Choosing the Right Separation Method
A decision framework
You have three problems: sand mixed in water, alcohol mixed in water, and iron filings mixed in sand. Would you use the same method to solve all three — or does each one need a completely different approach?
In a real laboratory, a chemist rarely uses just one separation technique. Purifying a drug might require: filtration (remove solid impurities) → extraction (separate organic from aqueous layers) → distillation (remove solvent) → crystallisation (obtain pure drug crystals) → chromatography (check purity). Each step targets a different property difference. Knowing which tool to use — and in what order — is as important as knowing how each tool works.
Choosing the Right Technique
The key question is always: What property difference exists between the components?
Use this as your decision guide:
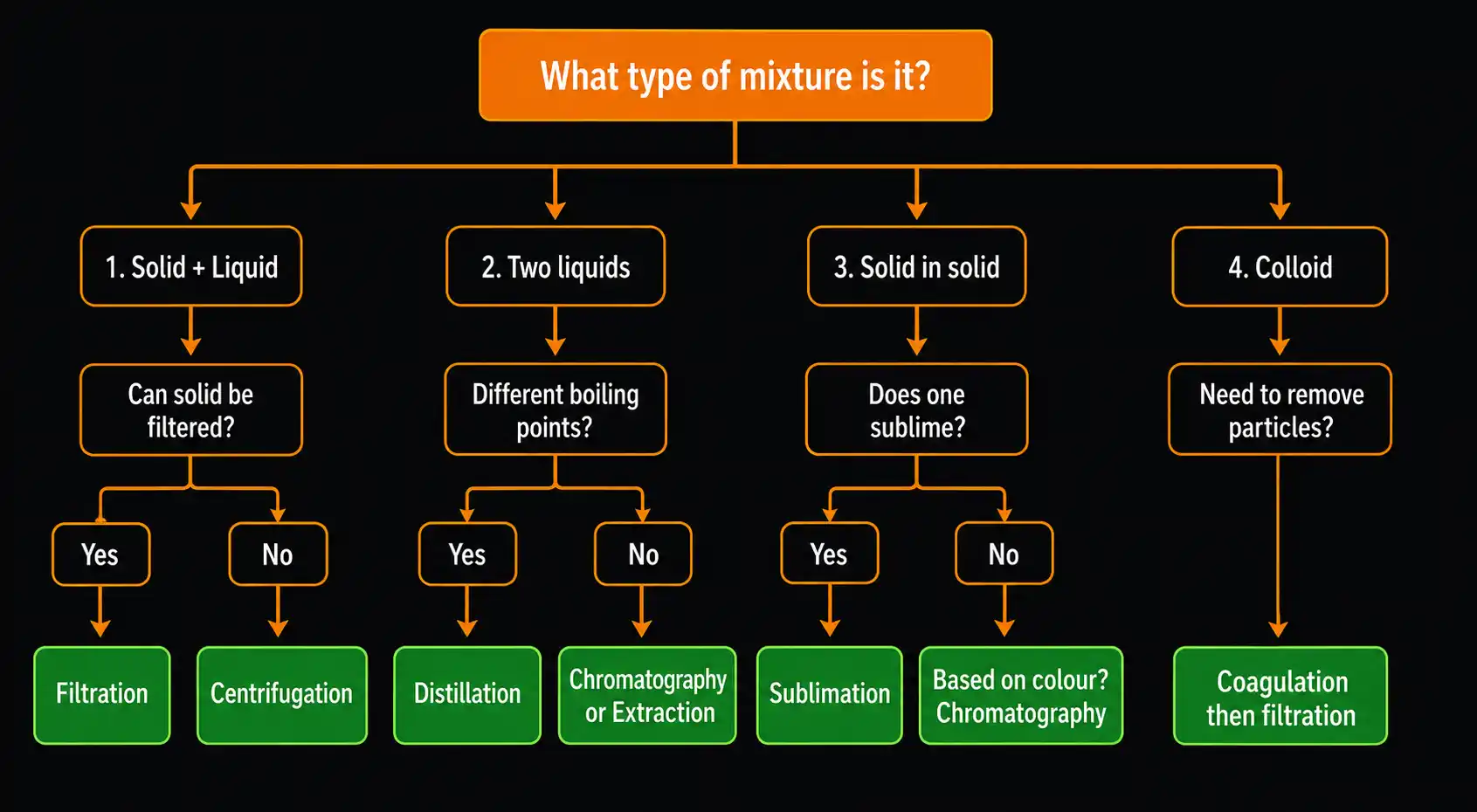
Industry Uses Multiple Techniques in Sequence
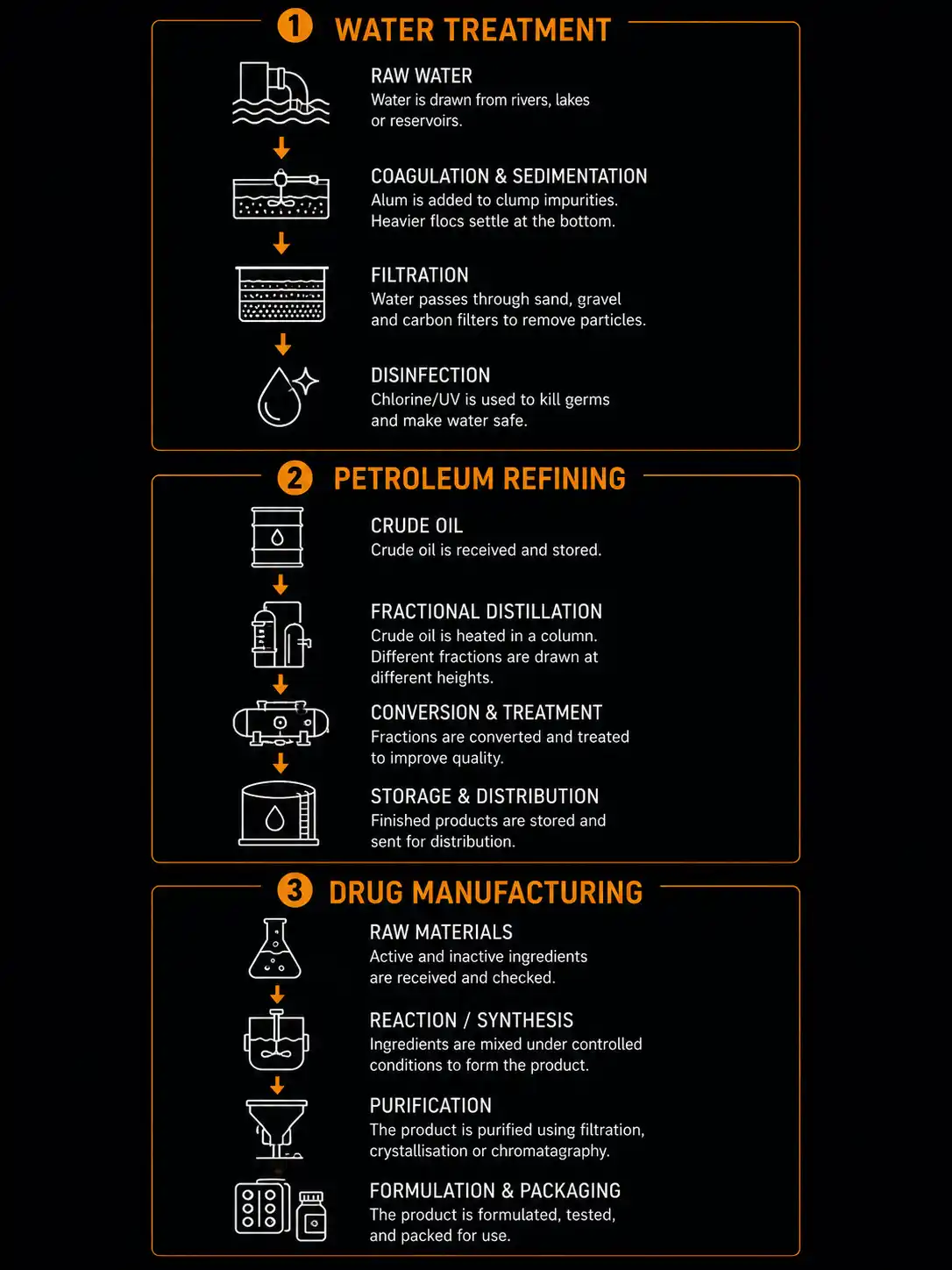
In real-world chemistry, no single separation technique is used in isolation. Industrial processes chain multiple methods together:
Water Treatment Plant (3-stage sequence):
- Coagulation + Sedimentation — alum is added, floc settles
- Filtration — water passes through sand and gravel filters
- Chlorination and UV — chemical and physical disinfection
Petroleum Refining (multi-stage):
- Centrifugation — removes water and solids from crude oil
- Fractional Distillation — separates fractions by boiling point
- Crystallisation (dewaxing) — removes waxy fractions from diesel
- Chromatographic testing — quality control at each stage
Drug Manufacturing (pharmaceutical purification):
- Filtration — removes catalyst after reaction
- Solvent extraction — pulls drug compound from aqueous phase
- Distillation — removes organic solvent
- Recrystallisation — achieves >99.9 % purity
- HPLC chromatography — final purity verification
The key skill is recognising which property difference to exploit at each step — boiling point, particle size, solubility, volatility, or charge.
Separation Beyond the Lab — In Your Body, Your City, Your Phone
Once you start looking for separation, you find it everywhere — even in places that have nothing to do with a school chemistry lab.
Inside your own body, separation is happening every second. Your kidneys filter your blood about 60 times a day, separating waste products like urea from the useful nutrients and water. Without that quiet biological separator, you would not survive 24 hours.
Inside your city, sewage water is treated before it can be released back into rivers. The treatment plant uses a sequence of techniques you have just learnt — sedimentation to settle heavy waste, coagulation with chemicals (like alum) to clump the fine particles, and filtration to remove the rest. The cleaned water is then reused for flushing toilets or watering plants. The same chemistry that gave you clean water from muddy water in the lab is what cleans the water of an entire city.
Inside the world's oceans, plastic waste is a separation problem on a planetary scale. Tiny pieces of plastic — microplastics — are mixed throughout the water and sediment. We do not yet have a good large-scale way to separate them out. This is one of the unsolved problems chemists, biologists, and engineers are working on right now.
Inside your home, waste segregation is separation done by hand. Dry waste (plastic, paper, glass, metal) can be recycled. Wet waste (food scraps, vegetable peels) can be composted. The few seconds it takes you to throw the right thing into the right bin make a real difference.
Inside an old phone, your discarded mobile or laptop battery contains tiny but valuable amounts of lithium, cobalt, and other rare metals. Researchers are now finding cleaner ways to recover these metals — using techniques that are extensions of the chemistry you've met in this chapter. As phones pile up, this kind of separation will only become more important.
Separation isn't a school topic — it's a survival skill for the 21st century.
Sewage to Safe Water — The Same Chapter on a City Scale
A modern sewage treatment plant uses, in order:
The Quest Continues — Pulling Lithium Back from a Mountain of Old Phones
The world is using more lithium-ion batteries every year — for phones, laptops, electric scooters and electric cars. India alone will have hundreds of millions of dead batteries by 2030. Lithium is precious; it cannot be wasted.
At a Glance — A Closing Recap
Before you turn the page, take a breath and look back. In this single chapter you have learnt:
A student has a cup of tea (liquid tea + dissolved sugar + tea leaves). She wants to separate all three. A classmate says: "Just filter it — done." What is the flaw in this plan, and what additional step is needed?
🏭 Real-World Impact
Chemical engineers at companies like Dr Reddy's Laboratories, Sun Pharma, and Lupin design multi-stage separation trains that run 24/7 to produce generic medicines for the world. India is the pharmacy of the world — supplying 40 % of US generic drugs and 25 % of UK medicines. This entire industry runs on the separation techniques in this chapter, applied in sequence.
Q1.Which separation method uses the difference in ability to convert directly from solid to vapour?
You have three problems: sand mixed in water, alcohol mixed in water, and iron filings mixed in sand. Would you use the same method to solve all three — or does each one need a completely different approach?
In a real laboratory, a chemist rarely uses just one separation technique. Purifying a drug might require: filtration (remove solid impurities) → extraction (separate organic from aqueous layers) → distillation (remove solvent) → crystallisation (obtain pure drug crystals) → chromatography (check purity). Each step targets a different property difference. Knowing which tool to use — and in what order — is as important as knowing how each tool works.
Choosing the Right Technique
The key question is always: What property difference exists between the components?
Use this as your decision guide:

Industry Uses Multiple Techniques in Sequence
In real-world chemistry, no single separation technique is used in isolation. Industrial processes chain multiple methods together:
Water Treatment Plant (3-stage sequence):
- Coagulation + Sedimentation — alum is added, floc settles
- Filtration — water passes through sand and gravel filters
- Chlorination and UV — chemical and physical disinfection
Petroleum Refining (multi-stage):
- Centrifugation — removes water and solids from crude oil
- Fractional Distillation — separates fractions by boiling point
- Crystallisation (dewaxing) — removes waxy fractions from diesel
- Chromatographic testing — quality control at each stage
Drug Manufacturing (pharmaceutical purification):
- Filtration — removes catalyst after reaction
- Solvent extraction — pulls drug compound from aqueous phase
- Distillation — removes organic solvent
- Recrystallisation — achieves >99.9 % purity
- HPLC chromatography — final purity verification
The key skill is recognising which property difference to exploit at each step — boiling point, particle size, solubility, volatility, or charge.
Separation Beyond the Lab — In Your Body, Your City, Your Phone
Once you start looking for separation, you find it everywhere — even in places that have nothing to do with a school chemistry lab.
Inside your own body, separation is happening every second. Your kidneys filter your blood about 60 times a day, separating waste products like urea from the useful nutrients and water. Without that quiet biological separator, you would not survive 24 hours.
Inside your city, sewage water is treated before it can be released back into rivers. The treatment plant uses a sequence of techniques you have just learnt — sedimentation to settle heavy waste, coagulation with chemicals (like alum) to clump the fine particles, and filtration to remove the rest. The cleaned water is then reused for flushing toilets or watering plants. The same chemistry that gave you clean water from muddy water in the lab is what cleans the water of an entire city.
Inside the world's oceans, plastic waste is a separation problem on a planetary scale. Tiny pieces of plastic — microplastics — are mixed throughout the water and sediment. We do not yet have a good large-scale way to separate them out. This is one of the unsolved problems chemists, biologists, and engineers are working on right now.
Inside your home, waste segregation is separation done by hand. Dry waste (plastic, paper, glass, metal) can be recycled. Wet waste (food scraps, vegetable peels) can be composted. The few seconds it takes you to throw the right thing into the right bin make a real difference.
Inside an old phone, your discarded mobile or laptop battery contains tiny but valuable amounts of lithium, cobalt, and other rare metals. Researchers are now finding cleaner ways to recover these metals — using techniques that are extensions of the chemistry you've met in this chapter. As phones pile up, this kind of separation will only become more important.
Separation isn't a school topic — it's a survival skill for the 21st century.
Sewage to Safe Water — The Same Chapter on a City Scale
A modern sewage treatment plant uses, in order:
The Quest Continues — Pulling Lithium Back from a Mountain of Old Phones
The world is using more lithium-ion batteries every year — for phones, laptops, electric scooters and electric cars. India alone will have hundreds of millions of dead batteries by 2030. Lithium is precious; it cannot be wasted.
At a Glance — A Closing Recap
Before you turn the page, take a breath and look back. In this single chapter you have learnt:
A student has a cup of tea (liquid tea + dissolved sugar + tea leaves). She wants to separate all three. A classmate says: "Just filter it — done." What is the flaw in this plan, and what additional step is needed?
Q1.Which separation method uses the difference in ability to convert directly from solid to vapour?