Paper Chromatography
Separating the invisible
Two black pens look identical. A detective needs to know if the same pen wrote two different notes. How could you possibly tell them apart — just from the ink — without a microscope?
Think about what "black" ink actually is.
Take a black marker and draw a spot on filter paper. Dip the edge of the paper in water. Watch what happens: the "black" ink separates into a rainbow — blue, red, yellow, violet — as the different dye components travel up the paper at different speeds. Black pen ink is actually a mixture of several coloured dyes. Chromatography reveals what's hidden in plain sight.
Why is it Called *Chromatography*?
The word chromatography comes from two Greek words — chroma (colour) and graphein (to write). Put together, it literally means writing with colour. The technique got its name when scientists first used it in the late 19th century to separate coloured substances like plant pigments and dyes. The name has stuck even though we now use it to separate colourless substances too — drugs, pesticides, even DNA fragments.
How Paper Chromatography Works
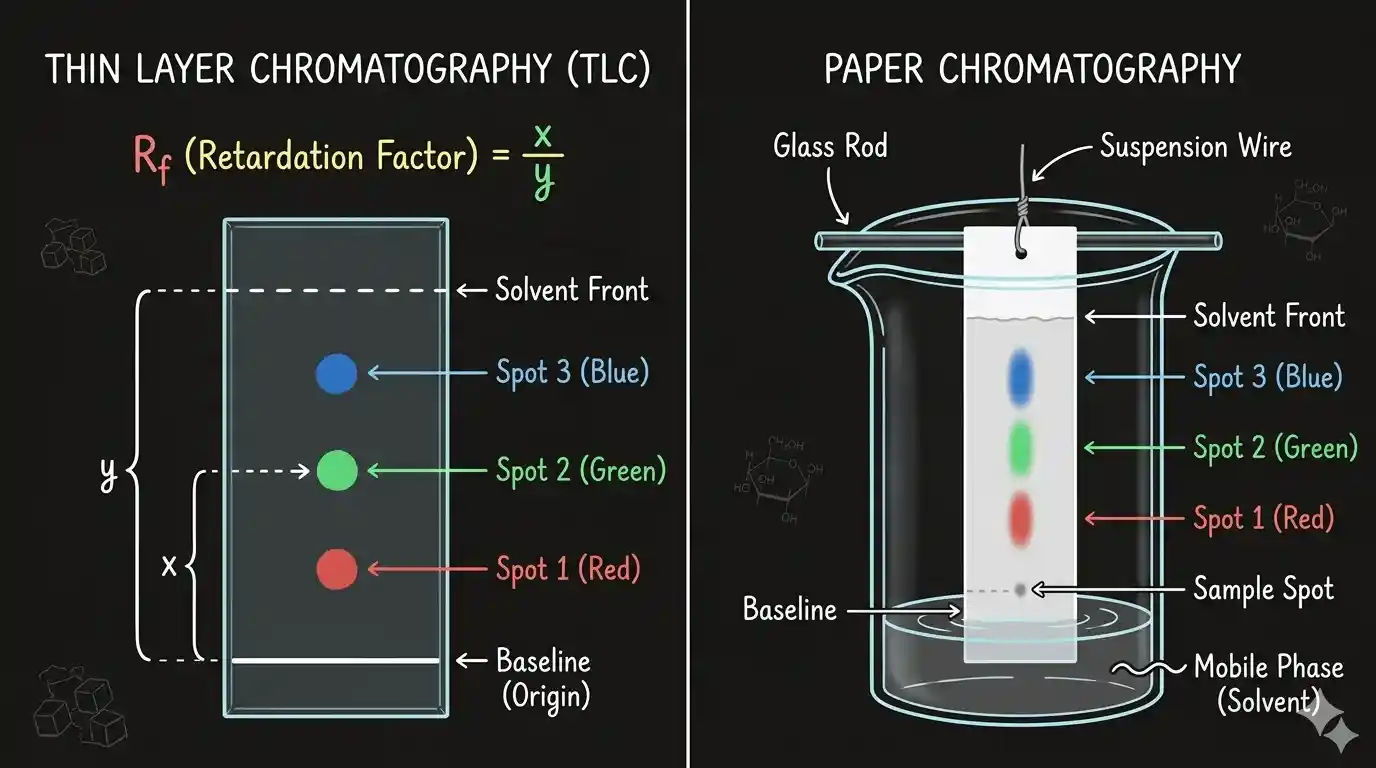
Paper chromatography separates a mixture of substances based on how strongly each component is attracted to the paper (stationary phase) vs. how soluble it is in the solvent (mobile phase).
The two phases:
- Stationary phase: The chromatography paper (which holds water in its fibres)
- Mobile phase: The solvent (water, ethanol, or a mixture) that moves up the paper by capillary action
What happens:
As the solvent travels up the paper, it carries the components of the mixture with it. Components that are more attracted to the paper move slowly. Components that are more soluble in the solvent move quickly. The result: the mixture separates into distinct spots at different heights.
Loading simulator…
Activity 5.5 — See Black Ink Split Into Its Hidden Colours
Here is the original chromatography experiment, simple enough to do in any school lab.
You will need: a strip of chromatographic paper (or filter paper) about 3 cm wide, a black sketch pen, a pencil, water, a tall gas jar (or a measuring cylinder, or a beaker).
Steps:
- Take the strip of paper and, using a pencil (never ink!), draw a straight horizontal line about 2 cm from the bottom edge.
- With a black sketch pen, place a single dark spot on the centre of that pencil line.
- Pour just enough water into the gas jar to make a thin layer at the bottom — about 1 cm deep.
- Stand the paper strip vertically inside the jar so its lower end just dips into the water. The water level must stay below the ink spot, not above it. (If the spot dips into the water, the ink will simply dissolve away — no separation.)
- Now wait. Watch as the water slowly creeps up through the paper.
- As the water passes through the ink spot, the spot starts to fan out into bands of different colours. What looked like plain black will reveal itself as a quiet rainbow.
Why does it work? A black sketch pen ink is a mixture of several coloured dyes. Each dye sticks to the paper differently and travels with the water at its own speed. The faster ones rise higher, the slower ones lag behind — and you end up seeing them as separate bands. Same trick chemists use to detect drugs in blood or pesticides in fruit.
Try Other Solvents and Other Mixtures
Water is just one possible solvent. The choice of solvent depends entirely on what you are trying to separate.
Applications:
- Identifying dyes in food colourings (food safety testing)
- Testing purity of drugs (FSSAI, pharmaceutical labs)
- Forensic analysis — identifying inks on disputed documents
- Testing blood plasma and urine samples
- Identifying plant pigments (chlorophyll a, b, carotenoids) in botany
A student runs paper chromatography on a black water-colour marker and a black permanent marker using water as the solvent. The water-colour ink separates into blue, red, and yellow spots. The permanent marker shows only one black spot that barely moves from the baseline. Why does the permanent marker behave differently?
Pause and Ponder — True or False?
For each statement below, decide whether it is True or False. If false, write the corrected version.
Q1.In paper chromatography, the Rf value of a substance is calculated as:
Where You See This Every Day
Chromatography is so versatile that it appears in settings as different as food labs and crime scenes:
- Food Safety in India — FSSAI (Food Safety and Standards Authority of India) uses chromatography to detect illegal artificial food colours in sweets, snacks, and beverages. Cadmium yellow, Sudan Red, and Rhodamine B are banned but found in street food — they are identified by their unique Rf values in chromatography.
- Anti-Doping in Sports — NADA (National Anti-Doping Agency) tests athletes' urine using High-Performance Liquid Chromatography (HPLC, an advanced version of paper chromatography). Prohibited substances leave a unique chromatographic fingerprint that cannot be masked.
- Forensic Science — The CBI and state forensic labs use chromatography to analyse ink in disputed documents (comparing Rf values of questioned ink with known samples), detect poisons in blood, and identify accelerants in arson cases.
- Blood Plasma Proteins — Hospitals separate blood plasma proteins by chromatography to diagnose conditions like multiple myeloma (abnormal protein bands on the chromatogram are diagnostic).
- Plant Pigments — Every time a leaf changes colour in autumn, it is revealing the chromatographic truth: the green chlorophyll was masking the yellow and orange carotenoids all along.
🔬 Real-World Impact
The ink on your Class 10 Board exam answer sheet can be forensically verified by chromatography. The Supreme Court has upheld cases where disputed signatures were authenticated by ink chromatography analysis. A technique invented in 1903 by a Russian botanist is now admissible evidence in the highest court of the land.

Two black pens look identical. A detective needs to know if the same pen wrote two different notes. How could you possibly tell them apart — just from the ink — without a microscope?
Think about what "black" ink actually is.
Take a black marker and draw a spot on filter paper. Dip the edge of the paper in water. Watch what happens: the "black" ink separates into a rainbow — blue, red, yellow, violet — as the different dye components travel up the paper at different speeds. Black pen ink is actually a mixture of several coloured dyes. Chromatography reveals what's hidden in plain sight.
Why is it Called *Chromatography*?
The word chromatography comes from two Greek words — chroma (colour) and graphein (to write). Put together, it literally means writing with colour. The technique got its name when scientists first used it in the late 19th century to separate coloured substances like plant pigments and dyes. The name has stuck even though we now use it to separate colourless substances too — drugs, pesticides, even DNA fragments.
How Paper Chromatography Works
Paper chromatography separates a mixture of substances based on how strongly each component is attracted to the paper (stationary phase) vs. how soluble it is in the solvent (mobile phase).
The two phases:
- Stationary phase: The chromatography paper (which holds water in its fibres)
- Mobile phase: The solvent (water, ethanol, or a mixture) that moves up the paper by capillary action
What happens:
As the solvent travels up the paper, it carries the components of the mixture with it. Components that are more attracted to the paper move slowly. Components that are more soluble in the solvent move quickly. The result: the mixture separates into distinct spots at different heights.

Loading simulator…
Activity 5.5 — See Black Ink Split Into Its Hidden Colours
Here is the original chromatography experiment, simple enough to do in any school lab.
You will need: a strip of chromatographic paper (or filter paper) about 3 cm wide, a black sketch pen, a pencil, water, a tall gas jar (or a measuring cylinder, or a beaker).
Steps:
- Take the strip of paper and, using a pencil (never ink!), draw a straight horizontal line about 2 cm from the bottom edge.
- With a black sketch pen, place a single dark spot on the centre of that pencil line.
- Pour just enough water into the gas jar to make a thin layer at the bottom — about 1 cm deep.
- Stand the paper strip vertically inside the jar so its lower end just dips into the water. The water level must stay below the ink spot, not above it. (If the spot dips into the water, the ink will simply dissolve away — no separation.)
- Now wait. Watch as the water slowly creeps up through the paper.
- As the water passes through the ink spot, the spot starts to fan out into bands of different colours. What looked like plain black will reveal itself as a quiet rainbow.
Why does it work? A black sketch pen ink is a mixture of several coloured dyes. Each dye sticks to the paper differently and travels with the water at its own speed. The faster ones rise higher, the slower ones lag behind — and you end up seeing them as separate bands. Same trick chemists use to detect drugs in blood or pesticides in fruit.
Try Other Solvents and Other Mixtures
Water is just one possible solvent. The choice of solvent depends entirely on what you are trying to separate.
Applications:
- Identifying dyes in food colourings (food safety testing)
- Testing purity of drugs (FSSAI, pharmaceutical labs)
- Forensic analysis — identifying inks on disputed documents
- Testing blood plasma and urine samples
- Identifying plant pigments (chlorophyll a, b, carotenoids) in botany
A student runs paper chromatography on a black water-colour marker and a black permanent marker using water as the solvent. The water-colour ink separates into blue, red, and yellow spots. The permanent marker shows only one black spot that barely moves from the baseline. Why does the permanent marker behave differently?
Pause and Ponder — True or False?
For each statement below, decide whether it is True or False. If false, write the corrected version.
Q1.In paper chromatography, the Rf value of a substance is calculated as:
Where You See This Every Day
Chromatography is so versatile that it appears in settings as different as food labs and crime scenes:
- Food Safety in India — FSSAI (Food Safety and Standards Authority of India) uses chromatography to detect illegal artificial food colours in sweets, snacks, and beverages. Cadmium yellow, Sudan Red, and Rhodamine B are banned but found in street food — they are identified by their unique Rf values in chromatography.
- Anti-Doping in Sports — NADA (National Anti-Doping Agency) tests athletes' urine using High-Performance Liquid Chromatography (HPLC, an advanced version of paper chromatography). Prohibited substances leave a unique chromatographic fingerprint that cannot be masked.
- Forensic Science — The CBI and state forensic labs use chromatography to analyse ink in disputed documents (comparing Rf values of questioned ink with known samples), detect poisons in blood, and identify accelerants in arson cases.
- Blood Plasma Proteins — Hospitals separate blood plasma proteins by chromatography to diagnose conditions like multiple myeloma (abnormal protein bands on the chromatogram are diagnostic).
- Plant Pigments — Every time a leaf changes colour in autumn, it is revealing the chromatographic truth: the green chlorophyll was masking the yellow and orange carotenoids all along.