shattered Thomson's model completely. A uniform, spread-out positive charge simply cannot produce that. You need something extremely dense, extremely small, and carrying all the atom's positive charge in one place — something that can exert a repulsive force of enormous magnitude at very short range.
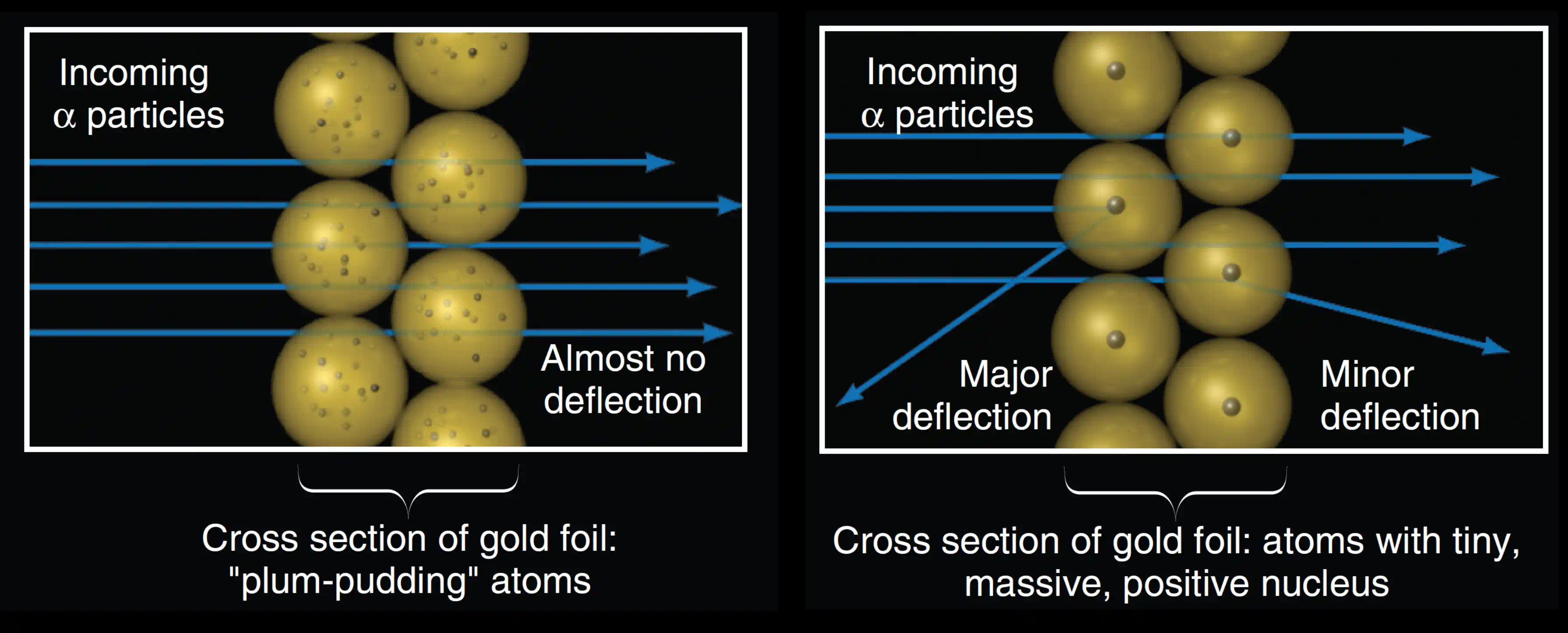
When Rutherford calculated the energy needed to deflect an α-particle by 150°, and compared it to what a uniform atom could deliver, he was stunned. His famous description of the discovery:
"It was quite the most incredible event that has ever happened to me in my life. It was almost as incredible as if you fired a 15-inch shell at a piece of tissue paper and it came back and hit you."
The tissue paper is Thomson's model. The 15-inch shell is the α-particle. The nucleus is what bounced it back.
See It for Yourself: Two Models, One Experiment
The simulator below fires α-particles at both atomic models simultaneously. Watch what happens in Thomson's atom (left) versus Rutherford's atom (right).
Use the Particle Gun Aim slider to aim the beam — try pointing it directly at the nucleus. In Thomson's model, even a direct hit barely deflects the particle. In Rutherford's model, a near-central hit sends the particle bouncing back. The Live Scattering Statistics tally each outcome in real time, exactly mirroring what Geiger and Marsden observed on their ZnS screen.
Loading simulator…
Rutherford's Conclusions from the Data
Rutherford analysed the scattering data mathematically and drew three inescapable conclusions:
1. Most of an atom is empty space.
The fact that the vast majority of α-particles passed straight through meant there was almost nothing for them to interact with. The atom is not a solid sphere — it is overwhelmingly vacuum.
2. All the positive charge (and most of the mass) is concentrated in an extremely tiny region.
Only a direct or near-direct hit could produce a large deflection. Rutherford calculated that the concentrating region — which he named the nucleus — must have a radius of roughly m, or about 1 femtometre. The rest of the atom (where the electrons roam) extends to about m.
3. The size difference is almost incomprehensible.
The ratio of atom radius to nucleus radius is about — a factor of 100,000. Rutherford offered an analogy: if the nucleus were the size of a cricket ball (~7 cm), the radius of the atom would be approximately 5 kilometres. The nucleus is a grain of sand in a football stadium.
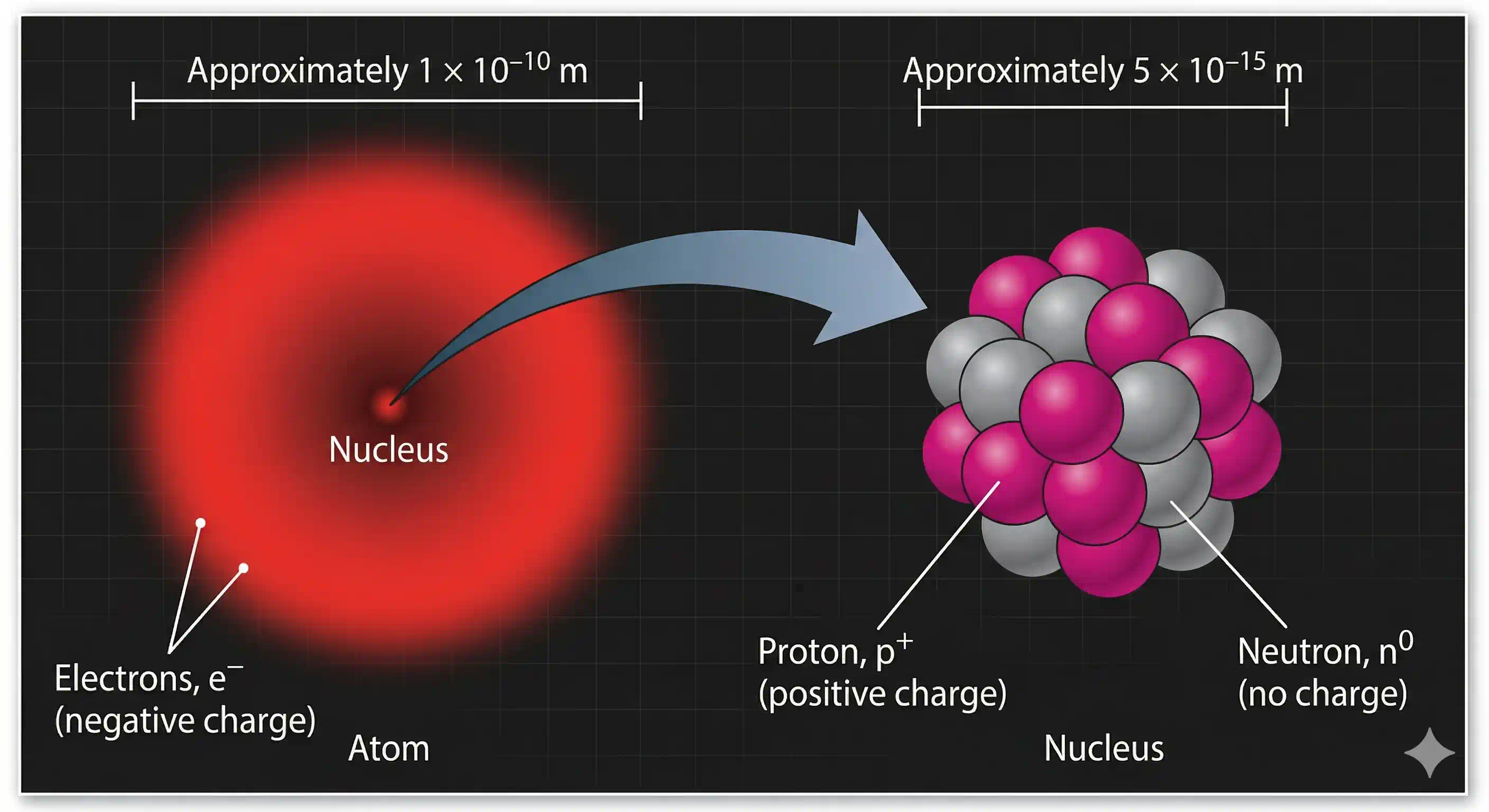
The Nuclear Model: Three Postulates
Based on these conclusions, Rutherford proposed the nuclear model of the atom (formally after the discovery of protons, 1919):
(i) Dense nucleus at the centre. The positive charge and almost all the mass of the atom are concentrated in an extremely small, dense region called the nucleus, with radius ~ m.
(ii) Electrons orbit the nucleus. The negative electrons occupy the vast empty space surrounding the nucleus, moving in circular orbits at very high speeds. This resembles a miniature solar system — the nucleus plays the role of the Sun, and the electrons are the planets.
(iii) Electrostatic attraction holds the system together. The negatively charged electrons are attracted to the positively charged nucleus by Coulomb's electrostatic force, which provides the centripetal force to keep them in their circular paths.
Comparing the sizes of the atom and the nucleus
| Feature | Typical Value | Analogy |
|---|---|---|
| Radius of atom | m (1 Å) | The entire cricket stadium |
| Radius of nucleus | m (1 fm) | A cricket ball at the centre |
| Ratio (atom/nucleus) | If nucleus = 7 cm, atom = 5 km radius | |
| Fraction of volume | Nucleus occupies of atom's volume | ~99.9999999999999% of atom is empty space |
The Cracks in Rutherford's Model
Rutherford's nuclear model was a massive leap forward — but it contained two deep, fatal problems that classical physics had no answer to.
Problem 1: Atomic instability (the spiral-in problem)
According to Maxwell's laws of electromagnetism, any charged particle that undergoes acceleration must radiate energy as electromagnetic radiation. An electron in a circular orbit is constantly changing direction — which means it is constantly accelerating (centripetal acceleration). So according to classical physics, the electron should continuously radiate energy, lose speed, spiral inward toward the nucleus, and collide with it in about seconds.
This means every atom in the universe should have collapsed to a point almost instantly after forming. Obviously, atoms are stable — so there must be something fundamentally wrong with applying Maxwell's equations to orbiting electrons.
Problem 2: It cannot explain atomic line spectra
When atoms are excited (by heat or electricity), they emit light — but not a smooth rainbow. They emit light at specific, discrete wavelengths only — sharp coloured lines. Hydrogen, for instance, has a precise set of lines in the visible range (the Balmer series).
In Rutherford's model, an electron losing energy continuously would emit a continuous spectrum of radiation as it spiralled inward — a smooth smear of all colours. It could never produce sharp discrete lines. The observed line spectra were therefore completely inexplicable within his framework.
Both problems required a fundamentally new kind of physics. That physics — quantum mechanics — arrived with Niels Bohr's model in 1913.
Loading simulator…

The Logic Chain to Remember
Radioactivity → Rutherford discovers α-particles with known properties
↓ Uses α-particles as probes → Geiger–Marsden experiment (1909)
↓ Observation: ~1 in 20,000 α-particles backscatter
↓ Conclusion: Positive charge cannot be spread out — it must be concentrated in a tiny nucleus
↓ Nuclear model: Tiny dense nucleus + electrons orbiting in mostly empty space
↓ Problem: Classical physics predicts electrons spiral in within s; can't explain line spectra → Bohr's model needed
Q1.In Rutherford's gold foil experiment, approximately what fraction of α-particles were deflected by angles greater than 90°?
Ernest Rutherford designed the gold foil experiment in 1909 not to discover a new structure of the atom — but to confirm his supervisor J.J. Thomson's plum pudding model. He expected to prove that atoms were soft, featureless spheres of spread-out charge that α-particles would glide through. What he found instead was so shocking that it rewrote the entire picture of matter — and gave us the nuclear atom we still use today.
Radioactivity: The Key That Unlocked the Atom
To understand why Rutherford's experiment worked, you first need to understand what he had in his hands: a gun that fired particles from inside atoms.
The story begins in 1895 when Wilhelm Röntgen discovered X-rays — invisible radiation produced when fast electrons smash into dense metal targets. These rays passed through skin but not bone, could not be deflected by electric or magnetic fields, and had wavelengths around 0.1 nm. Nobody knew exactly what they were, which is why Röntgen named them X-rays.
One year later, Henri Becquerel (1852–1908) stumbled upon something even stranger. Certain heavy elements emitted radiation spontaneously, without any external energy input. He called this phenomenon radioactivity and the elements responsible radioactive elements.
Becquerel's discovery was taken up by Marie Curie, Pierre Curie, Rutherford, and Frederick Soddy. It was Rutherford who sorted the radiation into three distinct types and named them:

Properties of the three types of radiation from radioactive elements
| Radiation | Nature | Charge | Mass | Penetrating Power | Deflection by Fields |
|---|---|---|---|---|---|
| α (alpha) | Helium nuclei | +2 | 4 u | Lowest — stopped by a sheet of paper | Yes — towards negative plate |
| β (beta) | Fast electrons | −1 | ~0 | ~100× more than α — stopped by Al foil | Yes — towards positive plate |
| γ (gamma) | High-energy EM radiation | 0 | 0 | ~1000× more than α — needs thick lead/concrete | No — not deflected |
The gold foil scattering experiment
Thomson's model of the atom was also known as the plum pudding model or a fruitcake model. Since there was no experimental evidence to support Thomson's model of the atom, Ernst Rutherford designed the gold foil experiment in 1909. If the atom was really like a fruit cake, then the most direct way to find out what is inside a fruitcake is to poke a finger into it, which is essentially what Hans Geiger and Ernest Marsden did in 1911. At the suggestion of Ernest Rutherford, they used as probes the fast alpha particles emitted by certain radioactive elements. Alpha particles are helium atoms that have lost two electrons each, leaving them with a charge of 2e.
Geiger and Marsden placed a sample of an alpha-emitting substance behind a lead screen with a small hole in it, so that a narrow beam of alpha particles was produced. This beam was directed at a thin gold foil. A zinc sulfide screen, which gives off a visible flash of light when struck by an alpha particle, was set on the other side of the foil with a microscope to see the flashes.
What Thomson's Model Predicted
If atoms were soft spheres of uniformly distributed positive charge (as Thomson proposed), the α-particles would encounter only a weak, spread-out electrostatic force at any point inside the atom. A fast, massive α-particle has enormous kinetic energy — far more than a diffuse cloud of charge could redirect.
The Geiger–Marsden Experiment (1909)

The apparatus was elegantly simple:
- A radioactive source (radium or polonium) inside a lead block emitted α-particles. A narrow slit in the lead acted as a collimator, producing a focused beam.
- The beam was aimed at an ultra-thin gold foil (~100 nm, equivalent to about 400 atoms thick).
- A circular ZnS fluorescent screen surrounded the foil. Every α-particle that struck the screen produced a faint but visible flash, which Geiger and Marsden counted by eye through a low-power microscope — in the dark, for hours at a time.
By rotating the screen to different angles, the team could measure how many particles were scattered in each direction, building up a complete picture of the scattering pattern.
Loading simulator…
Why α-Particles Were the Perfect Atomic Probe
Rutherford understood α-particles better than anyone alive. They were:
- Massive: 4 atomic mass units — about 8000 times heavier than an electron
- Fast: emitted at ~ m/s (about 5% of the speed of light) — enough kinetic energy to probe atomic interiors
- Positively charged: with a charge of +2, they would experience strong electrostatic forces near any concentrated positive charge
- Individually detectable: each α-particle that struck a zinc sulphide (ZnS) screen produced a tiny flash of light, allowing Geiger and Marsden to count each one
This combination made α-particles ideal atomic bullets. If you wanted to map the interior of an atom, there was no better tool available in 1909.
Gold was chosen for the foil because it is extraordinarily malleable — it can be beaten into a sheet just ~100 nm thick (a few hundred atoms), thin enough for most particles to pass through while still presenting a real target.
What Rutherford's Team Actually Observed
The results were so unexpected that Rutherford initially didn't believe them. Three distinct patterns emerged:
(i) Most α-particles passed straight through — undeflected, as if the foil weren't there at all. This confirmed that atoms are mostly empty space.
(ii) A small fraction were deflected by small angles — a few degrees. This was consistent with weak electrostatic interactions near the edges of some concentrated charge.
(iii) About 1 in every 20,000 particles bounced nearly straight back — deflected by more than 90°, some by almost 180°. This was the observation that shattered Thomson's model completely. A uniform, spread-out positive charge simply cannot produce that. You need something extremely dense, extremely small, and carrying all the atom's positive charge in one place — something that can exert a repulsive force of enormous magnitude at very short range.

When Rutherford calculated the energy needed to deflect an α-particle by 150°, and compared it to what a uniform atom could deliver, he was stunned. His famous description of the discovery:
"It was quite the most incredible event that has ever happened to me in my life. It was almost as incredible as if you fired a 15-inch shell at a piece of tissue paper and it came back and hit you."
The tissue paper is Thomson's model. The 15-inch shell is the α-particle. The nucleus is what bounced it back.
See It for Yourself: Two Models, One Experiment
The simulator below fires α-particles at both atomic models simultaneously. Watch what happens in Thomson's atom (left) versus Rutherford's atom (right).
Use the Particle Gun Aim slider to aim the beam — try pointing it directly at the nucleus. In Thomson's model, even a direct hit barely deflects the particle. In Rutherford's model, a near-central hit sends the particle bouncing back. The Live Scattering Statistics tally each outcome in real time, exactly mirroring what Geiger and Marsden observed on their ZnS screen.
Loading simulator…
Rutherford's Conclusions from the Data
Rutherford analysed the scattering data mathematically and drew three inescapable conclusions:
1. Most of an atom is empty space.
The fact that the vast majority of α-particles passed straight through meant there was almost nothing for them to interact with. The atom is not a solid sphere — it is overwhelmingly vacuum.
2. All the positive charge (and most of the mass) is concentrated in an extremely tiny region.
Only a direct or near-direct hit could produce a large deflection. Rutherford calculated that the concentrating region — which he named the nucleus — must have a radius of roughly m, or about 1 femtometre. The rest of the atom (where the electrons roam) extends to about m.
3. The size difference is almost incomprehensible.
The ratio of atom radius to nucleus radius is about — a factor of 100,000. Rutherford offered an analogy: if the nucleus were the size of a cricket ball (~7 cm), the radius of the atom would be approximately 5 kilometres. The nucleus is a grain of sand in a football stadium.

The Nuclear Model: Three Postulates
Based on these conclusions, Rutherford proposed the nuclear model of the atom (formally after the discovery of protons, 1919):
(i) Dense nucleus at the centre. The positive charge and almost all the mass of the atom are concentrated in an extremely small, dense region called the nucleus, with radius ~ m.
(ii) Electrons orbit the nucleus. The negative electrons occupy the vast empty space surrounding the nucleus, moving in circular orbits at very high speeds. This resembles a miniature solar system — the nucleus plays the role of the Sun, and the electrons are the planets.
(iii) Electrostatic attraction holds the system together. The negatively charged electrons are attracted to the positively charged nucleus by Coulomb's electrostatic force, which provides the centripetal force to keep them in their circular paths.
Comparing the sizes of the atom and the nucleus
| Feature | Typical Value | Analogy |
|---|---|---|
| Radius of atom | m (1 Å) | The entire cricket stadium |
| Radius of nucleus | m (1 fm) | A cricket ball at the centre |
| Ratio (atom/nucleus) | If nucleus = 7 cm, atom = 5 km radius | |
| Fraction of volume | Nucleus occupies of atom's volume | ~99.9999999999999% of atom is empty space |
The Cracks in Rutherford's Model
Rutherford's nuclear model was a massive leap forward — but it contained two deep, fatal problems that classical physics had no answer to.
Problem 1: Atomic instability (the spiral-in problem)
According to Maxwell's laws of electromagnetism, any charged particle that undergoes acceleration must radiate energy as electromagnetic radiation. An electron in a circular orbit is constantly changing direction — which means it is constantly accelerating (centripetal acceleration). So according to classical physics, the electron should continuously radiate energy, lose speed, spiral inward toward the nucleus, and collide with it in about seconds.
This means every atom in the universe should have collapsed to a point almost instantly after forming. Obviously, atoms are stable — so there must be something fundamentally wrong with applying Maxwell's equations to orbiting electrons.
Problem 2: It cannot explain atomic line spectra
When atoms are excited (by heat or electricity), they emit light — but not a smooth rainbow. They emit light at specific, discrete wavelengths only — sharp coloured lines. Hydrogen, for instance, has a precise set of lines in the visible range (the Balmer series).
In Rutherford's model, an electron losing energy continuously would emit a continuous spectrum of radiation as it spiralled inward — a smooth smear of all colours. It could never produce sharp discrete lines. The observed line spectra were therefore completely inexplicable within his framework.
Both problems required a fundamentally new kind of physics. That physics — quantum mechanics — arrived with Niels Bohr's model in 1913.
Loading simulator…

Q1.In Rutherford's gold foil experiment, approximately what fraction of α-particles were deflected by angles greater than 90°?