The Tyndall Effect
Why colloids scatter light
What is the Tyndall Effect?
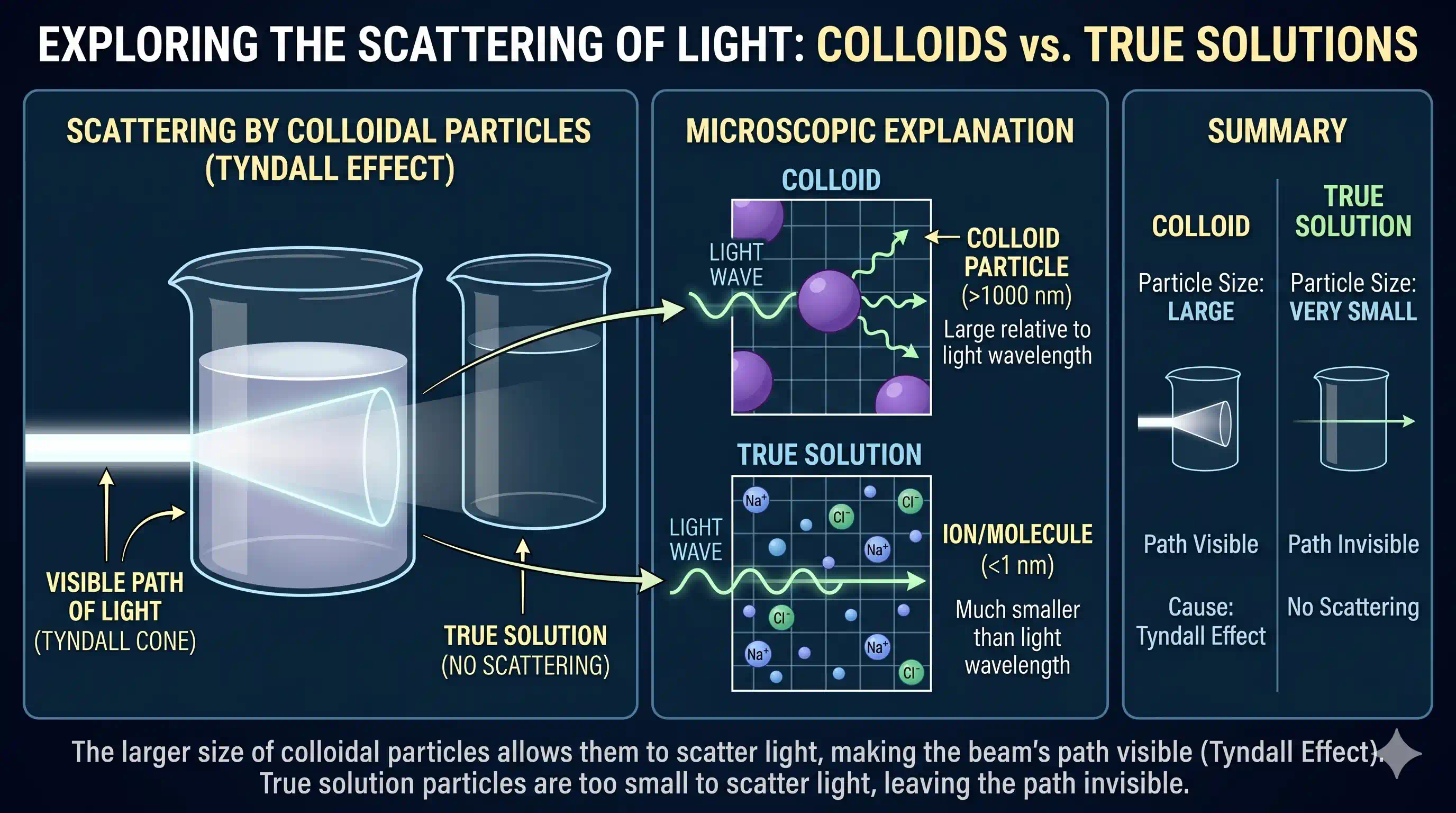
When a beam of light passes through a colloid, the colloidal particles scatter the light in all directions — making the beam's path visible when viewed from the side. This phenomenon is called the Tyndall Effect, named after physicist John Tyndall who studied it in 1869.
Why does scattering happen in colloids but not true solutions?
Think of it this way: visible light has a wavelength of roughly 400–700 nm. Colloidal particles (1–1000 nm) are comparable in size to this wavelength. When light encounters a particle of similar size, it bounces off in all directions — just like a stone thrown into a pond makes ripples spread outward.
In a true solution, the dissolved particles (ions or molecules) are smaller than 1 nm — far too small to interact with visible light. The light wave simply passes through without "noticing" them. No scattering, no visible beam.
In a suspension, particles are large enough to scatter light, but they eventually settle to the bottom. Any scattering effect is temporary.
Only the colloid — with particles in the 1–1000 nm range — produces the steady, persistent glowing beam that is the Tyndall Effect.
How to observe it at home:
Shine a torch through a glass of milk (colloid) — you see a bright beam inside. Now shine it through a glass of clear water (solution) — the beam is invisible inside the glass. This simple test distinguishes a colloid from a true solution.
Why It is Called the Tyndall Effect
The effect is named after John Tyndall, a 19th-century Irish physicist who first explained why a beam of light becomes visible when it travels through certain mixtures. He was studying why the sky is blue and why distant mountains look hazy, and he traced both back to the same idea — tiny particles in the air scatter light in every direction.
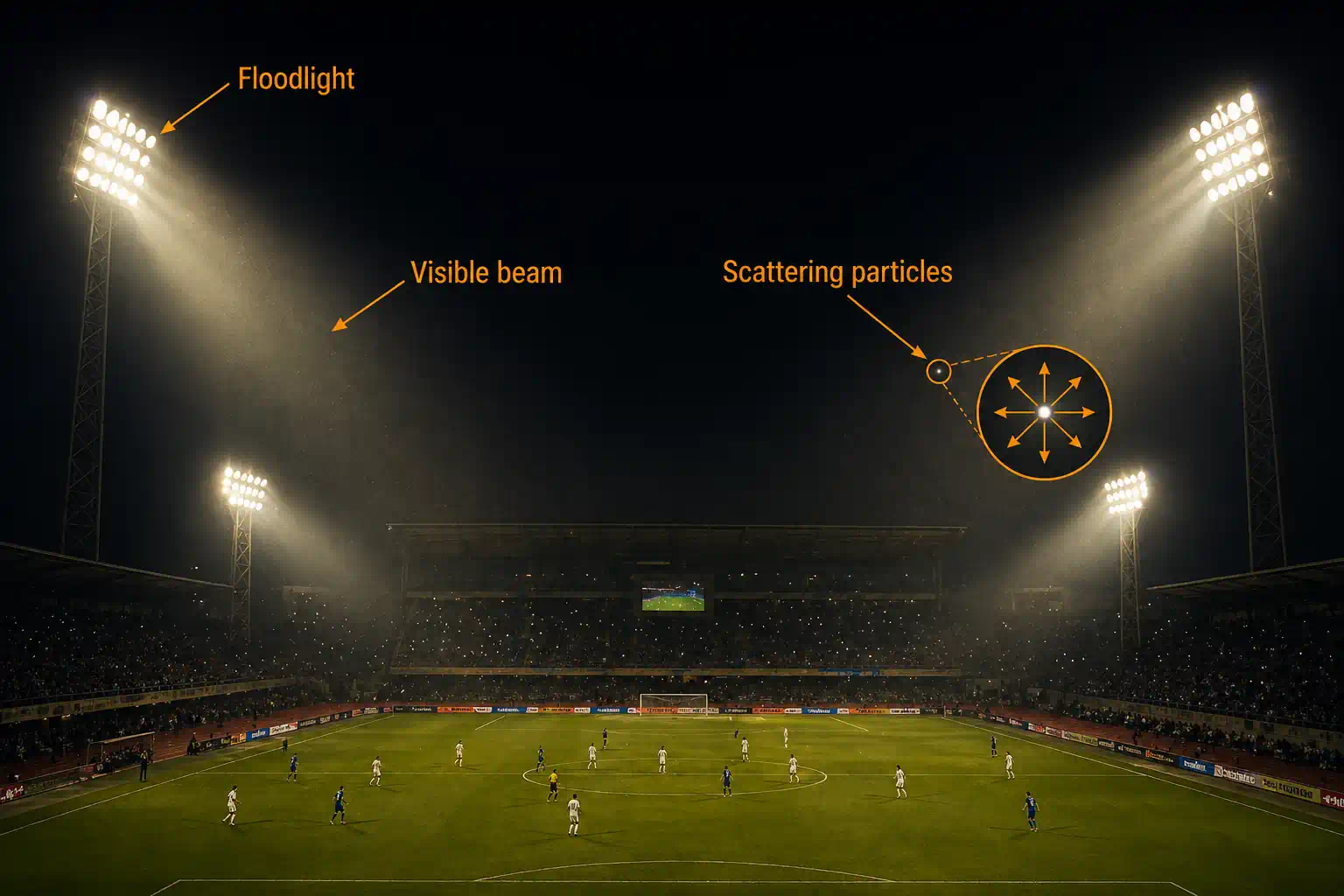
He went one step further and proved that the bigger the dust or smoke particles in the air, the brighter the scattering. That's why a sunbeam through a clean room is invisible, but a sunbeam through a dusty room glows so clearly you can almost touch it.
The Two Parts of Every Colloid
Every colloid has two parts. The tiny particles that are spread around — like the fat droplets in milk, or the smoke particles in air — are called the dispersed phase. The substance that they are spread through — the water in milk, or the air around the smoke — is called the dispersion medium.
Think of it as the guest and the host: the dispersed phase is the guest, the dispersion medium is the host who is letting the guest hang around without fully dissolving them.
- In milk, fat droplets (dispersed phase) float through water (dispersion medium).
- In smoke, carbon particles (dispersed phase) float through air (dispersion medium).
- In fog, tiny water droplets (dispersed phase) float through air (dispersion medium).
Knowing these two names will help you make sense of every colloid you meet from now on.
Where You See This Every Day
The Tyndall Effect is one of those phenomena you have been seeing your whole life without knowing the name:
- Blue sky — sunlight scatters off tiny air molecules and dust particles (colloid-sized). Shorter blue wavelengths scatter more than red, making the sky appear blue from below.
- Red sunset — at sunrise/sunset, light travels through more atmosphere. Blue scatters away; only the longer red wavelengths reach your eyes.
- Car headlights in fog — the beam is dramatically visible in fog (water droplets = colloid) but invisible on a clear night (clean air = no scattering).
- Laser pointer in a dusty room — you can trace the entire path of the beam because dust particles scatter the light.
- Blue eyes — the iris of blue-eyed people contains colloidal particles that scatter blue light by the Tyndall Effect. There is actually no blue pigment — it is all scattering!
What is the Tyndall Effect?
When a beam of light passes through a colloid, the colloidal particles scatter the light in all directions — making the beam's path visible when viewed from the side. This phenomenon is called the Tyndall Effect, named after physicist John Tyndall who studied it in 1869.
Why does scattering happen in colloids but not true solutions?
Think of it this way: visible light has a wavelength of roughly 400–700 nm. Colloidal particles (1–1000 nm) are comparable in size to this wavelength. When light encounters a particle of similar size, it bounces off in all directions — just like a stone thrown into a pond makes ripples spread outward.
In a true solution, the dissolved particles (ions or molecules) are smaller than 1 nm — far too small to interact with visible light. The light wave simply passes through without "noticing" them. No scattering, no visible beam.
In a suspension, particles are large enough to scatter light, but they eventually settle to the bottom. Any scattering effect is temporary.
Only the colloid — with particles in the 1–1000 nm range — produces the steady, persistent glowing beam that is the Tyndall Effect.
How to observe it at home:
Shine a torch through a glass of milk (colloid) — you see a bright beam inside. Now shine it through a glass of clear water (solution) — the beam is invisible inside the glass. This simple test distinguishes a colloid from a true solution.
Why It is Called the Tyndall Effect
The effect is named after John Tyndall, a 19th-century Irish physicist who first explained why a beam of light becomes visible when it travels through certain mixtures. He was studying why the sky is blue and why distant mountains look hazy, and he traced both back to the same idea — tiny particles in the air scatter light in every direction.
He went one step further and proved that the bigger the dust or smoke particles in the air, the brighter the scattering. That's why a sunbeam through a clean room is invisible, but a sunbeam through a dusty room glows so clearly you can almost touch it.
The Two Parts of Every Colloid
Every colloid has two parts. The tiny particles that are spread around — like the fat droplets in milk, or the smoke particles in air — are called the dispersed phase. The substance that they are spread through — the water in milk, or the air around the smoke — is called the dispersion medium.
Think of it as the guest and the host: the dispersed phase is the guest, the dispersion medium is the host who is letting the guest hang around without fully dissolving them.
- In milk, fat droplets (dispersed phase) float through water (dispersion medium).
- In smoke, carbon particles (dispersed phase) float through air (dispersion medium).
- In fog, tiny water droplets (dispersed phase) float through air (dispersion medium).
Knowing these two names will help you make sense of every colloid you meet from now on.
Where You See This Every Day
The Tyndall Effect is one of those phenomena you have been seeing your whole life without knowing the name:
- Blue sky — sunlight scatters off tiny air molecules and dust particles (colloid-sized). Shorter blue wavelengths scatter more than red, making the sky appear blue from below.
- Red sunset — at sunrise/sunset, light travels through more atmosphere. Blue scatters away; only the longer red wavelengths reach your eyes.
- Car headlights in fog — the beam is dramatically visible in fog (water droplets = colloid) but invisible on a clear night (clean air = no scattering).
- Laser pointer in a dusty room — you can trace the entire path of the beam because dust particles scatter the light.
- Blue eyes — the iris of blue-eyed people contains colloidal particles that scatter blue light by the Tyndall Effect. There is actually no blue pigment — it is all scattering!