Concentration of Solutions
Mass%, volume%, and how to calculate them

If you keep adding salt to a glass of water, does there come a point where no more salt will dissolve? What do you think happens to the extra salt — and to the solution itself?
Oral Rehydration Solution (ORS) must contain exactly 2.6 g NaCl, 2.9 g sodium citrate, 1.5 g KCl, and 13.5 g glucose per litre of water. Too little sodium = ineffective. Too much sodium = dangerous. The precise concentration is what makes it work. Indian scientist Dr. Dilip Mahalanabis (Padma Vibhushan) used this solution during the 1971 cholera epidemic — saving thousands of lives with a calculated mixture.
Meet the Scientist — Dr. Dilip Mahalanabis (1934–2022)

Behind the boring word concentration sits one of India's greatest medical stories.
What is Concentration?
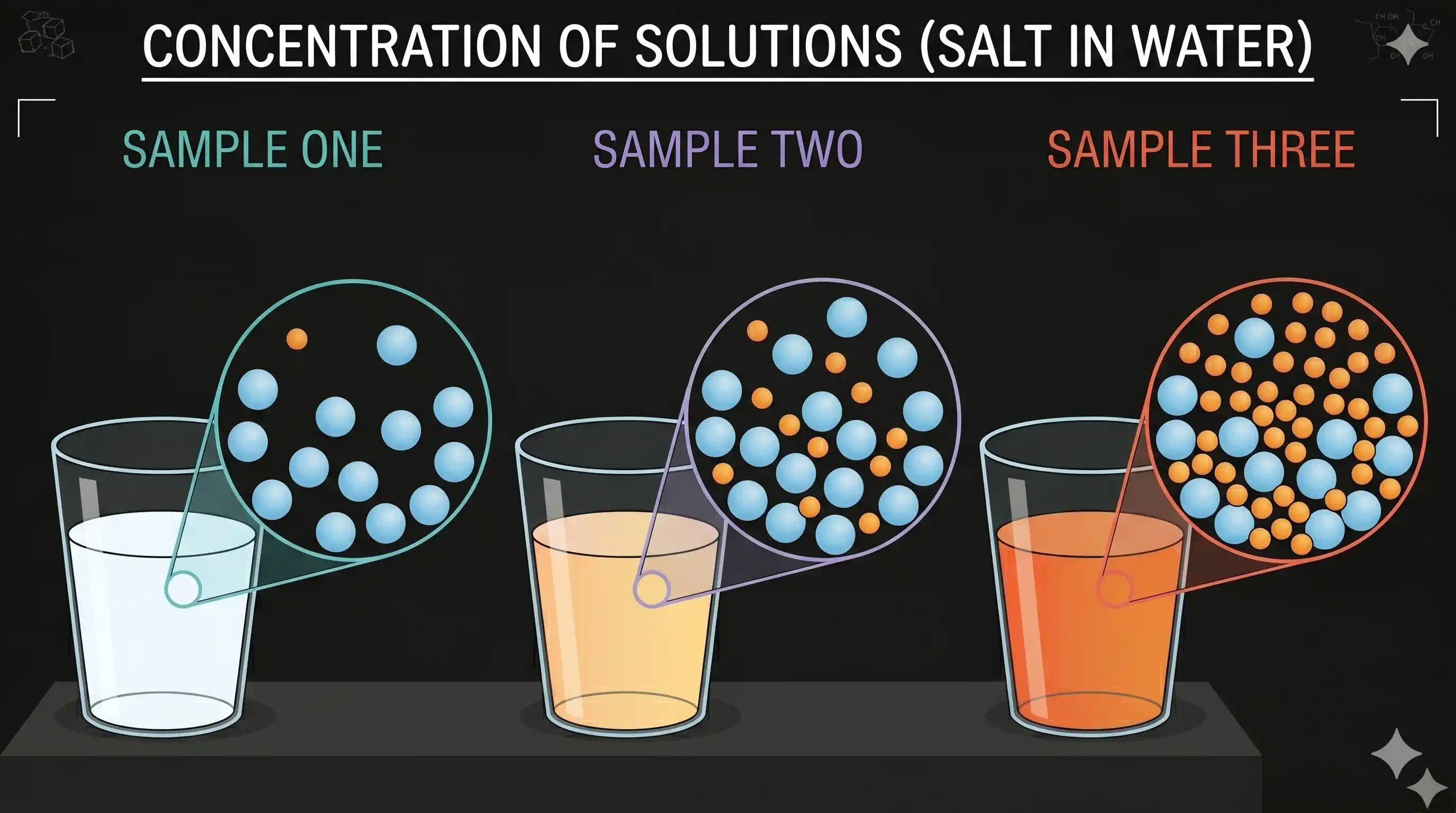
Concentration tells us how much solute is dissolved in a given amount of solution or solvent. A concentrated solution has a lot of solute; a dilute solution has little.
There are several ways to express concentration:
Mass Percentage (% w/w)
Volume Percentage (% v/v)
A Third Way — Mass by Volume Percentage (% m/v)
Sometimes the solute is a solid, but the solution is mostly liquid — and weighing the liquid every time is a hassle. In hospitals and chemistry labs, it is much easier to measure the volume of the final solution. For these cases we use Mass by Volume Percentage, written as % m/v (or % w/v). It tells you how many grams of solute are present in 100 mL of the solution.
You will see it on every glucose drip in a hospital — "5% m/v glucose" means 5 g of glucose dissolved in enough water to make 100 mL of solution.
Mass by Volume Percentage (% m/v)
Used when the solute is solid and the solution is a liquid — common for medicines, IV drips, and lab reagents.
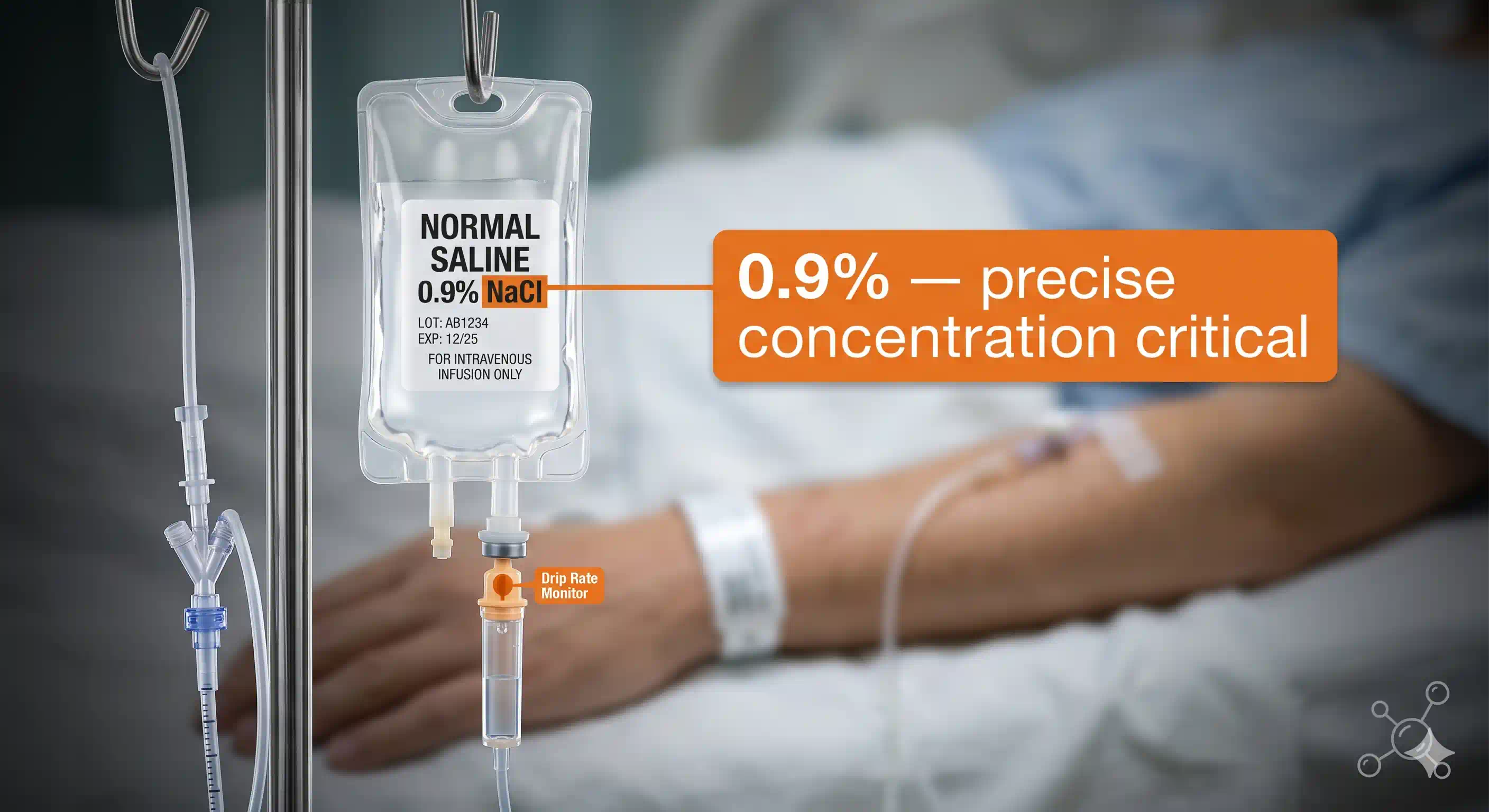
The Saline Drip — A Tiny but Critical 0.9%
Look closely at any saline drip in a hospital and you will see the words 0.9% m/v sodium chloride. That means 0.9 g of common salt dissolved in every 100 mL of solution.
Pause and Ponder
Try these three before moving on:
Where You See This Every Day
Getting concentration exactly right is a matter of life and death in several fields:
- Intravenous (IV) Drips in Hospitals — Normal saline is exactly 0.9 % w/v NaCl. A solution even 20 % above this osmotic concentration can cause cells to shrink (crenation); below it, cells swell and burst. Hospital pharmacists calculate concentration for every drip.
- ORS Sachets — The WHO-recommended formula contains exactly 2.6 g NaCl, 2.9 g trisodium citrate, 1.5 g KCl, and 13.5 g glucose per litre. The glucose: sodium ratio activates a specific gut transporter. Too concentrated = doesn't work; too dilute = doesn't hydrate fast enough.
- Swimming Pool Chlorination — Pool water must be maintained at 1–3 ppm free chlorine. Above 5 ppm, it irritates eyes and skin. Below 0.5 ppm, bacteria and algae grow. The pool operator calculates mass per volume of chlorine daily.
- Fertilizer Application — Farmers must dissolve urea at the correct concentration for drip irrigation. Too concentrated burns roots (chemical scorching); too dilute gives no benefit.

A student prepares a "10% salt solution" by dissolving 10 g of salt in 100 g of water. Her classmate says she made a mistake — a correct 10% solution needs 10 g in only 90 g of water. Who is right?
💉 Real-World Impact
During India's Covid-19 pandemic, a critical shortage of oxygen was matched by another crisis: incorrect IV fluid concentrations in overloaded hospitals. Pharmacy teams working with military precision to calculate concentrations under stress saved countless lives. Concentration chemistry is not abstract — it is urgent.
Q1.Which formula correctly defines mass percentage (w/w%) of a solute in a solution?

If you keep adding salt to a glass of water, does there come a point where no more salt will dissolve? What do you think happens to the extra salt — and to the solution itself?
Oral Rehydration Solution (ORS) must contain exactly 2.6 g NaCl, 2.9 g sodium citrate, 1.5 g KCl, and 13.5 g glucose per litre of water. Too little sodium = ineffective. Too much sodium = dangerous. The precise concentration is what makes it work. Indian scientist Dr. Dilip Mahalanabis (Padma Vibhushan) used this solution during the 1971 cholera epidemic — saving thousands of lives with a calculated mixture.
Meet the Scientist — Dr. Dilip Mahalanabis (1934–2022)

Behind the boring word concentration sits one of India's greatest medical stories.
What is Concentration?

Concentration tells us how much solute is dissolved in a given amount of solution or solvent. A concentrated solution has a lot of solute; a dilute solution has little.
There are several ways to express concentration:
Mass Percentage (% w/w)
Volume Percentage (% v/v)
A Third Way — Mass by Volume Percentage (% m/v)
Sometimes the solute is a solid, but the solution is mostly liquid — and weighing the liquid every time is a hassle. In hospitals and chemistry labs, it is much easier to measure the volume of the final solution. For these cases we use Mass by Volume Percentage, written as % m/v (or % w/v). It tells you how many grams of solute are present in 100 mL of the solution.
You will see it on every glucose drip in a hospital — "5% m/v glucose" means 5 g of glucose dissolved in enough water to make 100 mL of solution.
Mass by Volume Percentage (% m/v)
Used when the solute is solid and the solution is a liquid — common for medicines, IV drips, and lab reagents.
The Saline Drip — A Tiny but Critical 0.9%
Look closely at any saline drip in a hospital and you will see the words 0.9% m/v sodium chloride. That means 0.9 g of common salt dissolved in every 100 mL of solution.
Pause and Ponder
Try these three before moving on:
Where You See This Every Day
Getting concentration exactly right is a matter of life and death in several fields:
- Intravenous (IV) Drips in Hospitals — Normal saline is exactly 0.9 % w/v NaCl. A solution even 20 % above this osmotic concentration can cause cells to shrink (crenation); below it, cells swell and burst. Hospital pharmacists calculate concentration for every drip.
- ORS Sachets — The WHO-recommended formula contains exactly 2.6 g NaCl, 2.9 g trisodium citrate, 1.5 g KCl, and 13.5 g glucose per litre. The glucose: sodium ratio activates a specific gut transporter. Too concentrated = doesn't work; too dilute = doesn't hydrate fast enough.
- Swimming Pool Chlorination — Pool water must be maintained at 1–3 ppm free chlorine. Above 5 ppm, it irritates eyes and skin. Below 0.5 ppm, bacteria and algae grow. The pool operator calculates mass per volume of chlorine daily.
- Fertilizer Application — Farmers must dissolve urea at the correct concentration for drip irrigation. Too concentrated burns roots (chemical scorching); too dilute gives no benefit.

A student prepares a "10% salt solution" by dissolving 10 g of salt in 100 g of water. Her classmate says she made a mistake — a correct 10% solution needs 10 g in only 90 g of water. Who is right?
Q1.Which formula correctly defines mass percentage (w/w%) of a solute in a solution?