Homogeneous and Heterogeneous Mixtures
Two types, one idea — composition
When you stir sugar into tea, it completely disappears — every sip is equally sweet. But stir sand into water and the sand is still visible, and settles to the bottom if you wait. What do you think is the fundamental difference between these two mixtures?
24-carat gold is pure gold — but pure gold is so soft it bends under finger pressure. 22-carat gold (91.7% gold + 8.3% silver or copper) is harder, more durable, and far more practical. The gold jewellery most Indians wear daily is a homogeneous mixture — a solid solution — where you cannot tell the metals apart even under a microscope. The entire history of metallurgy is the art of making homogeneous mixtures.

Mixtures are normally classified into two types — homogeneous and heterogeneous. To understand what these terms mean, we first need to understand what composition means for a mixture.
Composition is simply the answer to two questions: what is a mixture made of, and how much of each component is present? Think of a glass of fresh orange juice — its composition includes water, natural sugars, vitamin C, and fruit acids, each in some proportion. Squeeze more orange into one glass, and its composition changes, even though both glasses look like juice.
Now comes the crucial question: is that composition the same at every point, or does it vary from place to place?
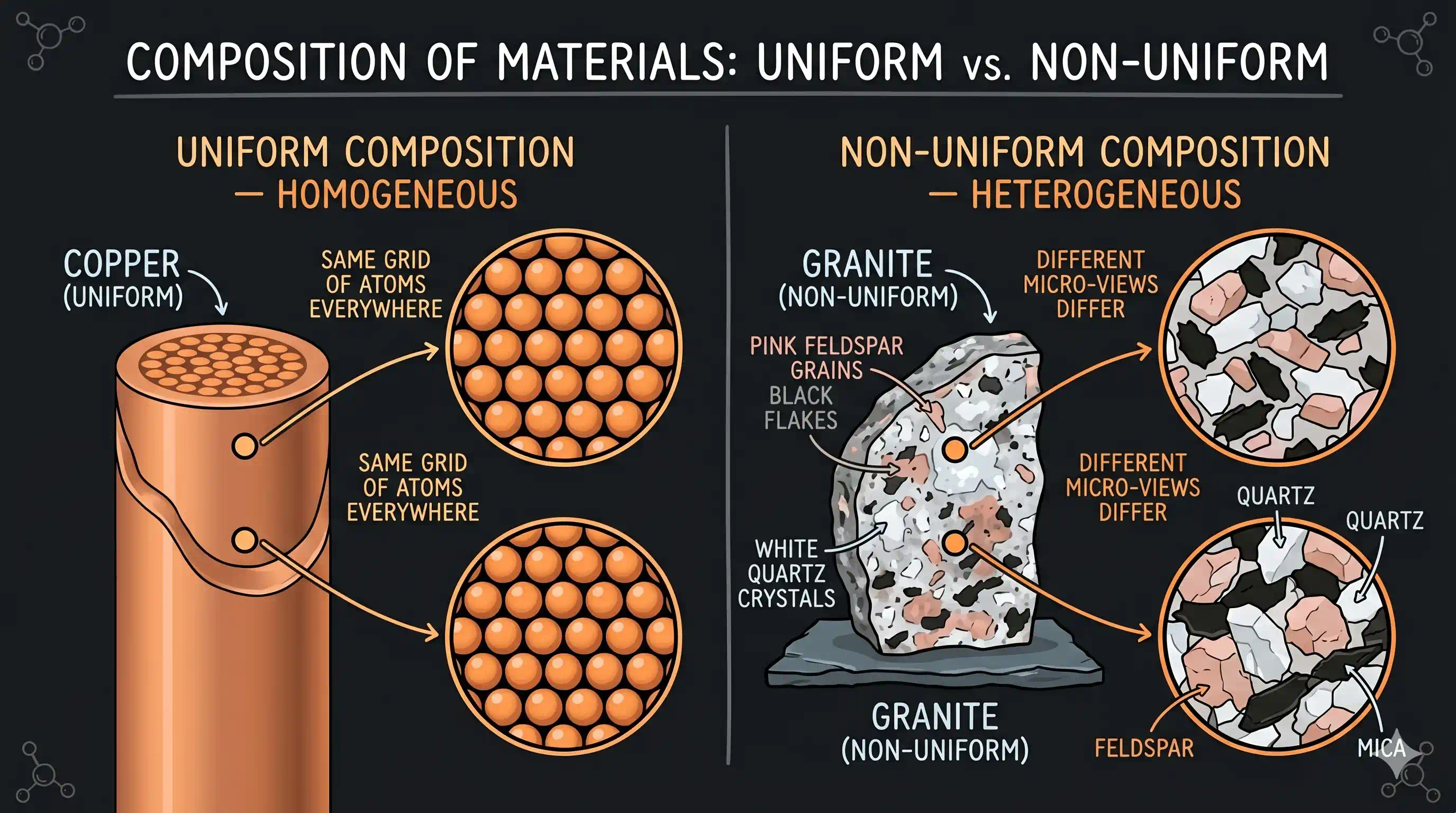
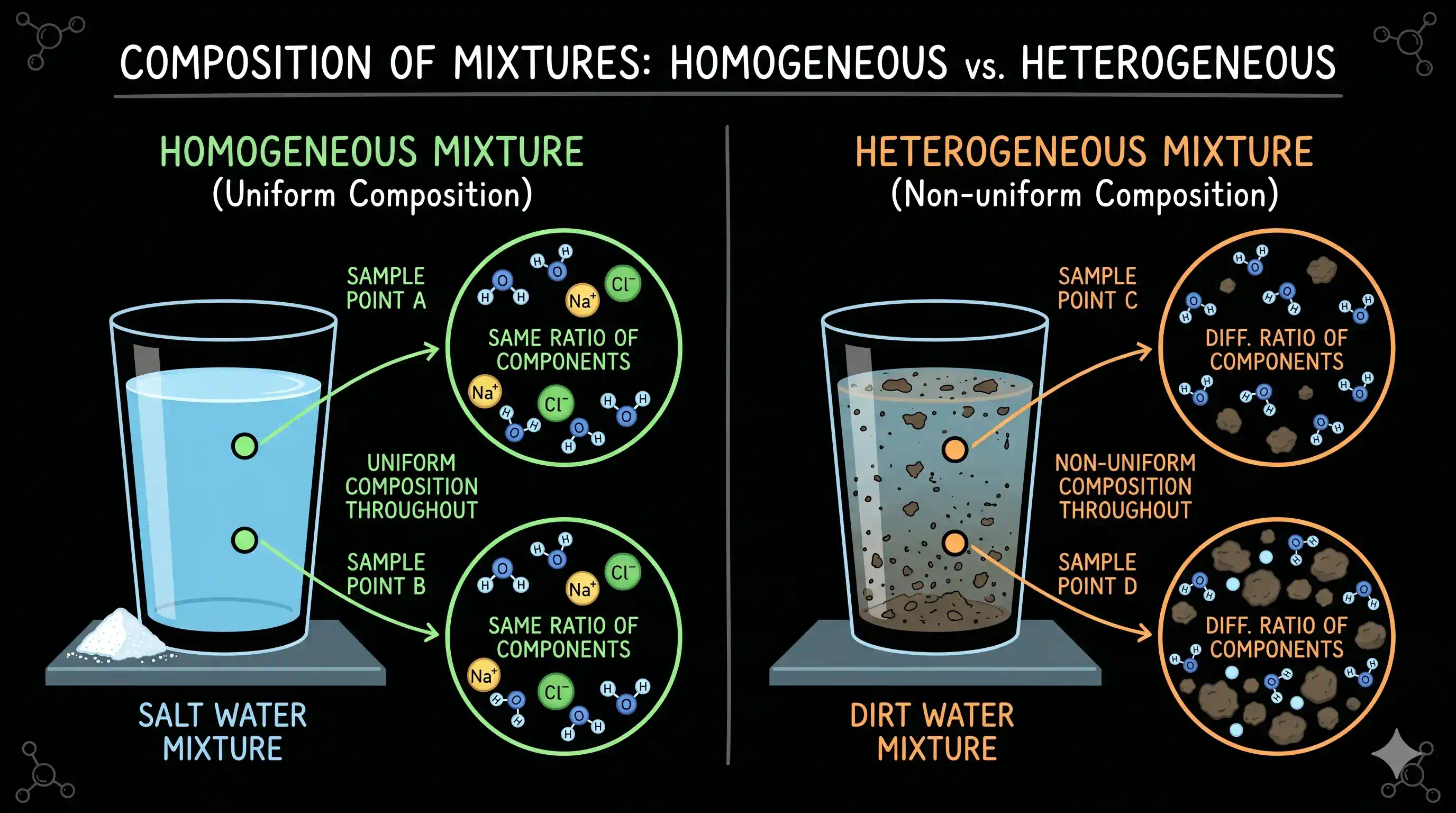
A homogeneous mixture has the same composition throughout — uniform, identical at every point. Take a sample from the top, middle, or bottom of a saltwater glass: every drop is equally salty. Every cubic metre of air — near the ceiling or the floor — has the same 78% nitrogen, 21% oxygen, 0.9% argon. Brass (the alloy in door handles and musical instruments) looks and behaves identically everywhere — you cannot see the copper and zinc as separate regions even under a microscope. If you cannot identify different parts, it is homogeneous.
A heterogeneous mixture has a composition that changes from part to part. In a handful of granite, you can see white quartz, pink feldspar, and black mica as visibly separate specks. In a glass of salad dressing left to stand, oil floats above vinegar in two distinct layers. In muddy river water, the mud concentrates at the bottom while the water above runs clearer. If you can identify different regions — by eye, by touch, or by chemical test — it is heterogeneous.

Activity 5.1 — See It For Yourself
The fastest way to feel the difference between homogeneous and heterogeneous mixtures is to make three of them yourself. In your science lab, split into three groups and prepare these in clean, transparent glass beakers:
- Group A — add one spatula of common salt to 50 mL of water and stir well.
- Group B — add one spatula of chalk powder to 50 mL of water and stir well.
- Group C — add a few drops of milk to 50 mL of water and stir well.
Now look closely. In Group A's beaker the salt has vanished — you cannot see a single grain. In Group B you can clearly see chalk floating around, and given some time, it settles at the bottom. Group C looks milky-white and uniform, almost like a solution.
Now shine a laser pointer through each beaker from the side. What do you observe?
- In Group A (salt water), the laser passes straight through. You cannot see the path of the beam from the side.
- In Group B (chalk water), the path of the laser is clearly visible — the suspended chalk particles scatter the light.
- In Group C (milk water), the path of the laser is also visible, even though the mixture looked perfectly uniform to your eye.
Surprised? You should be. The salt solution is homogeneous — the salt has dissolved at the level of individual particles, far smaller than the wavelength of light, so the laser sees nothing to scatter off. The chalk water is openly heterogeneous. But the milk-water — which looked like a clean solution — turns out to be hiding particles big enough to scatter light. You will meet that hidden middle category on the next page.
Finally, set up a filter paper and pour each mixture through it. The chalk gets caught on the paper. The salt water and the milk water both pass through — but for very different reasons, as you will soon see.

Safety First
Never look directly into the laser beam — even a low-power laser pointer can cause permanent eye damage. Always shine the laser horizontally through the side of the beaker, and observe its path from a safe angle.
What About Oil and Water?
Pour a little oil into a glass of water and you don't even need a laser — the oil simply refuses to mix. It floats on top as a separate yellow layer. Two liquids that refuse to dissolve into each other like this are called immiscible. Sand and water behave similarly, and so do iron filings and sulfur powder. Heterogeneous mixtures come in many shapes — sometimes the components are visibly separate, sometimes they are too small for the eye to catch but still scatter light. Stay with this — it's the doorway into solutions, suspensions, and colloids.
Q1.Which of the following is a heterogeneous mixture?
When you stir sugar into tea, it completely disappears — every sip is equally sweet. But stir sand into water and the sand is still visible, and settles to the bottom if you wait. What do you think is the fundamental difference between these two mixtures?
24-carat gold is pure gold — but pure gold is so soft it bends under finger pressure. 22-carat gold (91.7% gold + 8.3% silver or copper) is harder, more durable, and far more practical. The gold jewellery most Indians wear daily is a homogeneous mixture — a solid solution — where you cannot tell the metals apart even under a microscope. The entire history of metallurgy is the art of making homogeneous mixtures.

Mixtures are normally classified into two types — homogeneous and heterogeneous. To understand what these terms mean, we first need to understand what composition means for a mixture.
Composition is simply the answer to two questions: what is a mixture made of, and how much of each component is present? Think of a glass of fresh orange juice — its composition includes water, natural sugars, vitamin C, and fruit acids, each in some proportion. Squeeze more orange into one glass, and its composition changes, even though both glasses look like juice.
Now comes the crucial question: is that composition the same at every point, or does it vary from place to place?
A homogeneous mixture has the same composition throughout — uniform, identical at every point. Take a sample from the top, middle, or bottom of a saltwater glass: every drop is equally salty. Every cubic metre of air — near the ceiling or the floor — has the same 78% nitrogen, 21% oxygen, 0.9% argon. Brass (the alloy in door handles and musical instruments) looks and behaves identically everywhere — you cannot see the copper and zinc as separate regions even under a microscope. If you cannot identify different parts, it is homogeneous.
A heterogeneous mixture has a composition that changes from part to part. In a handful of granite, you can see white quartz, pink feldspar, and black mica as visibly separate specks. In a glass of salad dressing left to stand, oil floats above vinegar in two distinct layers. In muddy river water, the mud concentrates at the bottom while the water above runs clearer. If you can identify different regions — by eye, by touch, or by chemical test — it is heterogeneous.



Activity 5.1 — See It For Yourself
The fastest way to feel the difference between homogeneous and heterogeneous mixtures is to make three of them yourself. In your science lab, split into three groups and prepare these in clean, transparent glass beakers:
- Group A — add one spatula of common salt to 50 mL of water and stir well.
- Group B — add one spatula of chalk powder to 50 mL of water and stir well.
- Group C — add a few drops of milk to 50 mL of water and stir well.
Now look closely. In Group A's beaker the salt has vanished — you cannot see a single grain. In Group B you can clearly see chalk floating around, and given some time, it settles at the bottom. Group C looks milky-white and uniform, almost like a solution.
Now shine a laser pointer through each beaker from the side. What do you observe?
- In Group A (salt water), the laser passes straight through. You cannot see the path of the beam from the side.
- In Group B (chalk water), the path of the laser is clearly visible — the suspended chalk particles scatter the light.
- In Group C (milk water), the path of the laser is also visible, even though the mixture looked perfectly uniform to your eye.
Surprised? You should be. The salt solution is homogeneous — the salt has dissolved at the level of individual particles, far smaller than the wavelength of light, so the laser sees nothing to scatter off. The chalk water is openly heterogeneous. But the milk-water — which looked like a clean solution — turns out to be hiding particles big enough to scatter light. You will meet that hidden middle category on the next page.
Finally, set up a filter paper and pour each mixture through it. The chalk gets caught on the paper. The salt water and the milk water both pass through — but for very different reasons, as you will soon see.

Safety First
Never look directly into the laser beam — even a low-power laser pointer can cause permanent eye damage. Always shine the laser horizontally through the side of the beaker, and observe its path from a safe angle.
What About Oil and Water?
Pour a little oil into a glass of water and you don't even need a laser — the oil simply refuses to mix. It floats on top as a separate yellow layer. Two liquids that refuse to dissolve into each other like this are called immiscible. Sand and water behave similarly, and so do iron filings and sulfur powder. Heterogeneous mixtures come in many shapes — sometimes the components are visibly separate, sometimes they are too small for the eye to catch but still scatter light. Stay with this — it's the doorway into solutions, suspensions, and colloids.
Q1.Which of the following is a heterogeneous mixture?