Crystallisation
How it works and where we use it
Sea water is full of dissolved salt — invisible, completely mixed in. Salt farmers somehow get dry, solid salt from this water without any electricity or machines. How do you think they do it?
Every snowflake is a crystal of water. They all have 6-fold symmetry — because water molecules form hexagonal arrangements when they freeze. No two snowflakes are exactly alike because each one grows in slightly different atmospheric conditions. Crystallisation produces some of nature's most precise geometric structures — from table salt cubes to diamond octahedra.
What is Crystallisation?
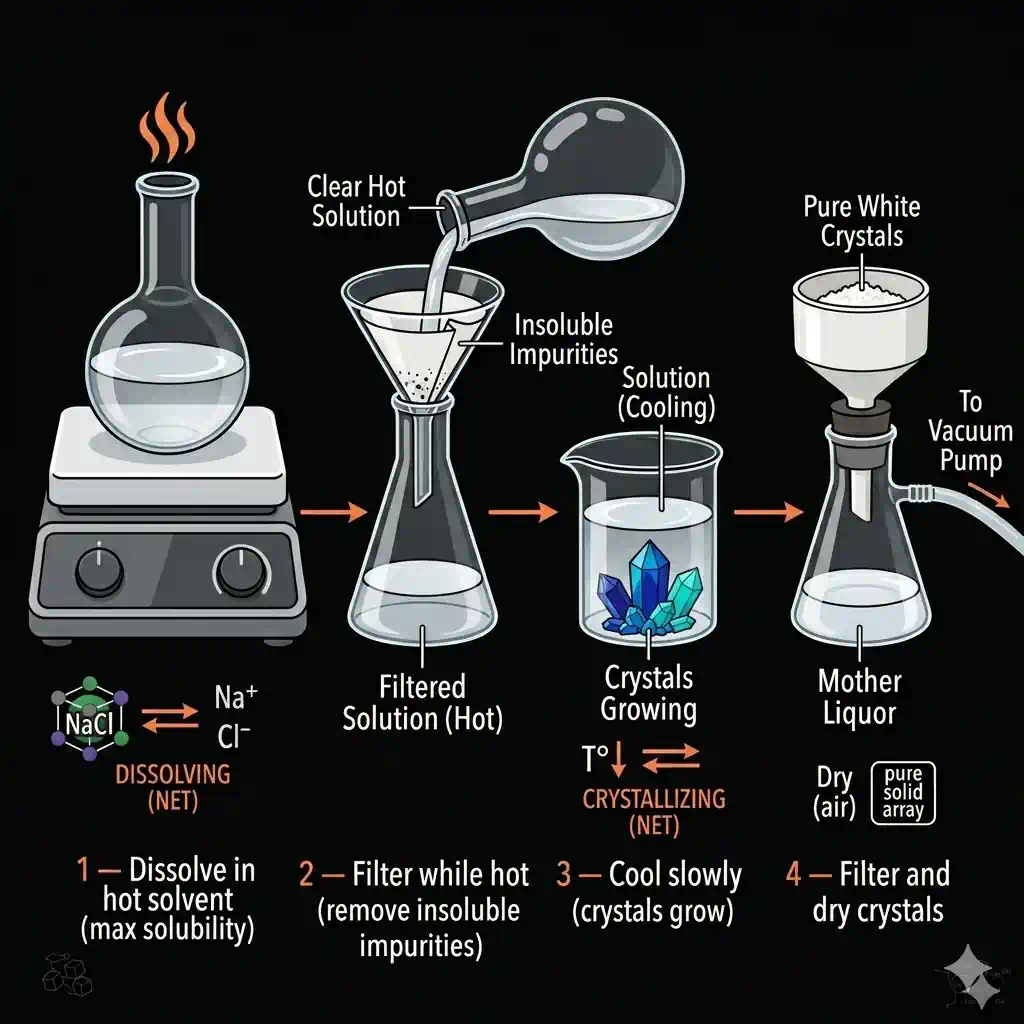
Crystallisation is a separation technique that uses the difference in solubility at different temperatures to separate a dissolved solid from its solution — and produces the solid in a pure, crystalline form.
How it works:
- Dissolve the impure solid in the minimum amount of hot solvent (near saturation)
- Filter the hot solution to remove insoluble impurities
- Allow to cool slowly — as temperature drops, solubility decreases
- Excess solute separates as pure crystals
- Filter and dry the crystals
Why crystallisation gives purer crystals than evaporation:
When you evaporate a solution to dryness, all dissolved substances (both wanted and unwanted) crystallise together. With controlled crystallisation, you can tune the temperature and rate so that primarily the target substance crystallises, leaving impurities in solution (in the mother liquor).
What Exactly is a Crystal?
Before we go further — what is a crystal? A crystal is a solid in which the particles are arranged in a beautiful, repeating geometric pattern. Look at any crystal under a magnifier and you will see flat faces and sharp angles, almost as if someone cut the solid by hand.
You have probably seen crystals all around you without naming them:
- Rock salt — those big pinkish chunks of sendha namak in the kitchen are natural salt crystals.
- Sugar candy (mishri in Hindi) — the clear, sparkling lumps you eat after meals are sugar crystals slowly grown from a hot sugar solution.
- Snowflakes — frozen water crystals that form when water vapour cools rapidly in the air. No two are quite the same.
- Frost on a window — tiny ice crystals deposited directly from water vapour in the air.
Nature has been growing crystals for billions of years. In the lab, we just learn to do it on demand.

You prepare a saturated copper sulphate solution in hot water and let it cool slowly overnight. Beautiful blue crystals appear. If instead you rapidly evaporated the same solution on high heat, predict what you would get and why it would look different.
Activity 5.3 — Grow Your Own Copper Sulfate Crystals
Now let us actually grow some crystals. The most beautiful and reliable choice for school labs is copper sulfate — also known as blue vitriol (neela thotha in Hindi). If copper sulfate isn't available, you can use common salt instead.
You will need: copper sulfate powder, a 100 mL beaker, water, a glass rod, dilute sulfuric acid (one drop only), a tripod stand with wire gauze, a burner, a conical funnel with filter paper, a watch glass.
Steps:
- Take 1 g of copper sulfate and place it in a 100 mL beaker. Add 25 mL of water, then a single drop of dilute sulfuric acid (your teacher will add this). Gently warm the beaker on a water bath while stirring with the glass rod. The sulfuric acid stops unwanted reactions and helps the final crystals stay pure.
- Slowly add more copper sulfate, stirring all the time, until no more dissolves — this is your saturated solution.
- Filter the hot solution through a conical funnel with filter paper to remove any insoluble dust or impurities. Collect the clear blue filtrate in a fresh beaker.
- Cover the beaker with a watch glass and let it cool slowly without disturbing it. Slow cooling gives time for the particles to arrange themselves into proper geometric shapes — you will get larger, shinier, well-formed crystals.
- Once cooled, filter the crystals, rinse them quickly with cold water, and let them dry on a watch glass.
You should now have a small heap of brilliant blue, well-shaped copper sulfate crystals — a result you grew yourself.
Safety First
- Copper sulfate is toxic. Don't touch it with bare hands and never taste it. Always work under adult supervision.
- Sulfuric acid is highly corrosive. Only your teacher should add the drop. If even a tiny splash touches your skin, wash with lots of cold water and tell your teacher immediately.
- Use tongs, not your fingers, to handle the hot beaker.
Think as a Scientist — Slow vs Fast Cooling
Suppose two friends do the same activity. Friend A lets her saturated copper sulfate solution cool slowly at room temperature, untouched. Friend B, in a hurry, dunks his beaker into ice-cold water to cool it quickly.

Activity 5.4 — From the Sea to the Salt Bowl
Most of the salt on your dining table started its journey in the sea. The process is just three steps:
Seawater → Saturated solution → Salt crystals
Salt farmers along the coasts of Gujarat, Tamil Nadu and Andhra Pradesh let seawater into shallow flat fields called salt pans. Sun and wind slowly evaporate the water. As the water leaves, the solution becomes more and more concentrated until it is saturated. Eventually, dazzling white salt crystals begin to appear on the bed of the pan. The farmers rake them up by hand. The same simple chemistry — solubility plus evaporation plus crystallisation — feeds an entire country.
India's Two Salts — Panga and Karkatch
Long before modern factories, coastal communities in India had perfected two slightly different ways of getting pure salt from the sea:
Ready to Go Beyond — Crystals That Grew Themselves
You don't have to grow crystals — sometimes nature does it for you, on a scale that is hard to believe.
Pause and Ponder
Two questions to chew on before the next page:
Real-world uses in India:
- Salt production: Sambhar Lake (Rajasthan) — India's largest inland saltwater lake — uses solar evaporation, then crystallisation to produce salt
- Sugar refining: Raw cane sugar is dissolved and recrystallised multiple times to get white refined sugar
- Alum (Phitkari): Crystallised from aluminium sulfate solutions — used in water purification
- Drug manufacturing: Most pharmaceutical crystals are produced by controlled crystallisation for consistent purity and particle size
From Lab to Industry — Crystallisation at Scale
The crystallisation process you study in lab (hot → filter → cool → collect) is used at massive industrial scale:
- Salt Production — India is the world's third-largest salt producer. The Rann of Kutch and Chilika Lake produce salt by solar evaporation of seawater followed by natural crystallisation as brine becomes saturated. Zero energy input — just sun and time.
- Sugar from Sugarcane — Sugarcane juice is concentrated by heating, then seeded with tiny sugar crystals to trigger crystallisation. The brown mother liquor (molasses) is separated, giving white sugar. Jaggery (gur) is made when crystallisation is incomplete — it retains impurities and minerals.
- Pharmaceutical Manufacturing — Drugs like aspirin, ibuprofen, and paracetamol are crystallised multiple times to achieve >99.9 % purity before being compressed into tablets. A single recrystallisation step can increase purity from 95 % to 99.5 %.
- Gemstones — Diamonds, rubies, sapphires, and emeralds are all crystals formed under extreme geological conditions. Lab-grown gemstones use controlled crystallisation from supersaturated solutions or melts.
💎 Real-World Impact
India's Sambhar Salt Lake produces over 190,000 tonnes of salt per year by solar crystallisation — the largest salt lake in the country. The technology is millennia old: ancient texts describe the process in detail. Modern pharmaceutical crystallisation is a billion-dollar industry built on the same fundamental principle you have just learned.
Q1.Which of the following best describes how crystallisation purifies a substance?
Sea water is full of dissolved salt — invisible, completely mixed in. Salt farmers somehow get dry, solid salt from this water without any electricity or machines. How do you think they do it?
Every snowflake is a crystal of water. They all have 6-fold symmetry — because water molecules form hexagonal arrangements when they freeze. No two snowflakes are exactly alike because each one grows in slightly different atmospheric conditions. Crystallisation produces some of nature's most precise geometric structures — from table salt cubes to diamond octahedra.
What is Crystallisation?
Crystallisation is a separation technique that uses the difference in solubility at different temperatures to separate a dissolved solid from its solution — and produces the solid in a pure, crystalline form.
How it works:
- Dissolve the impure solid in the minimum amount of hot solvent (near saturation)
- Filter the hot solution to remove insoluble impurities
- Allow to cool slowly — as temperature drops, solubility decreases
- Excess solute separates as pure crystals
- Filter and dry the crystals
Why crystallisation gives purer crystals than evaporation:
When you evaporate a solution to dryness, all dissolved substances (both wanted and unwanted) crystallise together. With controlled crystallisation, you can tune the temperature and rate so that primarily the target substance crystallises, leaving impurities in solution (in the mother liquor).
What Exactly is a Crystal?
Before we go further — what is a crystal? A crystal is a solid in which the particles are arranged in a beautiful, repeating geometric pattern. Look at any crystal under a magnifier and you will see flat faces and sharp angles, almost as if someone cut the solid by hand.
You have probably seen crystals all around you without naming them:
- Rock salt — those big pinkish chunks of sendha namak in the kitchen are natural salt crystals.
- Sugar candy (mishri in Hindi) — the clear, sparkling lumps you eat after meals are sugar crystals slowly grown from a hot sugar solution.
- Snowflakes — frozen water crystals that form when water vapour cools rapidly in the air. No two are quite the same.
- Frost on a window — tiny ice crystals deposited directly from water vapour in the air.
Nature has been growing crystals for billions of years. In the lab, we just learn to do it on demand.

You prepare a saturated copper sulphate solution in hot water and let it cool slowly overnight. Beautiful blue crystals appear. If instead you rapidly evaporated the same solution on high heat, predict what you would get and why it would look different.

Activity 5.3 — Grow Your Own Copper Sulfate Crystals
Now let us actually grow some crystals. The most beautiful and reliable choice for school labs is copper sulfate — also known as blue vitriol (neela thotha in Hindi). If copper sulfate isn't available, you can use common salt instead.
You will need: copper sulfate powder, a 100 mL beaker, water, a glass rod, dilute sulfuric acid (one drop only), a tripod stand with wire gauze, a burner, a conical funnel with filter paper, a watch glass.
Steps:
- Take 1 g of copper sulfate and place it in a 100 mL beaker. Add 25 mL of water, then a single drop of dilute sulfuric acid (your teacher will add this). Gently warm the beaker on a water bath while stirring with the glass rod. The sulfuric acid stops unwanted reactions and helps the final crystals stay pure.
- Slowly add more copper sulfate, stirring all the time, until no more dissolves — this is your saturated solution.
- Filter the hot solution through a conical funnel with filter paper to remove any insoluble dust or impurities. Collect the clear blue filtrate in a fresh beaker.
- Cover the beaker with a watch glass and let it cool slowly without disturbing it. Slow cooling gives time for the particles to arrange themselves into proper geometric shapes — you will get larger, shinier, well-formed crystals.
- Once cooled, filter the crystals, rinse them quickly with cold water, and let them dry on a watch glass.
You should now have a small heap of brilliant blue, well-shaped copper sulfate crystals — a result you grew yourself.
Safety First
- Copper sulfate is toxic. Don't touch it with bare hands and never taste it. Always work under adult supervision.
- Sulfuric acid is highly corrosive. Only your teacher should add the drop. If even a tiny splash touches your skin, wash with lots of cold water and tell your teacher immediately.
- Use tongs, not your fingers, to handle the hot beaker.
Think as a Scientist — Slow vs Fast Cooling
Suppose two friends do the same activity. Friend A lets her saturated copper sulfate solution cool slowly at room temperature, untouched. Friend B, in a hurry, dunks his beaker into ice-cold water to cool it quickly.

Activity 5.4 — From the Sea to the Salt Bowl
Most of the salt on your dining table started its journey in the sea. The process is just three steps:
Seawater → Saturated solution → Salt crystals
Salt farmers along the coasts of Gujarat, Tamil Nadu and Andhra Pradesh let seawater into shallow flat fields called salt pans. Sun and wind slowly evaporate the water. As the water leaves, the solution becomes more and more concentrated until it is saturated. Eventually, dazzling white salt crystals begin to appear on the bed of the pan. The farmers rake them up by hand. The same simple chemistry — solubility plus evaporation plus crystallisation — feeds an entire country.
India's Two Salts — Panga and Karkatch
Long before modern factories, coastal communities in India had perfected two slightly different ways of getting pure salt from the sea:
Ready to Go Beyond — Crystals That Grew Themselves
You don't have to grow crystals — sometimes nature does it for you, on a scale that is hard to believe.
Pause and Ponder
Two questions to chew on before the next page:
Real-world uses in India:
- Salt production: Sambhar Lake (Rajasthan) — India's largest inland saltwater lake — uses solar evaporation, then crystallisation to produce salt
- Sugar refining: Raw cane sugar is dissolved and recrystallised multiple times to get white refined sugar
- Alum (Phitkari): Crystallised from aluminium sulfate solutions — used in water purification
- Drug manufacturing: Most pharmaceutical crystals are produced by controlled crystallisation for consistent purity and particle size
From Lab to Industry — Crystallisation at Scale
The crystallisation process you study in lab (hot → filter → cool → collect) is used at massive industrial scale:
- Salt Production — India is the world's third-largest salt producer. The Rann of Kutch and Chilika Lake produce salt by solar evaporation of seawater followed by natural crystallisation as brine becomes saturated. Zero energy input — just sun and time.
- Sugar from Sugarcane — Sugarcane juice is concentrated by heating, then seeded with tiny sugar crystals to trigger crystallisation. The brown mother liquor (molasses) is separated, giving white sugar. Jaggery (gur) is made when crystallisation is incomplete — it retains impurities and minerals.
- Pharmaceutical Manufacturing — Drugs like aspirin, ibuprofen, and paracetamol are crystallised multiple times to achieve >99.9 % purity before being compressed into tablets. A single recrystallisation step can increase purity from 95 % to 99.5 %.
- Gemstones — Diamonds, rubies, sapphires, and emeralds are all crystals formed under extreme geological conditions. Lab-grown gemstones use controlled crystallisation from supersaturated solutions or melts.
Q1.Which of the following best describes how crystallisation purifies a substance?