Distillation
The science behind purification
Sailors used to die of thirst surrounded by ocean water. Sea water makes you sicker the more you drink. But the ocean has no shortage of water. What would you need to do to make it drinkable?
The earliest evidence of distillation apparatus has been found at Taxila (modern Pakistan, ancient India) — terracotta stills dating to around 3000 BCE, used to distil fermented beverages. The Arthashastra (Kautilya, ~300 BCE) describes distillation of medicinal preparations in detail. India was distilling substances more than 2,000 years before the concept became central to European chemistry.
The Principle of Distillation
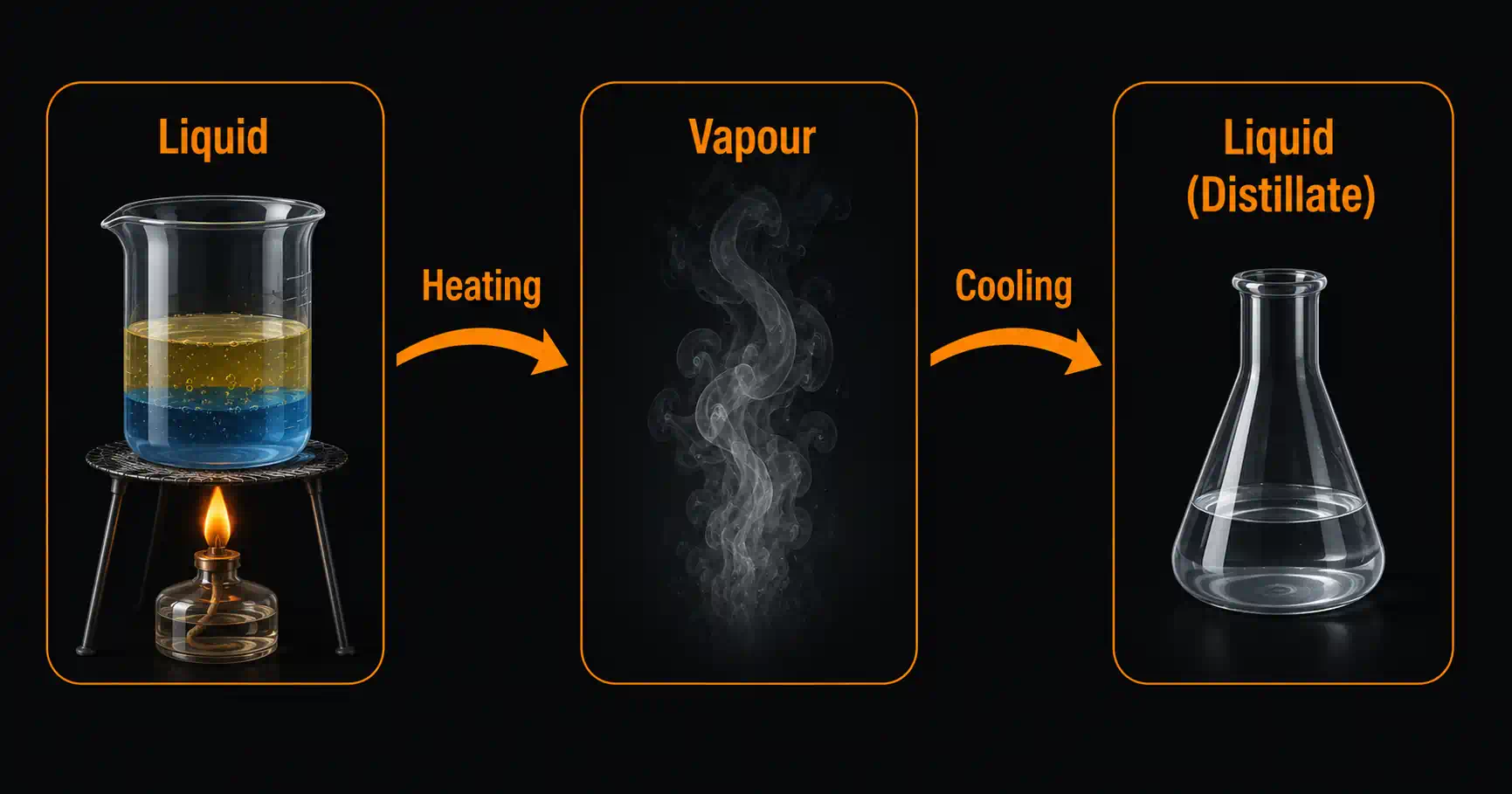
Distillation separates a mixture of liquids (or a dissolved solid from a liquid) using differences in their boiling points.
Basic steps:
- The mixture is heated in a flask
- The component with the lower boiling point vaporises first
- The vapour travels into a condenser (cooled tube)
- The condenser cools the vapour back to liquid — condensate/distillate collects in a receiving flask
- The component with the higher boiling point remains behind
Example: Separating water from a salt solution — water (bp 100 °C) evaporates, condenses, and is collected pure. Salt remains in the flask.

A Worked Example — Separating Acetone and Water
Imagine a student in a chemistry lab accidentally mixes a beaker of acetone (a clear, sharp-smelling liquid used in nail-polish remover) with water. The two are completely miscible — they have dissolved into each other and look like a single liquid. How do we get them apart again?
The trick is that the two liquids boil at very different temperatures:
- Acetone boils at about 56 °C.
- Water boils at 100 °C.
If we heat the mixture gently, acetone vaporises first while the water is still happily liquid. The acetone vapour rises, hits a cold condenser, turns back into pure liquid acetone, and drips into a clean flask. The water, meanwhile, stays behind in the original flask. Two pure liquids, separated.
The 25 °C rule. Distillation works cleanly only when the two liquids' boiling points differ by at least about 25 °C. If the difference is smaller, both liquids start vaporising together and the separation gets messy. (For those harder cases — boiling points closer than 25 °C — chemists use a fancier version called fractional distillation, which you'll meet at the bottom of this page.)
Loading simulator…
Applications of distillation in India:
- Water purification: Distilled water for medical use, batteries, and laboratories
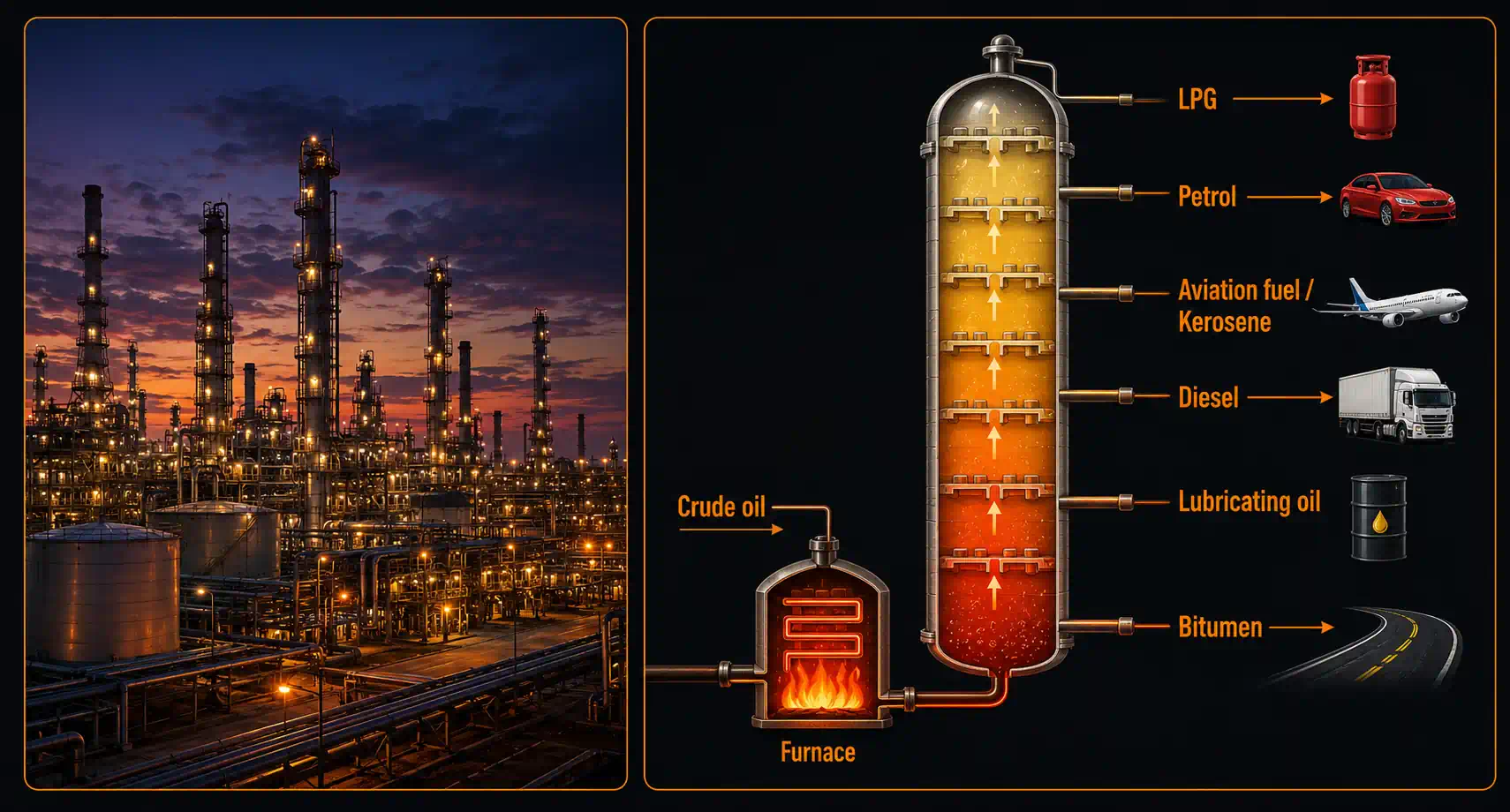
- Petroleum refining: Crude oil is fractionally distilled at Indian Oil refineries to produce LPG, petrol, diesel, kerosene, and bitumen
- Perfumery: Rose water (gulab jal) is produced by steam distillation of rose petals — a centuries-old Indian practice
- Industrial alcohol: Fermented molasses is distilled in sugar mills across Maharashtra and UP
- Essential oils: Distillation of sandalwood, lavender, and eucalyptus for pharmaceuticals and cosmetics
India's Scientific Contributions — The Smell of Wet Earth
Mitti ka Ittar — Bottling the Smell of First Rain

Have you ever noticed the gentle, earthy smell that rises from the soil after the first rain of the season? In Kannauj, a town in Uttar Pradesh, this fragrance has been bottled into a famous natural perfume called Mitti ka Ittar — literally, the perfume of earth.
Ready to Go Beyond — How Crude Oil Becomes LPG, Petrol, and Diesel
Drive past a petroleum refinery at night and you'll see a forest of giant towers lit up like a small city. Inside those towers, simple distillation alone wouldn't work — because crude oil is a mixture of dozens of liquids whose boiling points lie within a narrow range, much closer than 25 °C apart. Time for a stronger tool.
A student says she can purify drinking water from a salt-water mixture by distillation. A classmate says distillation won't work because "salt dissolves in water — you can't just boil it out." Who is right, and what is the flaw in the classmate's reasoning?
Where You See This Every Day
Distillation is one of the most economically important separation techniques in the world:
- Municipal Drinking Water — Cities like Chennai, which faces acute water scarcity, use large-scale desalination plants that are essentially industrial distillation units. The Nemmeli Desalination Plant (100 MLD capacity) distils seawater to provide freshwater to 5 lakh residents daily.
- Petroleum Refining — The Jamnagar Refinery in Gujarat (Reliance Industries) is the world's largest integrated refinery. It processes ~1.2 million barrels of crude oil per day through fractional distillation columns that are 60+ metres tall, separating petrol, diesel, kerosene, LPG, and bitumen.
- Perfume Industry — Rose, jasmine, and sandalwood essential oils are extracted from flowers and bark by steam distillation (a variant you will study in Class 11). The scent molecules are too delicate to survive direct heating. India's Kannauj district (UP) is the "Perfume Capital of India" and uses traditional deg-bhapka copper distillation stills.
- Traditional Spirits — Coconut toddy from Kerala, mahua from Madhya Pradesh, and rice wine from the Northeast have been distilled for centuries using clay and copper apparatus — a real-world application of vapour pressure differences your ancestors understood empirically.
🏭 Real-World Impact
The Jamnagar Refinery generates over ₹4 lakh crore in annual revenue from a single chemistry process — fractional distillation. Every litre of petrol in your family's vehicle passed through a distillation column. The person who maintains and optimises those columns is a chemical engineer — and their career starts with exactly the concept you just learned.

Q1.Distillation is most useful for separating:
Sailors used to die of thirst surrounded by ocean water. Sea water makes you sicker the more you drink. But the ocean has no shortage of water. What would you need to do to make it drinkable?
The earliest evidence of distillation apparatus has been found at Taxila (modern Pakistan, ancient India) — terracotta stills dating to around 3000 BCE, used to distil fermented beverages. The Arthashastra (Kautilya, ~300 BCE) describes distillation of medicinal preparations in detail. India was distilling substances more than 2,000 years before the concept became central to European chemistry.
The Principle of Distillation
Distillation separates a mixture of liquids (or a dissolved solid from a liquid) using differences in their boiling points.
Basic steps:
- The mixture is heated in a flask
- The component with the lower boiling point vaporises first
- The vapour travels into a condenser (cooled tube)
- The condenser cools the vapour back to liquid — condensate/distillate collects in a receiving flask
- The component with the higher boiling point remains behind
Example: Separating water from a salt solution — water (bp 100 °C) evaporates, condenses, and is collected pure. Salt remains in the flask.

A Worked Example — Separating Acetone and Water
Imagine a student in a chemistry lab accidentally mixes a beaker of acetone (a clear, sharp-smelling liquid used in nail-polish remover) with water. The two are completely miscible — they have dissolved into each other and look like a single liquid. How do we get them apart again?
The trick is that the two liquids boil at very different temperatures:
- Acetone boils at about 56 °C.
- Water boils at 100 °C.
If we heat the mixture gently, acetone vaporises first while the water is still happily liquid. The acetone vapour rises, hits a cold condenser, turns back into pure liquid acetone, and drips into a clean flask. The water, meanwhile, stays behind in the original flask. Two pure liquids, separated.
The 25 °C rule. Distillation works cleanly only when the two liquids' boiling points differ by at least about 25 °C. If the difference is smaller, both liquids start vaporising together and the separation gets messy. (For those harder cases — boiling points closer than 25 °C — chemists use a fancier version called fractional distillation, which you'll meet at the bottom of this page.)

Loading simulator…
Applications of distillation in India:
- Water purification: Distilled water for medical use, batteries, and laboratories
- Petroleum refining: Crude oil is fractionally distilled at Indian Oil refineries to produce LPG, petrol, diesel, kerosene, and bitumen
- Perfumery: Rose water (gulab jal) is produced by steam distillation of rose petals — a centuries-old Indian practice
- Industrial alcohol: Fermented molasses is distilled in sugar mills across Maharashtra and UP
- Essential oils: Distillation of sandalwood, lavender, and eucalyptus for pharmaceuticals and cosmetics
India's Scientific Contributions — The Smell of Wet Earth
Mitti ka Ittar — Bottling the Smell of First Rain

Have you ever noticed the gentle, earthy smell that rises from the soil after the first rain of the season? In Kannauj, a town in Uttar Pradesh, this fragrance has been bottled into a famous natural perfume called Mitti ka Ittar — literally, the perfume of earth.
Ready to Go Beyond — How Crude Oil Becomes LPG, Petrol, and Diesel
Drive past a petroleum refinery at night and you'll see a forest of giant towers lit up like a small city. Inside those towers, simple distillation alone wouldn't work — because crude oil is a mixture of dozens of liquids whose boiling points lie within a narrow range, much closer than 25 °C apart. Time for a stronger tool.

A student says she can purify drinking water from a salt-water mixture by distillation. A classmate says distillation won't work because "salt dissolves in water — you can't just boil it out." Who is right, and what is the flaw in the classmate's reasoning?
Where You See This Every Day
Distillation is one of the most economically important separation techniques in the world:
- Municipal Drinking Water — Cities like Chennai, which faces acute water scarcity, use large-scale desalination plants that are essentially industrial distillation units. The Nemmeli Desalination Plant (100 MLD capacity) distils seawater to provide freshwater to 5 lakh residents daily.
- Petroleum Refining — The Jamnagar Refinery in Gujarat (Reliance Industries) is the world's largest integrated refinery. It processes ~1.2 million barrels of crude oil per day through fractional distillation columns that are 60+ metres tall, separating petrol, diesel, kerosene, LPG, and bitumen.
- Perfume Industry — Rose, jasmine, and sandalwood essential oils are extracted from flowers and bark by steam distillation (a variant you will study in Class 11). The scent molecules are too delicate to survive direct heating. India's Kannauj district (UP) is the "Perfume Capital of India" and uses traditional deg-bhapka copper distillation stills.
- Traditional Spirits — Coconut toddy from Kerala, mahua from Madhya Pradesh, and rice wine from the Northeast have been distilled for centuries using clay and copper apparatus — a real-world application of vapour pressure differences your ancestors understood empirically.
Q1.Distillation is most useful for separating: